Introduction
In the realm of medical device software development, compliance with regulatory standards is critical to ensuring patient safety. Adherence to these standards and effective risk management are essential; failure to comply can lead to significant risks for patient safety. By exploring best practices in this field, developers can gain invaluable insights into optimizing compliance, enhancing product quality, and safeguarding sensitive data. As regulations change, developers must adapt their strategies to mitigate risks effectively.
Understand Regulatory Standards for Medical Device Software
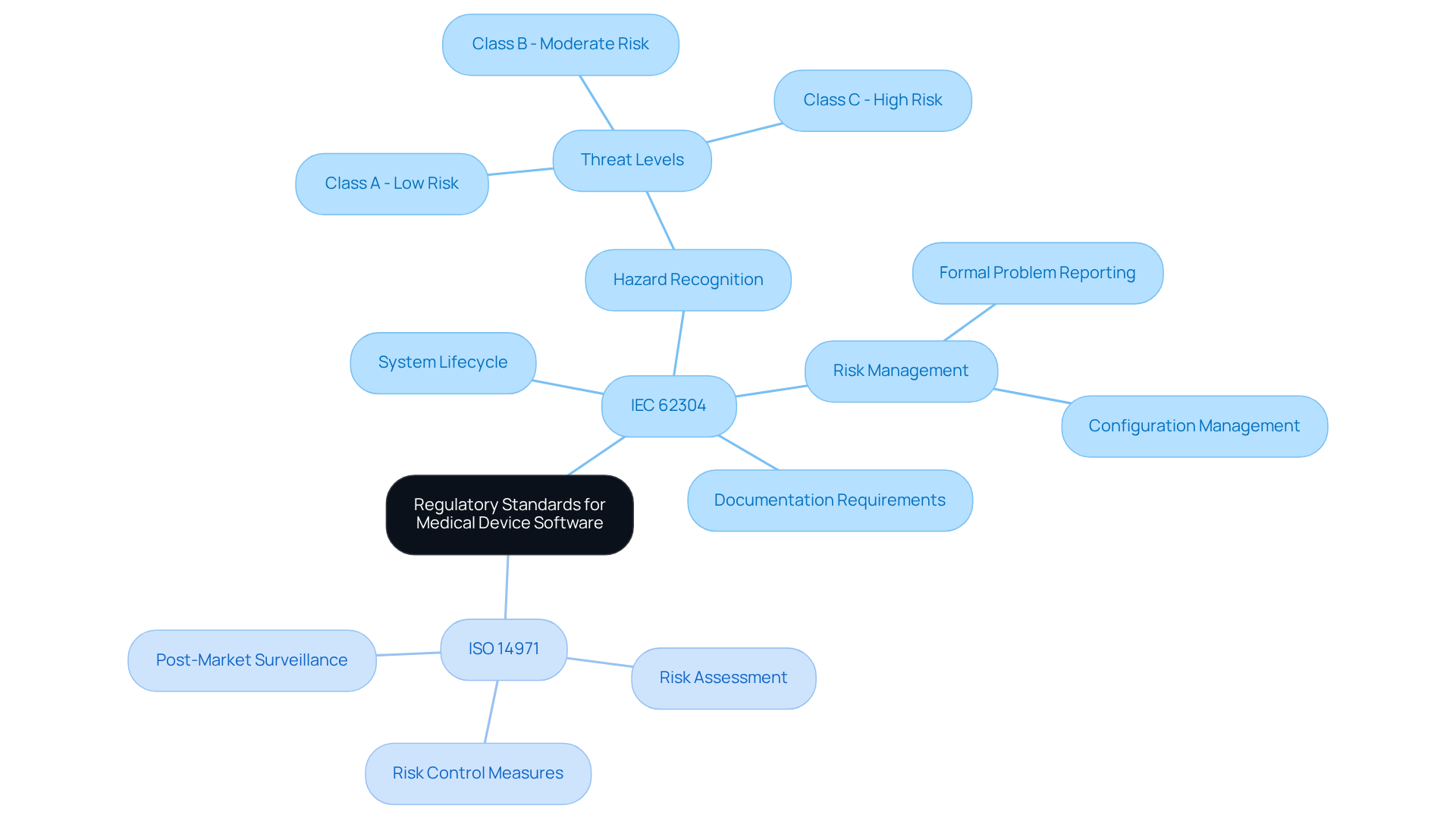
A comprehensive understanding of regulatory standards such as IEC 62304 and ISO 14971 is critical for successful software development for medical devices. These standards outline the requirements for the system lifecycle, encompassing design, creation, testing, and maintenance phases. Adhering to these regulations enhances compliance while significantly improving the quality and safety of software development for medical devices.
