Introduction
Regulatory compliance in medical software development serves as a fundamental pillar that ensures the integrity and safety of healthcare solutions. As developers navigate the intricate web of regulatory requirements, understanding the implications of regulations like HIPAA and FDA guidelines becomes paramount.
However, navigating the intricate web of regulatory requirements poses significant challenges for developers. Failure to adapt could result in non-compliance and compromised patient safety.
This article explores best practices that address regulatory adherence while leveraging innovative technologies and user-centered design to enhance quality and safety in medical applications.
Understand Regulatory Compliance in Medical Software Development
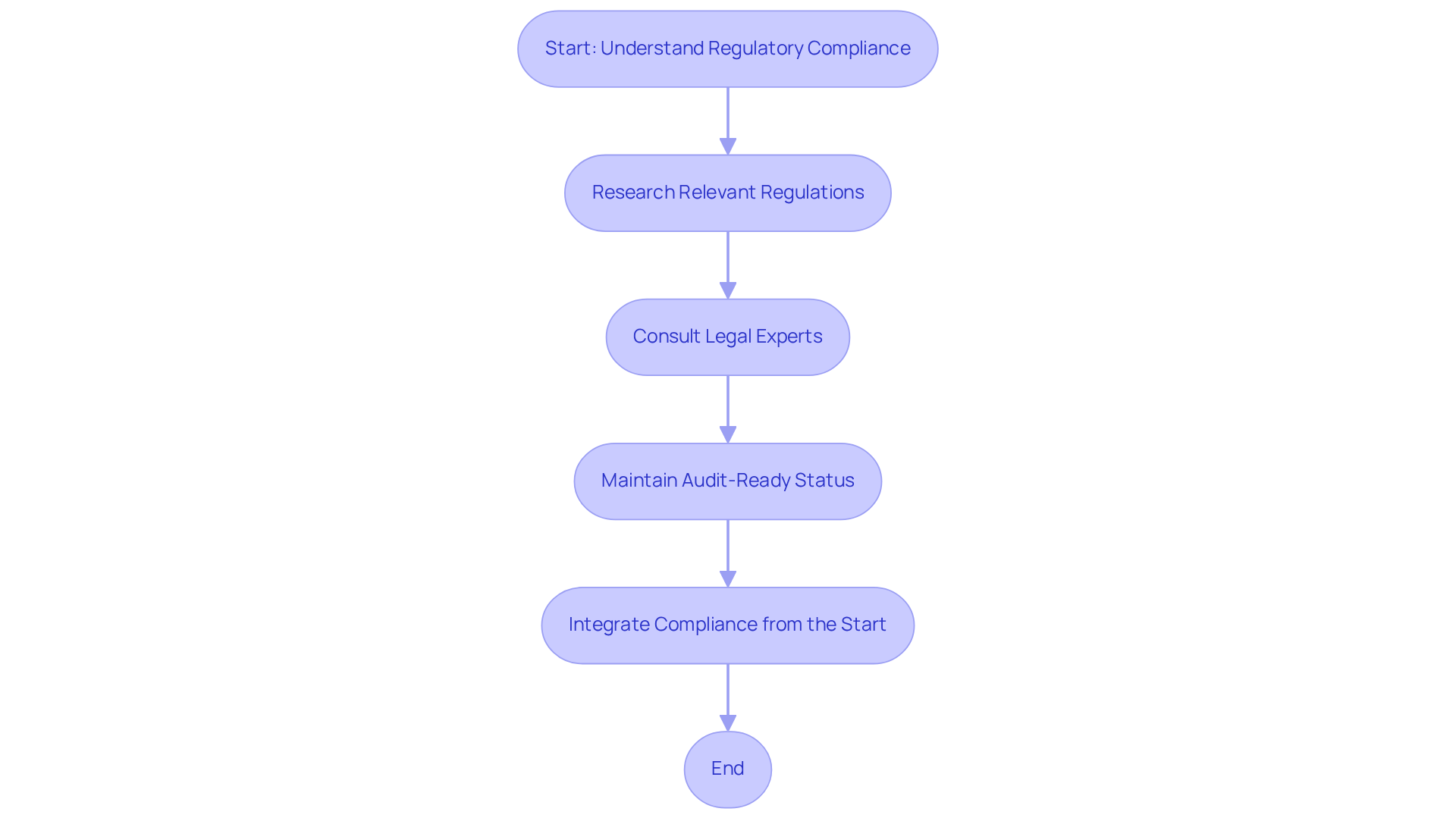
Regulatory compliance in software development for medical applications is not just important; it is a fundamental requirement that shapes the integrity of healthcare solutions. It encompasses adherence to various laws and standards governing the healthcare industry. The Health Insurance Portability and Accountability Act (HIPAA) plays a crucial role in safeguarding patient information, while the FDA provides guidelines for applications classified as a medical device (SaMD). Developers must grasp these regulations to ensure that their software development for medical is both functional and legally compliant.
