Introduction
Navigating the complex landscape of medical device software design compliance presents significant challenges for developers and organizations. Understanding regulatory frameworks such as IEC 62304 and ISO 13485 is crucial, not only for legal compliance but also for enhancing patient safety and product efficacy. As the industry evolves and regulations change, it becomes imperative for developers to ensure they are not merely meeting standards but also fostering innovation.
This article explores four best practices that can guide medical device software teams through the intricacies of compliance, quality assurance, and user-centric design, ultimately leading to improved patient outcomes and successful product launches.
Understand Regulatory Requirements for Medical Device Software
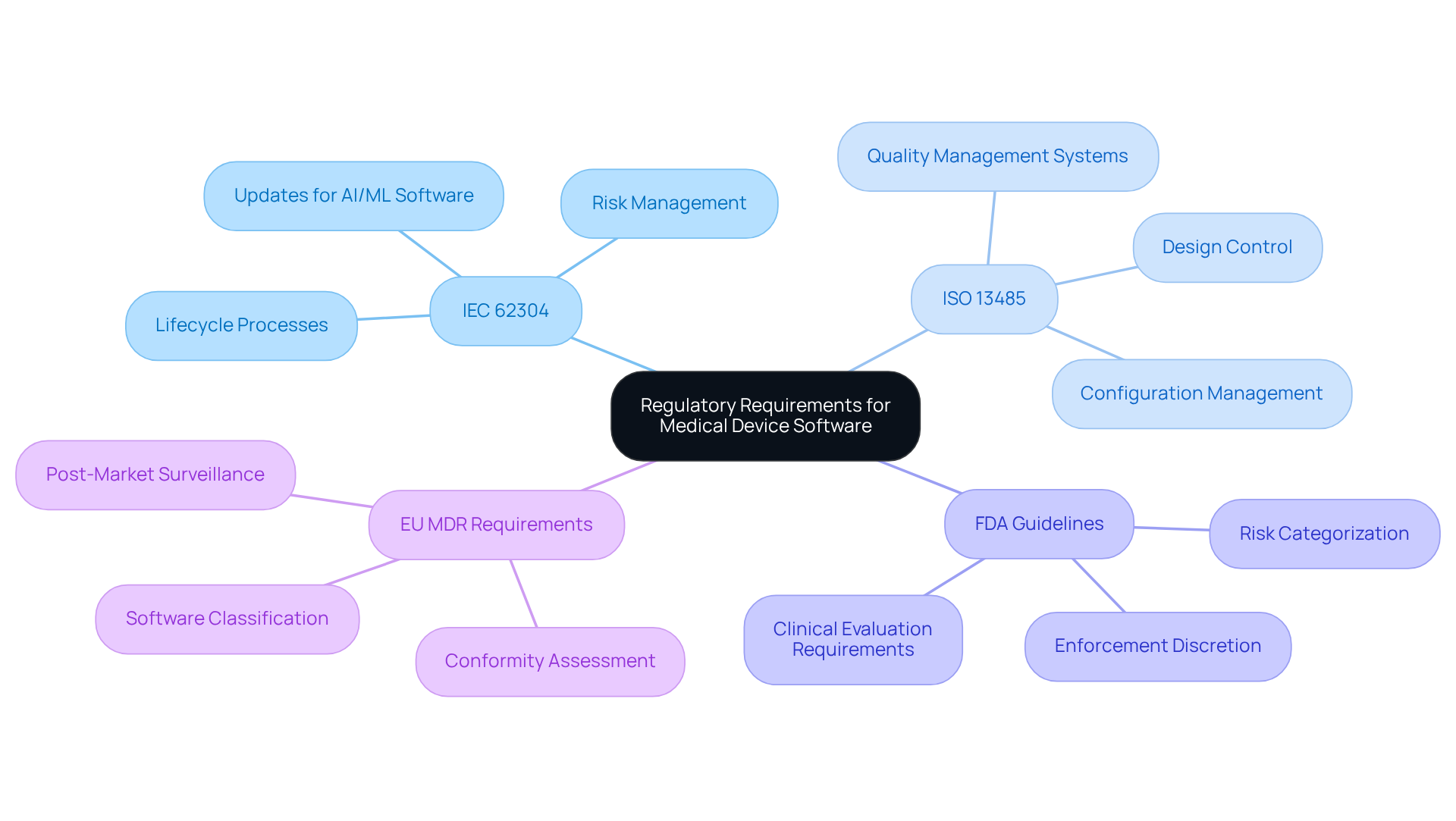
Achieving compliance in medical device software design necessitates a thorough understanding of key regulatory frameworks, particularly IEC 62304 and ISO 13485. IEC 62304 delineates the lifecycle processes for application development, while ISO 13485 emphasizes the establishment of effective quality management systems. Familiarity with these standards is instrumental in defining the project’s scope, identifying requisite documentation, and implementing robust risk management protocols related to medical device software design.