For instance, IEC 62304 specifies the essential procedures for medical device program creation, requiring that all possible hazards be recognized and addressed throughout the program’s lifecycle. Furthermore, applications must be categorized into threat levels (Class A, B, and C) based on possible damage, which is essential for comprehending compliance needs.
Without early engagement with regulatory agencies, developers may struggle to keep up with evolving compliance requirements. As highlighted by the U.S. Food and Drug Administration, effective risk management in software development for medical devices following ISO 14971 is essential for ensuring that programs meet safety and quality standards.
Additionally, the FDA’s Computer Program Assurance (CSA) guidance, completed in September 2025, emphasizes the changing regulatory environment and its effects on application creation practices. This proactive strategy for adherence is essential for preserving the integrity of medical technology in a swiftly changing regulatory environment.
It is also crucial to highlight that delays in documentation can hinder the approval process and impact project timelines. The approval for the updated IEC 62304 standard is set to start on May 22, 2026, emphasizing the urgency of compliance. As the regulatory landscape evolves, adherence to these standards will be paramount for maintaining the safety and efficacy of medical technologies.
Implement Effective Risk Management Strategies
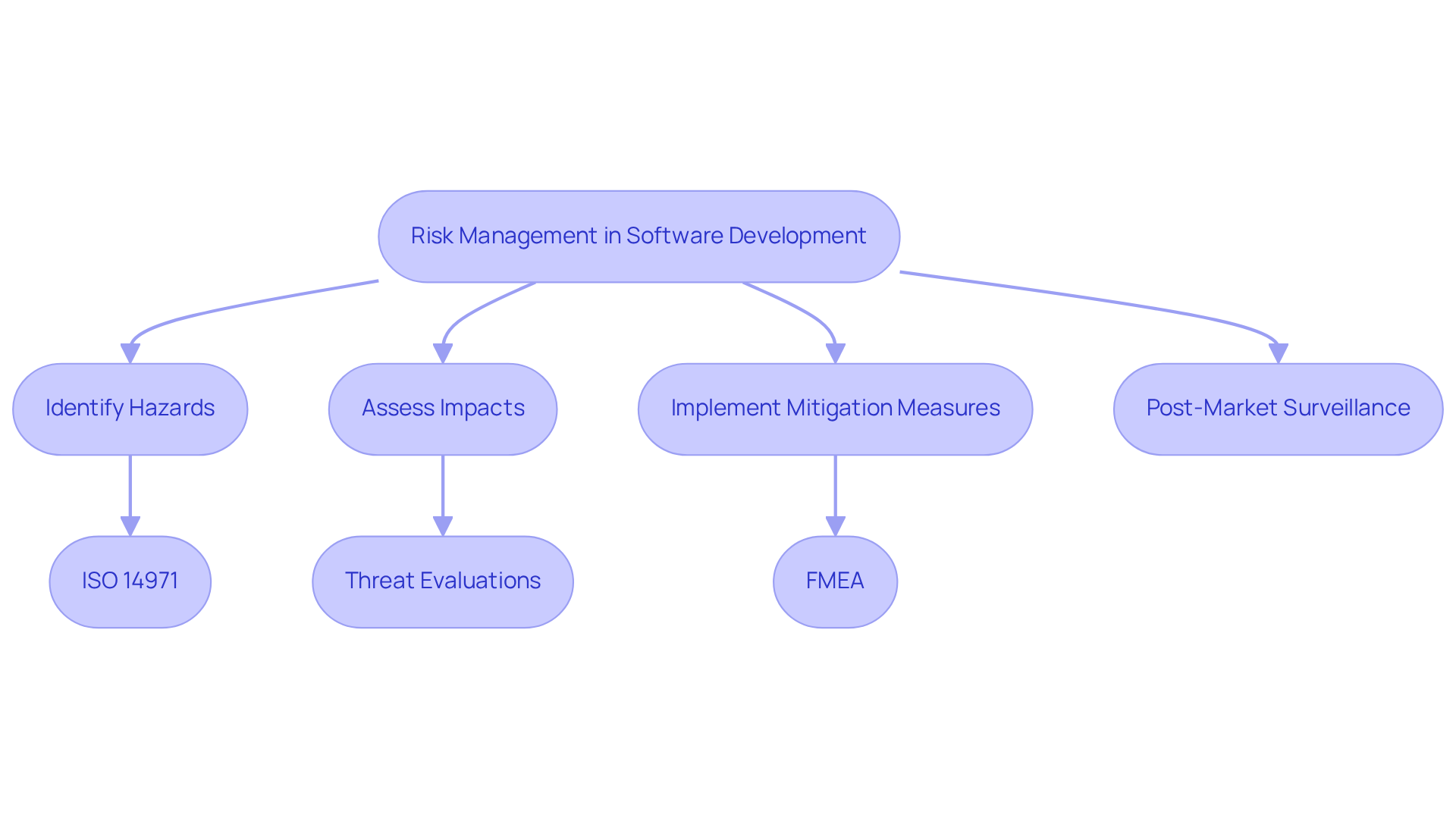
In the realm of software development for medical devices, effective hazard management is not just beneficial; it is essential for ensuring safety and compliance. It involves recognizing potential dangers, assessing their impacts, and implementing measures to mitigate these risks.