Developers must start by thoroughly researching relevant regulations and standards to ensure compliance. Consulting legal experts can clarify complex regulatory requirements, ensuring accurate interpretation. Furthermore, maintaining an audit-ready status throughout the software lifecycle is essential. This can be achieved through strong documentation practices and regular adherence checks, which are crucial for proving compliance with regulatory standards.
Case studies, such as the compliance of Electronic Health Record (EHR) systems with HIPAA and Dexcom Glucose Monitors, clearly show why regulatory considerations must be integrated from the very beginning of the software development for medical process. These examples demonstrate how prioritizing regulatory adherence not only reduces risks but also boosts the credibility of technological solutions. As the healthcare environment changes, especially with the expected revisions to HIPAA and FDA regulations in 2026, the cost of non-compliance will only increase, making proactive adherence not just beneficial but essential for success in the healthcare technology landscape. Additionally, the introduction of new Process Rigor Levels, which categorize applications into Class A (Low risk) and Classes B & C (High risk), along with the requirement for multifactor authentication for systems handling PHI, are crucial considerations for developers in this evolving regulatory environment.
Implement User-Centered Design for Enhanced Quality
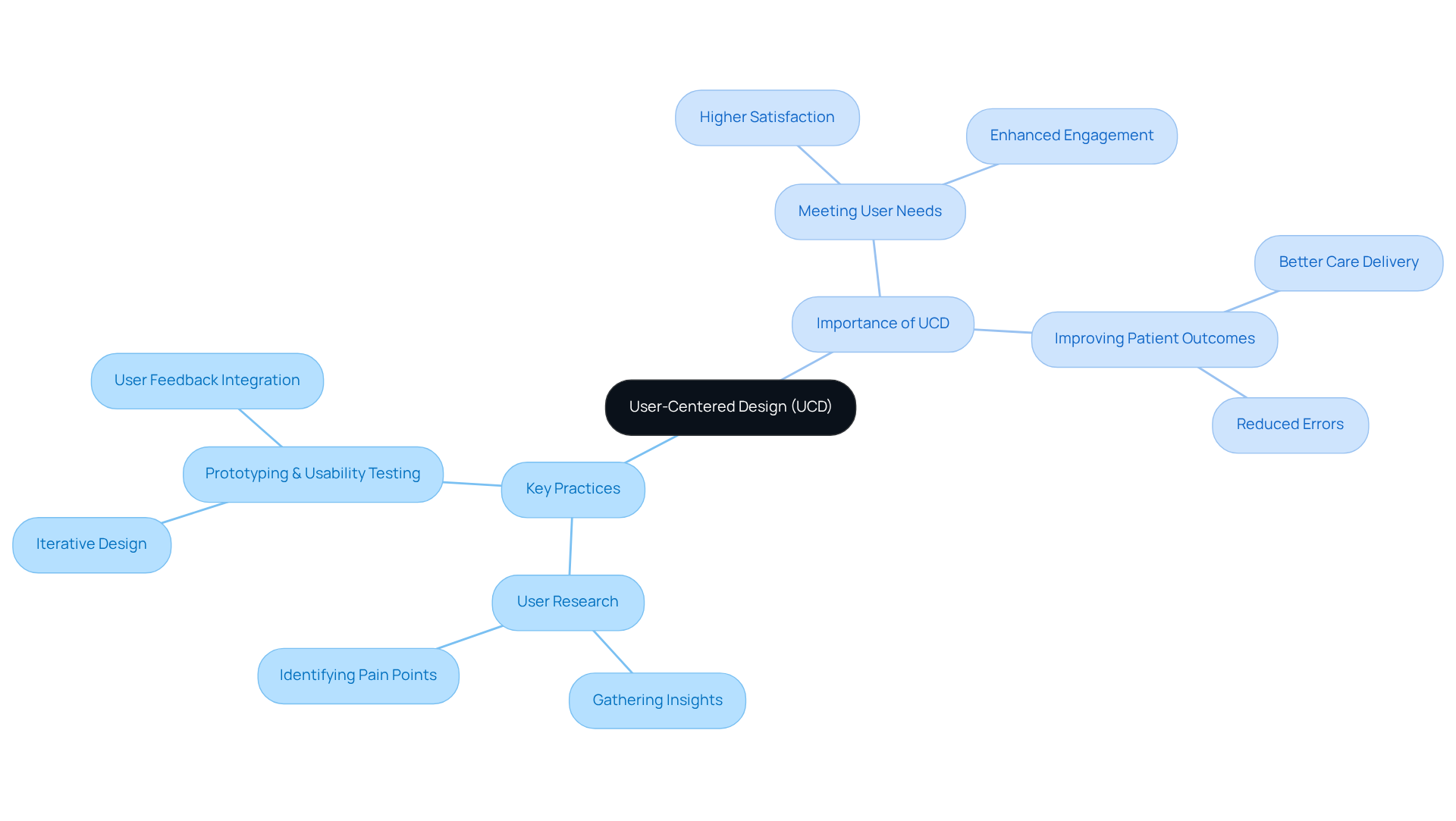
Neglecting user input in medical application development can lead to systems that fail to meet user needs effectively. Implementing user-centered design (UCD) involves actively engaging end-users throughout the design process. This approach ensures the system meets users’ specific needs and preferences, leading to higher satisfaction and better patient outcomes.
Key practices for effective UCD include:
- Conducting user research to gather insights on user needs, preferences, and pain points.
- Prototyping and usability testing, which are essential stages that enable creators to enhance their designs based on actual user feedback.
For instance, a study on telehealth applications revealed that incorporating user feedback significantly improved the usability and adoption rates of the program. Failing to prioritize user-centered design risks creating applications that may hinder patient care and satisfaction.
Conduct Comprehensive Testing and Validation Procedures
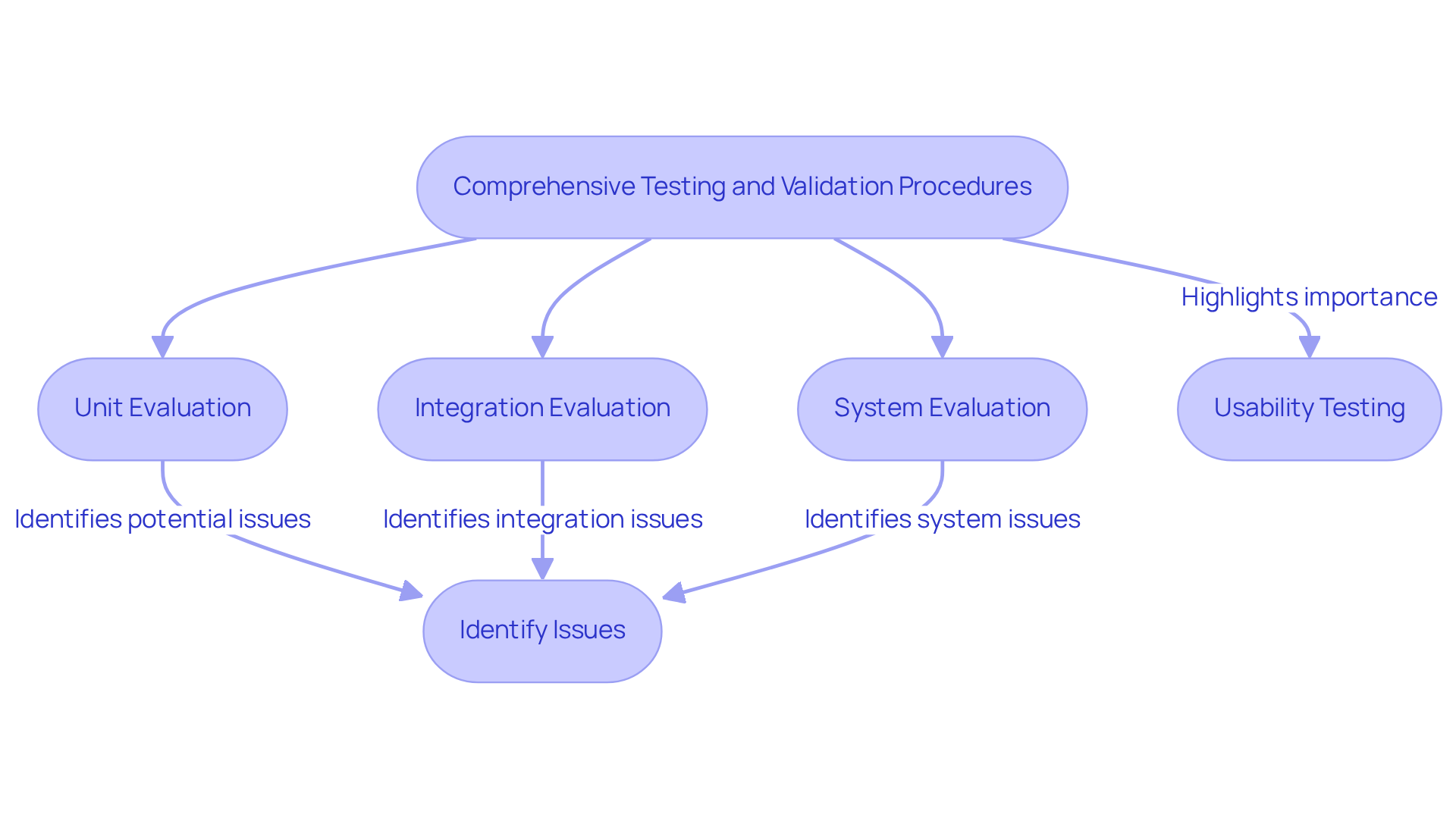
Thorough assessment and validation processes are crucial in the software development for medical applications lifecycle, as they ensure compliance with regulatory standards. This process guarantees that the system meets all functional requirements. Key evaluation methods include:
- Unit evaluation
- Integration evaluation
- System evaluation
Each serving to identify and rectify potential issues before deployment.
Validation should also include usability testing to ensure that the application is user-friendly and meets the needs of healthcare professionals and patients. The FDA highlights the significance of validation in its guidelines for Software as a Medical Device (SaMD), necessitating that applications be tested under conditions that mimic real-world use. Additionally, the FDA’s revised guidance on Computer Programs Assurance for Production and Quality System Applications, effective September 24, 2025, emphasizes the necessity for a modernized approach to validation.
By implementing a structured assessment and validation framework, developers in software development for medical can significantly reduce the risk of software failures, enhance patient safety, and ensure compliance with industry regulations. Furthermore, leveraging vendor documentation effectively minimizes unnecessary evaluations, streamlining the validation process. Research indicates that around 25% of companies investing in test automation experience immediate returns on investment, highlighting the benefits of adopting structured testing frameworks. It is also essential to ensure that products do not replace FDA-authorized devices or encourage specific clinical actions, as these common pitfalls can result in regulatory issues. By proactively addressing these challenges, developers can foster innovation while ensuring patient safety and regulatory compliance.