Moreover, staying informed about the latest FDA guidelines and EU MDR requirements is vital, as these regulations are continually evolving to accommodate new technologies and market demands. Engaging with regulatory consultants or participating in industry workshops can provide invaluable insights into these requirements, ensuring that your application not only complies with regulations but also enhances patient safety and efficacy.
Recent case studies illustrate the successful application of ISO 13485 within medical equipment companies, highlighting its role in optimizing compliance procedures and improving product quality. Looking ahead to 2026, adoption rates for ISO 13485 among medical product firms are projected to rise, reflecting a growing recognition of its significance in program development.
Implement Quality Assurance Throughout the Development Lifecycle
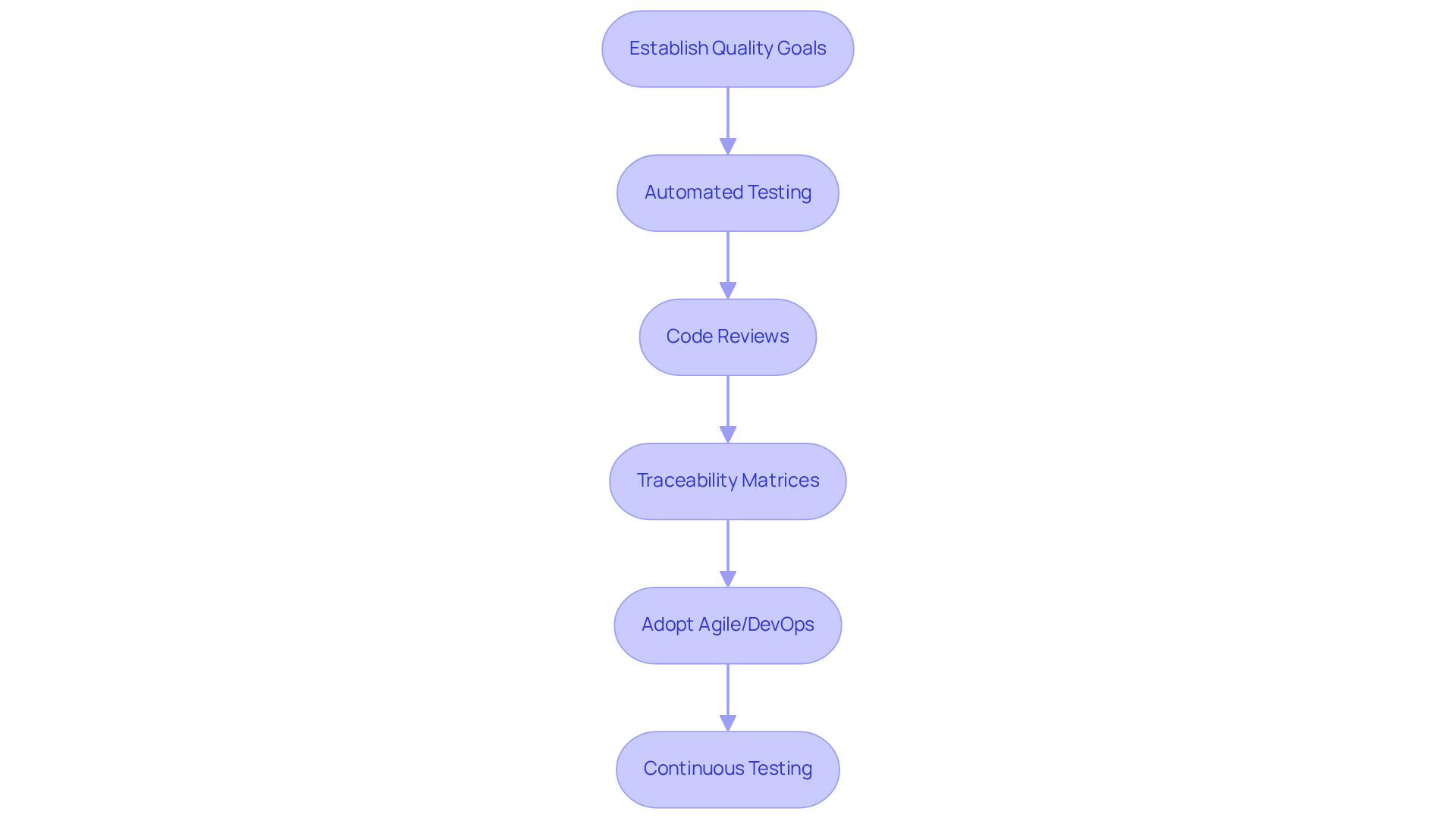
Incorporating quality assurance (QA) into every stage of the medical device software design lifecycle is essential for ensuring compliance and achieving high-quality results. It begins with establishing clear quality goals and metrics during the planning phase, which directs the creation process effectively.