Utilizing frameworks such as ISO 14971 enables developers to systematically manage uncertainties effectively. For example, conducting regular threat evaluations and maintaining a management file aids in monitoring identified hazards and their mitigation strategies.
Furthermore, incorporating post-market surveillance into management systems is crucial for ongoing monitoring and enhancement based on user feedback, as highlighted in recent guidelines.
Employing methods like Failure Mode and Effects Analysis (FMEA) provides deeper insights into potential failure points within the system, allowing teams to address these issues proactively.
Integrating risk management at every stage of the application lifecycle in software development for medical devices enhances product safety and ensures compliance with evolving regulatory standards, ultimately safeguarding patient outcomes.
Follow a Structured Software Development Lifecycle
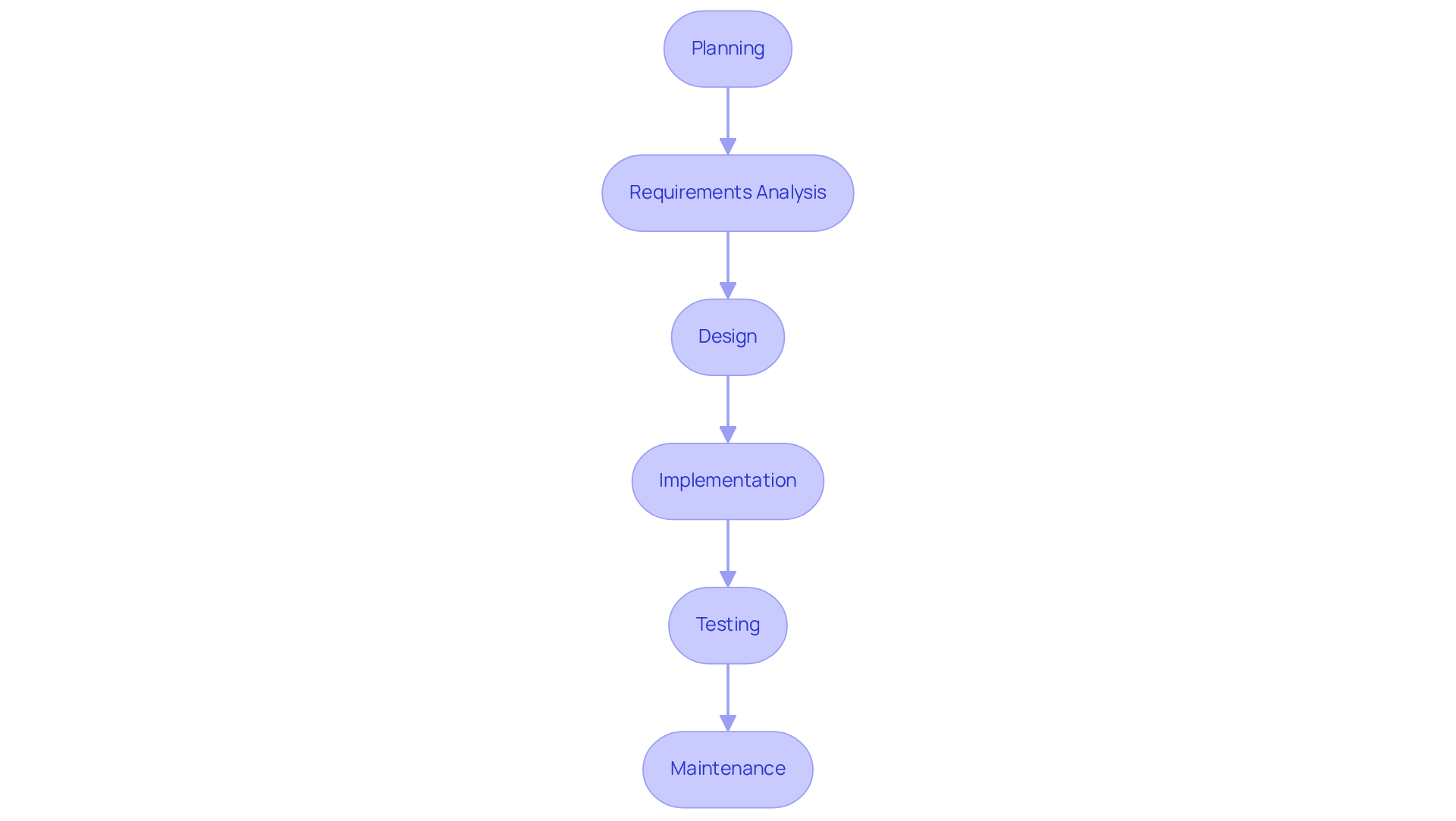
A structured program lifecycle (SDLC) is crucial for the successful software development for medical devices, ensuring compliance and quality throughout the process. This lifecycle encompasses critical phases such as:
- Planning
- Requirements analysis
- Design
- Implementation
- Testing
- Maintenance
Thorough documentation of each phase is essential for ensuring compliance with regulatory standards and maintaining traceability. The V-Model is particularly effective in this context, as it emphasizes verification and validation at every stage of progress. By adhering to a structured SDLC, teams can ensure that all requirements are fulfilled, identify potential issues early, and conduct rigorous testing prior to deployment. Ultimately, neglecting a structured SDLC can lead to significant risks in product safety and efficacy, undermining stakeholder confidence.
Prioritize Cybersecurity and Data Protection
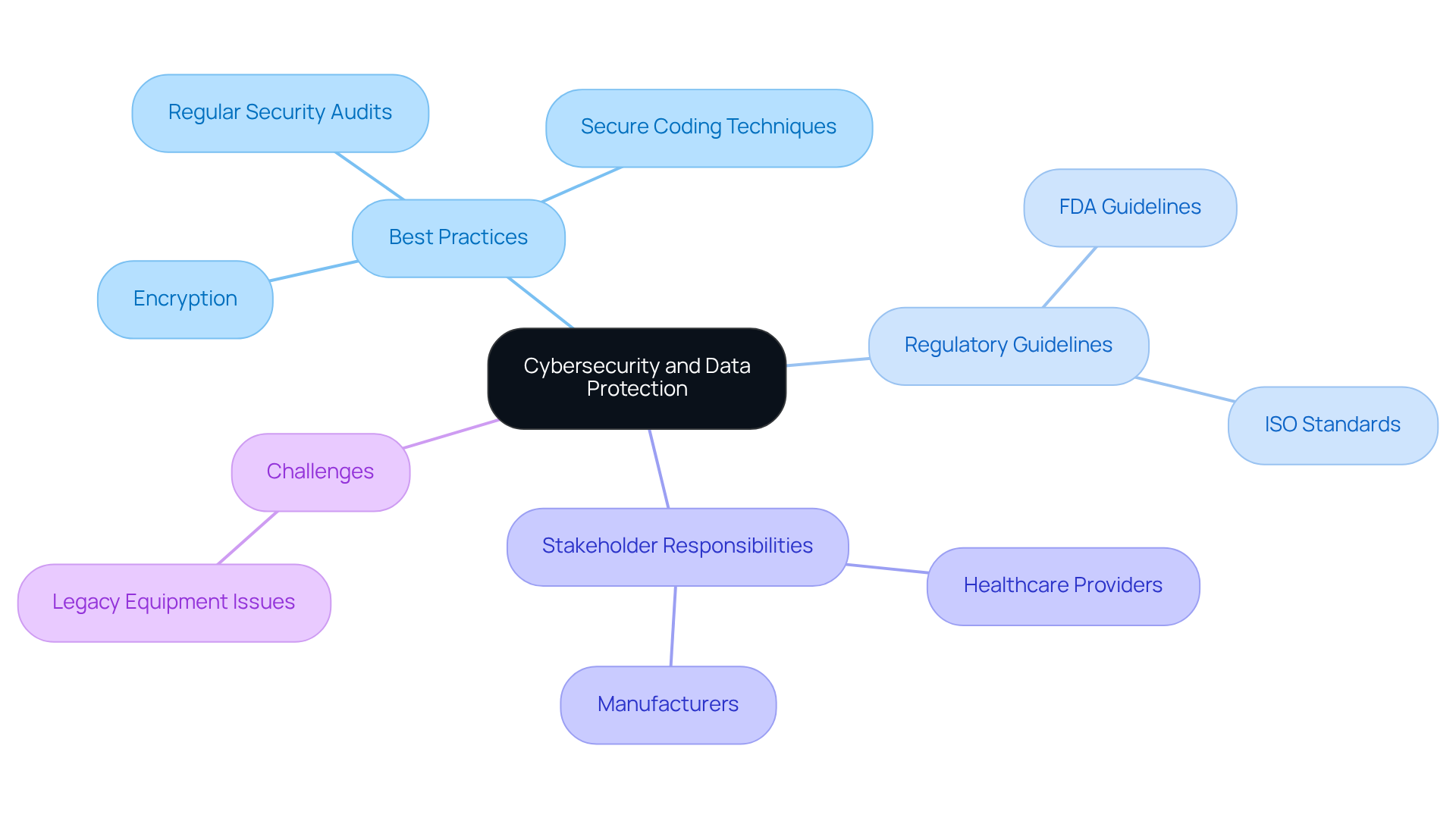
