Introduction
Navigating the intricate landscape of medical device software compliance necessitates a thorough understanding of regulatory frameworks and established practices. As the industry continues to evolve, developers must place a premium on usability and rigorous testing. This ensures that their products not only adhere to compliance standards but also provide exceptional user experiences.
However, with the swift pace of technological advancements and regulatory updates, how can developers effectively balance compliance with innovation while minimizing risks?
This article explores essential strategies for achieving compliance and enhancing usability in medical device software, offering insights that are crucial for success in this vital field.
Understand Regulatory Frameworks for Medical Device Software
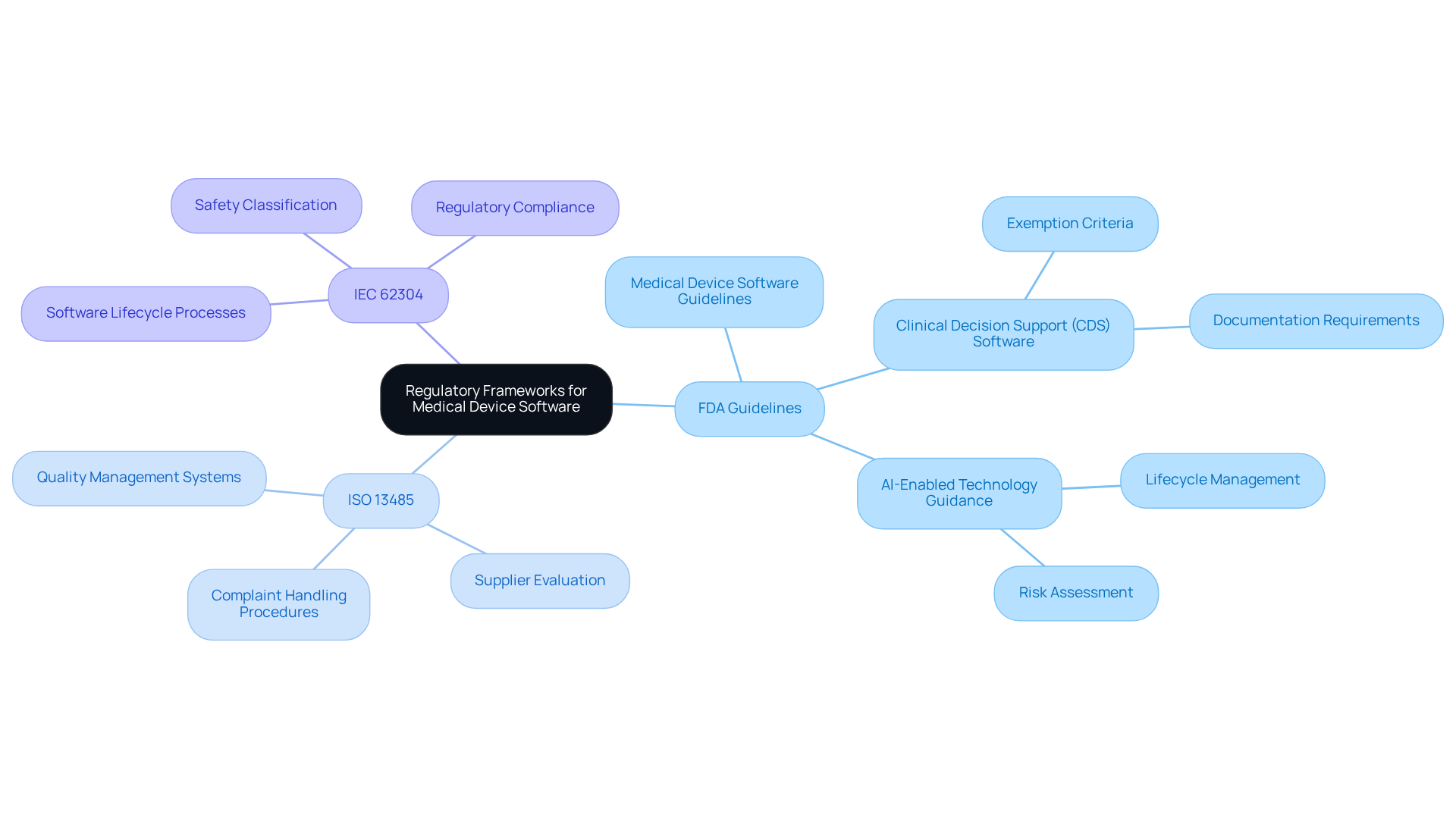
Understanding the governing regulations is crucial for ensuring adherence to compliance standards. Key regulations include:
- The FDA
- IEC 62304 for software development
Familiarity with these regulations is essential for identifying the necessary documentation and testing requirements.