- Automated testing tools are pivotal in streamlining testing, offering comprehensive coverage of functionalities while minimizing manual effort.
- Regular code reviews and peer assessments are vital for the early detection of issues, promoting a proactive approach to quality assurance.
- Implementing traceability matrices connects requirements directly to test cases, ensuring that all specifications are validated thoroughly.
Adopting methodologies such as Agile or DevOps enhances the iterative nature of testing, facilitating continuous feedback and the rapid identification of defects. This proactive strategy not only improves quality but also cultivates a culture of accountability and excellence within the development team.
As the landscape of medical device software design evolves, integrating QA throughout the SDLC will be crucial for meeting regulatory standards, particularly with the FDA’s updated Quality Management System Regulation (QMSR) set to take effect in February 2026. Continuous testing, now integrated into CI/CD pipelines, is becoming standard practice, driven by the necessity for rapid feedback and compliance. By addressing potential pitfalls in QA implementation, organizations can avoid common missteps and ensure successful product outcomes.
Establish Post-Market Monitoring and Continuous Improvement
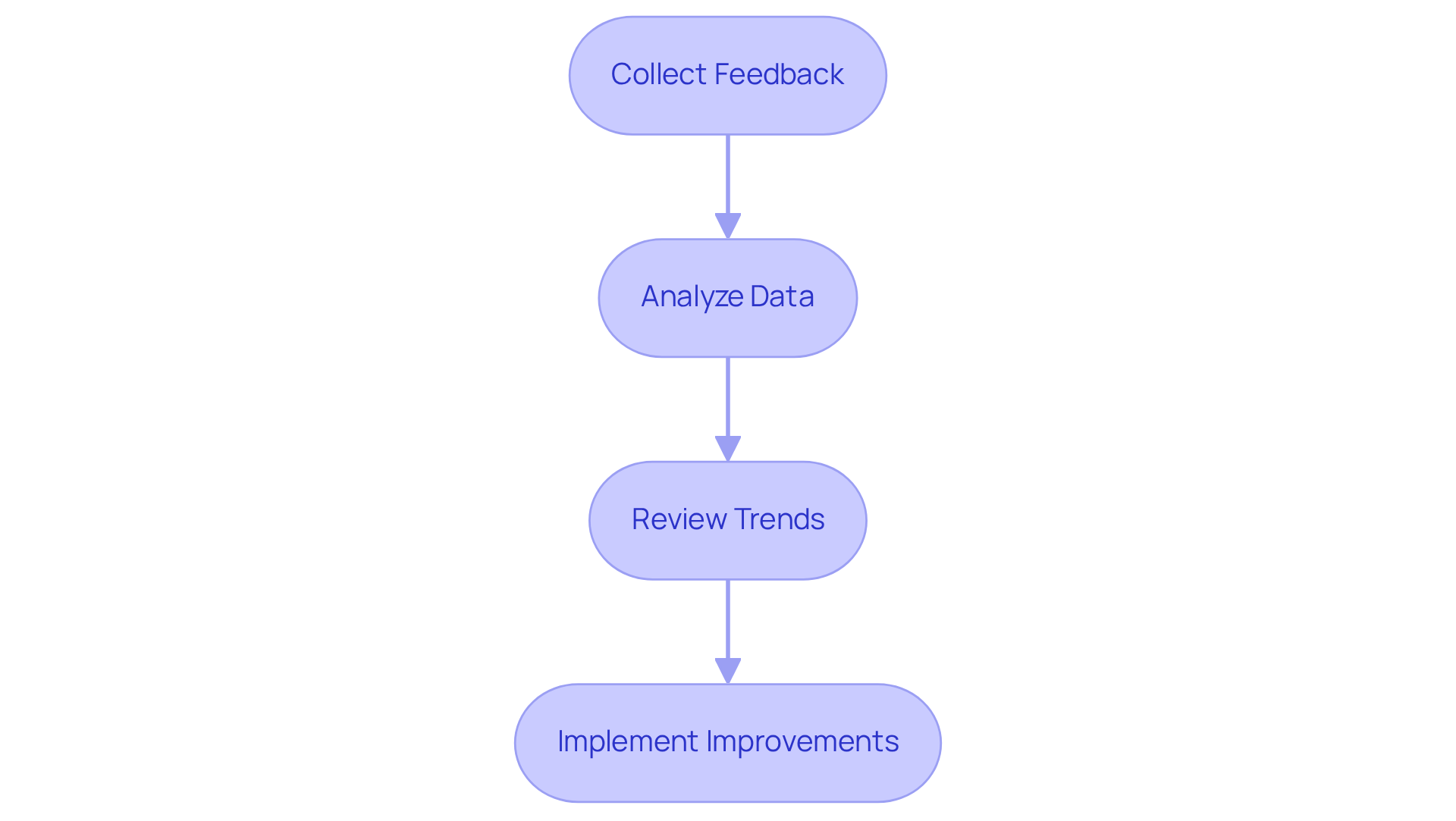
Post-market monitoring serves as a critical component in the lifecycle management of medical device software design. It is essential to implement a robust system for collecting and analyzing feedback, adverse event reports, and performance data. Tools such as participant surveys, product registries, and clinical follow-up studies should be utilized to gather comprehensive insights.
A dedicated team must be established to review this data regularly, identifying trends or issues that may require intervention. Additionally, a feedback mechanism should be put in place to inform future application updates and improvements. This ensures that the product evolves in response to audience needs and regulatory changes.