Inadequate cybersecurity measures expose sensitive patient information to significant risks, making it imperative to prioritize cybersecurity in the software development for medical devices. Best practices, including:
- Encryption
- Secure coding techniques
- Regular security audits
significantly mitigate vulnerabilities. Adhering to guidelines from regulatory bodies like the FDA and ISO is essential for establishing a robust cybersecurity framework. The FDA’s recent focus on a risk-based strategy for managing cybersecurity threats throughout the product lifecycle highlights the need for incorporating security measures from the beginning. This proactive strategy enhances protection against breaches and ensures compliance with evolving regulations, thereby safeguarding patient safety.
Furthermore, healthcare providers and manufacturers share a mutual responsibility to ensure the security of equipment throughout its lifecycle. With over 70% of companies reporting moderate to severe financial effects from incidents in the past two years, the urgency for robust cybersecurity measures cannot be overstated. Additionally, the FDA’s new requirements for manufacturers to provide a Software Bill of Materials (SBOM) and manage risks associated with their components underscore the critical need for compliance.
Addressing the challenges presented by legacy medical equipment is also essential, as these older systems often lack sufficient security features, rendering them susceptible to cyber threats. As Phil Englert, director of medical device security at Health-ISAC, emphasizes, securing connected devices is essential not only for data protection but also for ensuring care delivery itself. The evolving landscape of cybersecurity threats demands that both healthcare providers and manufacturers take immediate and collaborative action to protect patient safety and ensure the integrity of medical devices.
Conclusion