For instance, the FDA’s recent draft guidance on medical device software highlights the significance of compliance. By staying informed about these regulations, developers can align their solutions with industry standards more effectively. This proactive approach not only mitigates the risk of non-compliance but also helps avoid potential legal issues.
Prioritize Usability and User Experience in Design
Usability must be a priority in the design of applications. By adopting best practices, developers can create software that is intuitive, significantly reducing the likelihood of user error. Early user tests are essential; they help identify potential issues before they escalate into costly problems. The FDA mandates guidelines for compliance to ensure that instruments can be utilized safely and effectively by their intended users. As highlighted in the FDA’s guidance, “Companies must demonstrate via quality systems and submission documentation that their device can withstand reasonable cyber attacks,” underscoring the importance of security.
Engaging actual users during the design phase can lead to substantial improvements in usability and adherence to regulatory standards. The 2026 FDA updates emphasize the necessity for user-centered design. Effective evaluations can be conducted using tools such as heuristic evaluations and cognitive walkthroughs, which provide valuable insights into user interactions and help refine the design for optimal performance.
Common pitfalls in design include neglecting to involve users and failing to iterate based on feedback, both of which can hinder the effectiveness of the design.
Implement Comprehensive Testing and Validation Protocols
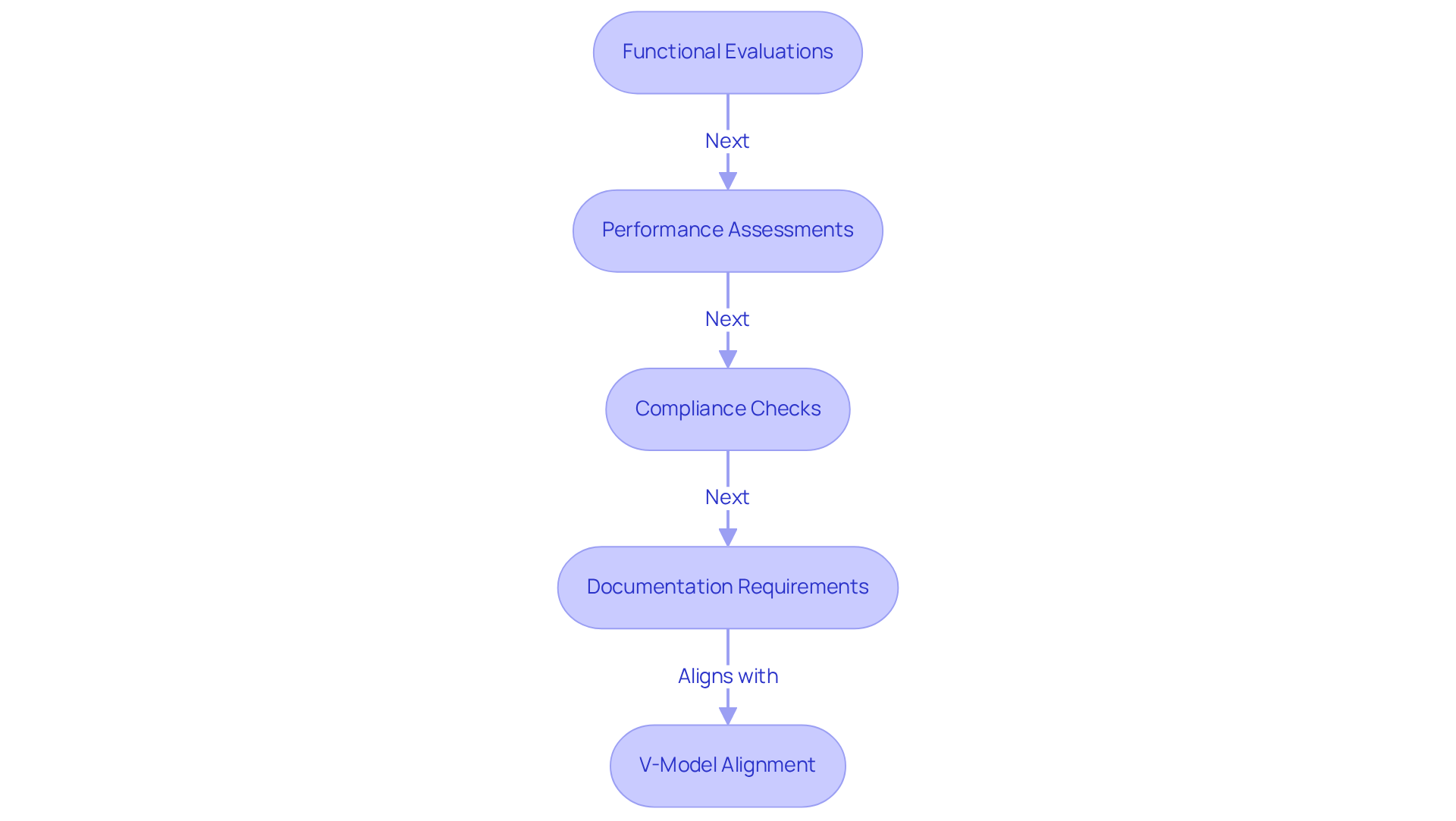
Strong testing protocols are essential for ensuring compliance with regulations and optimal performance of medical device software. These procedures encompass functional evaluations, performance assessments, and compliance checks that align with industry standards.
A well-organized testing plan includes success criteria and documentation requirements. For example, employing the V-model of application development facilitates the alignment of evaluation activities with development stages, ensuring that each component is validated before progressing to the next phase.
Implementation of testing procedures is critical for maintaining compliance and enhancing program quality over time. Notably, comprehensive testing has been shown to significantly improve compliance rates in the development of medical device software, underscoring the necessity of integrating these practices into the development lifecycle.