By prioritizing continuous improvement, organizations can enhance product safety, efficacy, and customer satisfaction. Ultimately, this commitment leads to better patient outcomes.
Incorporate Key Design Considerations for User-Centric Software
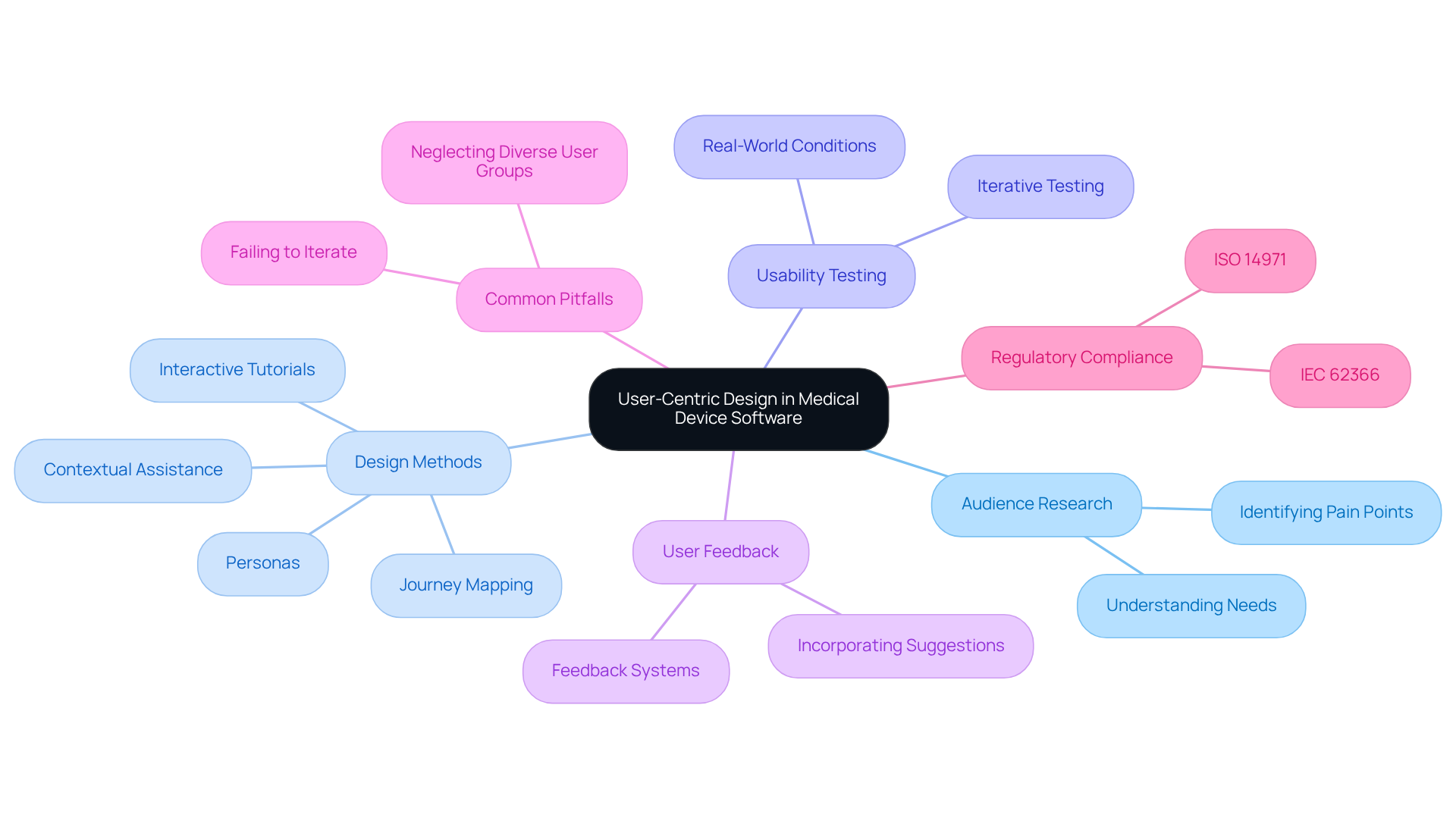
User-centric design is essential in the medical device software design process. It begins with comprehensive audience research to understand the needs, preferences, and pain points of the target group. Employing methods such as personas, journey mapping, and usability testing can effectively guide design choices. The interface must be intuitive and accessible, catering to individuals with varying levels of technical proficiency. As Sophia Andrus notes, “When interfaces are clear, consistent, and tested in real-world conditions, clinicians make fewer errors, patients feel more confident, and teams avoid costly setbacks.”
To enhance engagement, it is crucial to implement features like interactive tutorials and contextual assistance. Additionally, prioritizing feedback systems allows users to report issues or suggest enhancements effortlessly. By incorporating user feedback into the design process, software can not only comply with regulatory standards such as IEC 62366 and ISO 14971 but also provide an improved experience for users, ultimately leading to better health outcomes.
Statistics indicate that effective usability testing is a vital component of medical device software design, as it can significantly reduce errors and enhance confidence. However, it is important to be aware of common pitfalls in user-centric design, such as neglecting to test with diverse user groups or failing to iterate based on feedback. By implementing these user-centric design practices, improved adoption rates and enhanced patient outcomes can be expected.
Conclusion
Achieving compliance in medical device software design transcends mere regulatory obligation; it is essential for ensuring patient safety and product efficacy. By comprehensively understanding and implementing the necessary regulatory frameworks, organizations can adeptly navigate the complexities of software design while adhering to standards that safeguard both users and stakeholders.