The complexities of software development for medical devices demand rigorous adherence to established best practices. Adhering to regulatory standards, implementing effective risk management strategies, following a structured software development lifecycle, and ensuring robust cybersecurity measures are critical components of successful software development for medical devices. These elements work in tandem to create a framework that not only meets regulatory requirements but also enhances the overall quality of medical software.
Key insights emerge, including:
- The necessity of engaging with regulatory bodies early in the development process
- The critical role of risk management frameworks like ISO 14971
- The significance of following a structured SDLC
- The importance of prioritizing cybersecurity to protect sensitive patient information and maintain trust in medical technologies
Each of these practices contributes to a comprehensive approach that safeguards patient outcomes and ensures the integrity of medical devices.
In conclusion, the evolving landscape of medical device software development underscores the urgency for adherence to these best practices. By committing to regulatory compliance, implementing effective risk management, following a structured development process, and prioritizing cybersecurity, developers can not only enhance product safety but also foster innovation in healthcare technology. Ultimately, the future of patient care hinges on the unwavering commitment to these best practices in medical device development.
Frequently Asked Questions
Why is understanding regulatory standards important for medical device software development?
A comprehensive understanding of regulatory standards such as IEC 62304 and ISO 14971 is critical for successful software development as they outline the requirements for the system lifecycle, including design, creation, testing, and maintenance phases. Adhering to these regulations enhances compliance and significantly improves the quality and safety of software development.
What does IEC 62304 specify regarding medical device software?