Furthermore, it is imperative to conduct thorough evaluations to meet regulatory requirements and ensure transparency. Ongoing revisions to the testing process, informed by production and post-production feedback, are vital for maintaining compliance and enhancing program quality.
Industry leaders emphasize that effective validation is not merely a regulatory checkbox; it is a fundamental aspect of ensuring safety and efficacy.
Ensure Continuous Compliance Through Maintenance and Updates
Ongoing adherence is essential for medical device software, particularly as technology and regulations evolve. It is imperative to plan and execute regular updates in accordance with industry standards to address compliance and usability. A robust maintenance plan is crucial, ensuring that all updates are documented, tested, and validated prior to deployment.
For example, the FDA recommends that manufacturers maintain a strategy that includes regular audits to ensure alignment with relevant regulations. By prioritizing maintenance and updates, developers can effectively mitigate risks, thereby enhancing the overall safety and effectiveness of medical devices.
Conclusion
Understanding and implementing best practices for medical device software compliance and usability is crucial for developers aiming to create safe and effective products. Adhering to regulatory frameworks, prioritizing user experience, establishing rigorous testing protocols, and ensuring continuous compliance are fundamental components that contribute to the overall success of medical device software.
This article outlines critical strategies, including:
- The necessity of understanding regulatory guidelines such as those from the FDA and ISO standards.
- The importance of user-centered design to enhance usability.
- The implementation of comprehensive testing and validation protocols.
By focusing on these areas, developers can mitigate risks, improve user satisfaction, and ensure that their products meet the required safety and efficacy standards.
Ultimately, the commitment to maintaining compliance through regular updates and evaluations is vital in an ever-evolving technological landscape. By adopting these best practices, developers not only fulfill regulatory obligations but also enhance the quality and reliability of medical device software. This paves the way for innovations that prioritize patient safety and user experience. Taking proactive steps today can lead to a more secure and efficient future in medical device development.