This article underscores key practices, including:
- A thorough understanding of regulatory requirements such as IEC 62304 and ISO 13485
- The integration of quality assurance throughout the development lifecycle
- The significance of post-market monitoring
Each of these components is vital in fostering an environment of continuous improvement and user-centric design, ultimately leading to enhanced product quality and user satisfaction.
As the landscape of medical device software evolves, it is imperative for organizations to adopt proactive compliance strategies. Embracing these best practices not only ensures adherence to current regulations but also positions companies to tackle future challenges and seize opportunities within the industry. By prioritizing quality, user experience, and ongoing feedback, the medical device sector can drive innovation while safeguarding the health and well-being of patients globally.
Frequently Asked Questions
What are the key regulatory frameworks for medical device software design?
The key regulatory frameworks for medical device software design are IEC 62304, which outlines the lifecycle processes for application development, and ISO 13485, which focuses on establishing effective quality management systems.
Why is familiarity with IEC 62304 and ISO 13485 important?
Familiarity with these standards is crucial for defining the project’s scope, identifying necessary documentation, and implementing robust risk management protocols related to medical device software design.
What additional guidelines should be considered in medical device software compliance?
It is important to stay informed about the latest FDA guidelines and EU MDR requirements, as these regulations are constantly evolving to address new technologies and market demands.
How can one gain insights into regulatory requirements for medical device software?
Engaging with regulatory consultants or participating in industry workshops can provide valuable insights into regulatory requirements, helping ensure compliance and enhancing patient safety and efficacy.