Leverage Advanced Technologies for Compliance and Quality Improvement
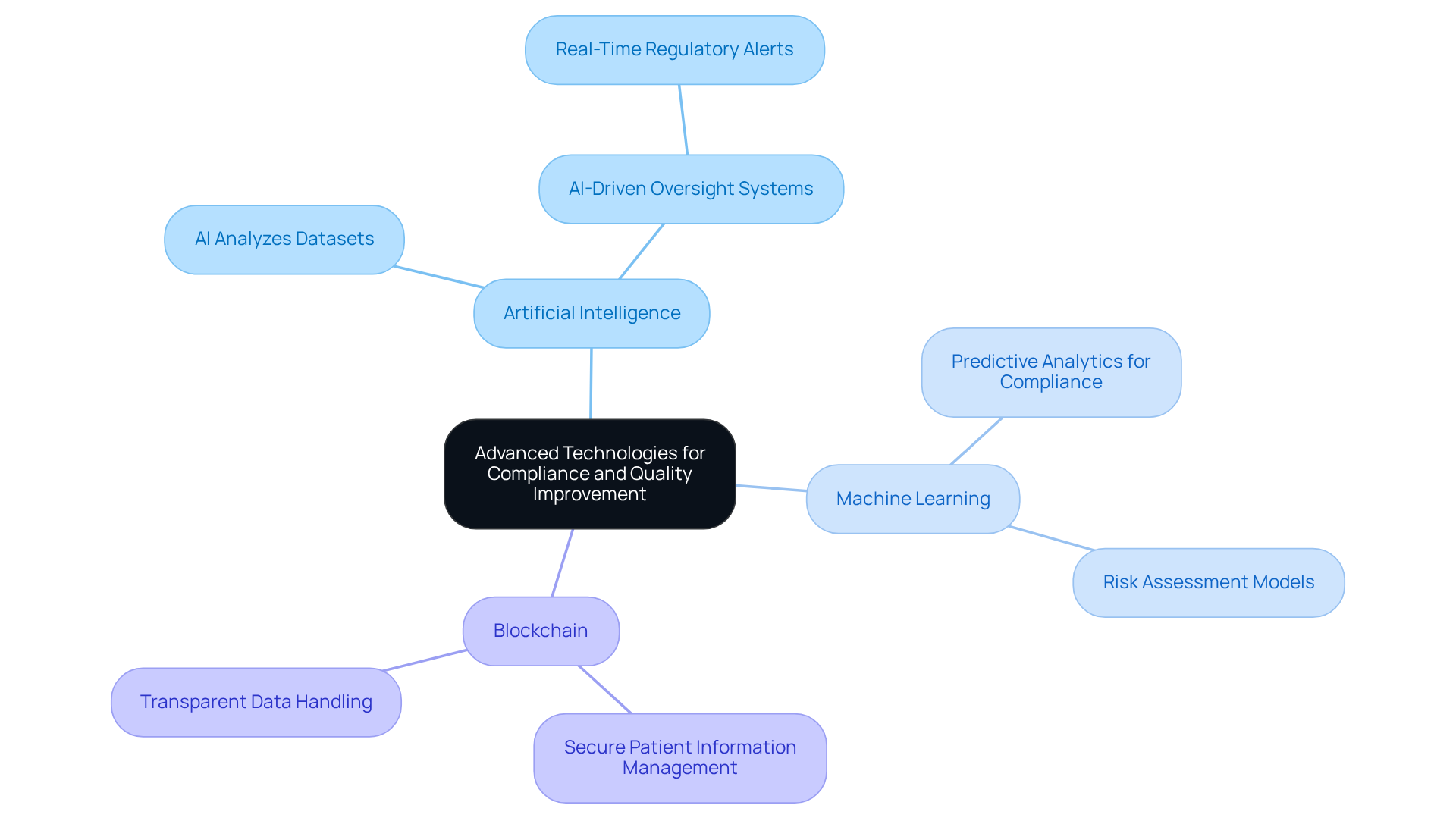
The integration of advanced technologies such as artificial intelligence, machine learning, and blockchain presents a significant opportunity to enhance compliance and quality in medical application development. These technologies automate regulatory checks, streamline documentation, and bolster data security, addressing critical challenges in the field.