Frequently Asked Questions
What are the key regulations for medical device software development?
The key regulations include the FDA’s guidelines for medical device software, ISO 13485 for quality management systems, and IEC 62304 for software lifecycle processes.
Why is it important to understand these regulatory frameworks?
Understanding these frameworks is crucial for ensuring adherence to industry standards, identifying necessary documentation, and meeting testing requirements.
What does the FDA’s recent draft guidance on AI-enabled technology emphasize?
It emphasizes the significance of lifecycle management and risk assessment in the development of AI-enabled medical device software.
How can staying informed about regulations benefit developers?
Staying informed allows developers to align their solutions with compliance standards, mitigating the risk of costly recalls and avoiding potential legal issues.
List of Sources
- Understand Regulatory Frameworks for Medical Device Software
- Key Updates in FDA’s 2026 General Wellness and Clinical Decision Support Software Guidance | Faegre Drinker Biddle & Reath LLP (https://faegredrinker.com/en/insights/publications/2026/1/key-updates-in-fdas-2026-general-wellness-and-clinical-decision-support-software-guidance)
- FDA Digital Health Guidance: 2026 Requirements Overview | IntuitionLabs (https://intuitionlabs.ai/articles/fda-digital-health-technology-guidance-requirements)
- greenlight.guru (https://greenlight.guru/blog/iso-13485-qms-medical-device)
- #iso13485 #medicaldevices #qualitymanagement #qms #regulatoryaffairs #medtech #qualityassurance | Uma Gaur, PhD, RAC (US, Europe, Canada) | 14 comments (https://linkedin.com/posts/uma-gaur-phd-rac-us-europe-canada-03334629_iso13485-medicaldevices-qualitymanagement-activity-7389968813863231488-e4Mj)
- Prioritize Usability and User Experience in Design
- FDA Digital Health Guidance: 2026 Requirements Overview | IntuitionLabs (https://intuitionlabs.ai/articles/fda-digital-health-technology-guidance-requirements)
- How Will FDA’s New Rules Impact Your Health Gadgets? (https://spectrum.ieee.org/fda-medical-device-rules)
- 30 Quotes on User-Centered Interaction Design (https://design4users.com/30-quotes-on-user-centered-interaction-design)
- Key Benefits of Usability Testing in Medical Devices (https://emergobyul.com/resources/importance-usability-testing-medical-devices)
- Implement Comprehensive Testing and Validation Protocols
- medicaldevicehq.com (https://medicaldevicehq.com/articles/the-illustrated-guide-to-risk-management-for-medical-devices-and-iso-14971)
- Utilizing statistical methodologies for efficient verification and validation of medical devices industry designs. (https://linkedin.com/pulse/utilizing-statistical-methodologies-rz8cf)
- lumafield.com (https://lumafield.com/case-studies/case-study-maintaining-fda-compliance-with-validated-ct-technology)
- greenlight.guru (https://greenlight.guru/blog/iso-14971-risk-management)
- FDA Digital Health Guidance: 2026 Requirements Overview | IntuitionLabs (https://intuitionlabs.ai/articles/fda-digital-health-technology-guidance-requirements)
- Ensure Continuous Compliance Through Maintenance and Updates
- Key Updates in FDA’s 2026 General Wellness and Clinical Decision Support Software Guidance | Faegre Drinker Biddle & Reath LLP (https://faegredrinker.com/en/insights/publications/2026/1/key-updates-in-fdas-2026-general-wellness-and-clinical-decision-support-software-guidance)
- Software as a Medical Device Market Size & Trends Report (https://rootsanalysis.com/reports/software-as-a-medical-device-market.html)
- FDA Updates Broaden Wellness and Clinical Decision Support Software Devices Under Enforcement (https://emergobyul.com/news/fda-updates-broaden-wellness-and-clinical-decision-support-software-devices-under-enforcement)
- ventionteams.com (https://ventionteams.com/blog/medical-device-software-statistics)
- Case Studies | Regulatory Compliance for Life Sciences | Arbour Group (https://arbourgroup.com/case-studies)