What do recent case studies reveal about the application of ISO 13485?
Recent case studies demonstrate the successful application of ISO 13485 within medical equipment companies, showcasing its role in optimizing compliance procedures and improving product quality.
What is the projected trend for ISO 13485 adoption among medical product firms by 2026?
Adoption rates for ISO 13485 among medical product firms are projected to rise by 2026, indicating a growing recognition of its importance in program development.
List of Sources
- Understand Regulatory Requirements for Medical Device Software
- IEC 62304 Update 2026: Key Changes & Compliance Tips (https://lfhregulatory.co.uk/iec-62304-update-2026)
- FDA’s 2026 Guidance Expands Pathway for Low-Risk Digital Health Products—But Caution Remains Essential | Berkley Lifesciences (https://berkleyls.com/blog/fdas-2026-guidance-expands-pathway-low-risk-digital-health-products-caution-remains-essential)
- SaMD EU MDR Compliance: Classification, IEC 62304 & CE Marking Guide (2026) (https://mdxcro.com/samd-compliance-guide-mdr-ai-act)
- IEC 62304 Edition 2: 2026 Medical Device Software Changes | IntuitionLabs (https://intuitionlabs.ai/articles/iec-62304-edition-2-medical-software-changes)
- FDA Digital Health Guidance: 2026 Requirements Overview | IntuitionLabs (https://intuitionlabs.ai/articles/fda-digital-health-technology-guidance-requirements)
- Implement Quality Assurance Throughout the Development Lifecycle
- A QMSR State of Mind: FDA Adopts New Inspection Approach for Medical Devices as Quality Management System Regulation Takes Effect | Insights | Ropes & Gray LLP (https://ropesgray.com/en/insights/alerts/2026/02/a-qmsr-state-of-mind-fda-adopts-new-inspection-approach-for-medical-devices)
- Healthcare Application Testing: All You Need to Know in 2026 (https://kms-technology.com/blog/healthcare-application-testing-all-you-need-to-know-in-2026)
- 2026 Medical Device Quality Trends (https://mastercontrol.com/gxp-lifeline/2026-medical-device-trends-quality-management)
- Top Software Testing Trends in 2026: The Future of Software – testomat.io (https://testomat.io/blog/software-testing-trends)
- Best Practices for Developing Medical Devices: Overcoming Challenges (https://volersystems.com/blog/best-practices-for-developing-medical-devices-overcoming-challenges)
- Establish Post-Market Monitoring and Continuous Improvement
- What Are The Emerging Global Medical Device Regulatory Trends For 2026? • Starodub (https://starodub.nl/blog/what-are-the-emerging-global-medical-device-regulatory-trends-for-2026)
- FDA’s 2026 Guidance Expands Pathway for Low-Risk Digital Health Products—But Caution Remains Essential | Berkley Lifesciences (https://berkleyls.com/blog/fdas-2026-guidance-expands-pathway-low-risk-digital-health-products-caution-remains-essential)
- FDA Digital Health Guidance: 2026 Requirements Overview | IntuitionLabs (https://intuitionlabs.ai/articles/fda-digital-health-technology-guidance-requirements)
- Post Market Surveys: A Cost-Effective Path to Real-World Evidence (https://mddionline.com/medical-device-regulations/capturing-real-world-data-why-post-market-surveys-for-medical-devices-are-gaining-ground)
- FDA 2026 Device Guidance Agenda: Key Takeaways | Innovenn posted on the topic | LinkedIn (https://linkedin.com/posts/innovenn-inc-_fda-device-guidance-agenda-what-to-watch-activity-7443007570618892288-kW8m)
- Incorporate Key Design Considerations for User-Centric Software
- Trends Shaping Patient-Centric Design in RPM Platforms (https://healtharc.io/blogs/trends-shaping-patient-centric-design-in-rpm-platforms)
- User-Centered Design in HealthTech in 2026: How UX Drives Safety, Compliance, and Adoption (https://linkedin.com/pulse/user-centered-design-healthtech-2026-how-ux-drives-safety-compliance-aj4pf)
- UX Design in Healthcare: Challenges and Trends Shaping 2026 | Orangesoft (https://orangesoft.co/blog/healthcare-ux-design)
- Medical Device UX Design: Complete Guide for 2026 [With Cases] (https://eleken.co/blog-posts/medical-device-ux-design)
- The Importance of User-Centered Design in Developing Medical Software (https://kandasoft.com/blog/user-centered-design-in-developing-medical-software)