IEC 62304 specifies essential procedures for medical device program creation, requiring that all possible hazards be recognized and addressed throughout the program’s lifecycle. It also mandates that applications be categorized into threat levels (Class A, B, and C) based on possible damage to understand compliance needs.
What role does ISO 14971 play in medical device software development?
ISO 14971 emphasizes effective risk management in software development for medical devices, which is essential for ensuring that programs meet safety and quality standards as highlighted by the U.S. Food and Drug Administration.
What is the significance of early engagement with regulatory agencies?
Early engagement with regulatory agencies is crucial as it helps developers keep up with evolving compliance requirements, preventing potential struggles during the software development process.
What is the FDA’s Computer Program Assurance (CSA) guidance?
The FDA’s CSA guidance, completed in September 2025, emphasizes the changing regulatory environment and its effects on application creation practices, promoting a proactive strategy for adherence to maintain the integrity of medical technology.
How can documentation delays affect the approval process for medical device software?
Delays in documentation can hinder the approval process and impact project timelines, making timely compliance essential for successful software development.
When is the approval for the updated IEC 62304 standard expected to start?
The approval for the updated IEC 62304 standard is set to start on May 22, 2026, highlighting the urgency of compliance as the regulatory landscape evolves.
List of Sources
- Understand Regulatory Standards for Medical Device Software
- IEC 62304 vs FDA CSA: A Medical Software Compliance Guide | IntuitionLabs (https://intuitionlabs.ai/articles/iec-62304-vs-fda-csa)
- IEC 62304 Update 2026: Key Changes & Compliance Tips (https://lfhregulatory.co.uk/iec-62304-update-2026)
- Medical Devices Compliance & Regulatory News – Newsletter Oct 2025 | Sushvin Consulting (https://sushvin.com/medical-devices-compliance-updates-newsletter-Oct-2025.html)
- What You need to know about IEC 62304: Medical Software Lifecycle (https://securitycompass.com/blog/iec-62304-medical-software-lifecycle)
- Medical Device Regulatory News and Updates (https://pureglobal.com/resources/regulatory-updates)
- Implement Effective Risk Management Strategies
- New FDA Cybersecurity Rules: Key Changes For Software Teams (https://punchthrough.com/new-fda-cybersecurity-rules)
- Implications of the New ISO 14971 Edition for Usability Engineering (https://emergobyul.com/news/implications-new-iso-14971-edition-usability-engineering)
- An investigation of the current status quo of ISO 14971 risk management challenges in the medical device industry (https://sciencedirect.com/org/science/article/pii/S0265671X26000113)
- Best Practices for Medical Device Software Validation and Risk Management (https://webinarwaves.com/blog/best-practices-for-medical-device-software-validation-and-risk-management)
- Medical Device Vendor Risk Management: FDA Compliance and Patient Safety Best Practices | Censinet, Inc. (https://censinet.com/perspectives/medical-device-vendor-risk-management-fda-compliance-patient-safety-best-practices)
- Follow a Structured Software Development Lifecycle
- Medical Device Software Development: FDA, SDLC & Cost (2026) – Tech Exactly (https://techexactly.com/blogs/medical-device-software-development-fda-cost)
- Software Development Lifecycle Regulatory Compliance for Medical Devices (https://emergobyul.com/services/software-development-lifecycle-regulatory-compliance-medical-devices)
- How is IEC 62304 transforming medical device software development? – Diffblue (https://diffblue.com/how-is-iec-62304-transforming-medical-device-software-development)
- What You need to know about IEC 62304: Medical Software Lifecycle (https://securitycompass.com/blog/iec-62304-medical-software-lifecycle)
- 5 phases of Medical Device Software Development (https://htdhealth.com/insights/medical-device-software-development-phases)
- Prioritize Cybersecurity and Data Protection
- FDA Tightens Its Medical Device Cybersecurity Guidance (https://health-isac.org/fda-tightens-its-medical-device-cybersecurity-guidance)
- The Life-or-Death Stakes of Medical Device Cybersecurity (https://latimes.com/doctors-scientists/innovations/technology/story/medical-device-cybersecurity-risks-reporting-accountability)
- MedTech and Medical Device Cybersecurity News | MedTech Dive (https://medtechdive.com/topic/cybersecurity)
- Customer Updates: Stryker Network Disruption (https://stryker.com/us/en/about/news/2026/a-message-to-our-customers-03-2026.html)
- FDA Tightens Its Medical Device Cybersecurity Guidance (https://fedtechmagazine.com/article/2026/03/fda-tightens-its-medical-device-cybersecurity-guidance-perfcon)