For example:
- AI analyzes large datasets to verify compliance with regulatory standards.
- Blockchain offers a secure and transparent method for managing patient information.
Practical applications include:
- AI-driven oversight systems that alert developers to potential regulatory violations in real-time, enhancing compliance efforts.
The strategic adoption of these technologies not only addresses compliance challenges but also sets a new standard for quality in software development for medical purposes.
Conclusion
Regulatory compliance serves as a foundational element in medical software development, ensuring the integrity and effectiveness of healthcare solutions. Developers must understand and adhere to regulations like HIPAA and FDA guidelines to create functional and legally compliant software. By prioritizing compliance from the outset, developers can safeguard patient information and enhance the credibility of their technological solutions.
Key practices discussed in this article include:
- Thorough research into regulations
- The implementation of user-centered design
- The importance of comprehensive testing and validation procedures
Involving end-users in the design process helps ensure that applications address real-world needs, and structured testing frameworks can pinpoint potential issues before deployment. Moreover, leveraging advanced technologies like AI and blockchain can significantly streamline compliance efforts and improve overall software quality.
The journey toward compliance in medical software development is ongoing and requires vigilance and proactive measures. Navigating the complexities of regulatory compliance can be daunting for developers. As regulations evolve and new technologies emerge, developers must remain adaptable and committed to best practices. Failure to comply can lead to significant legal repercussions and undermine patient trust. By embracing these strategies, the industry can enhance patient safety and satisfaction while fostering innovation that meets the ever-changing demands of healthcare.
Frequently Asked Questions
What is the importance of regulatory compliance in medical software development?
Regulatory compliance is fundamental in medical software development as it shapes the integrity of healthcare solutions and ensures adherence to laws and standards governing the healthcare industry.
Which regulations are critical for medical software developers to understand?
Key regulations include the Health Insurance Portability and Accountability Act (HIPAA), which safeguards patient information, and guidelines from the FDA for applications classified as medical devices (SaMD).
How can developers ensure compliance with regulatory standards?
Developers should thoroughly research relevant regulations, consult legal experts for clarification, and maintain an audit-ready status through strong documentation practices and regular adherence checks.
Why is it important to integrate regulatory considerations from the beginning of the software development process?
Integrating regulatory considerations early reduces risks and boosts the credibility of technological solutions, as demonstrated by case studies like the compliance of Electronic Health Record (EHR) systems with HIPAA.
What are the potential consequences of non-compliance in medical software development?
The cost of non-compliance is expected to increase, making proactive adherence essential for success in the healthcare technology landscape.
What are the new Process Rigor Levels introduced in the regulatory environment?
The new Process Rigor Levels categorize applications into Class A (Low risk) and Classes B & C (High risk), which developers must consider.
What security measures are required for systems handling Protected Health Information (PHI)?
Multifactor authentication is required for systems that handle PHI to enhance security and compliance.
List of Sources
- Understand Regulatory Compliance in Medical Software Development
- IEC 62304 Update 2026: Key Changes & Compliance Tips (https://lfhregulatory.co.uk/iec-62304-update-2026)
- HIPAA Security Rule Update Targets May 2026 Final Rule (https://brightdefense.com/news/hipaa-security-rule-update)
- FDA Digital Health Guidance: 2026 Requirements Overview | IntuitionLabs (https://intuitionlabs.ai/articles/fda-digital-health-technology-guidance-requirements)
- Modernizing HIPAA: Are You Ready? (https://bankinfosecurity.com/blogs/modernizing-hipaa-are-you-ready-p-4061)
- Implement User-Centered Design for Enhanced Quality
- User-Centered Design in HealthTech in 2026: How UX Drives Safety, Compliance, and Adoption (https://linkedin.com/pulse/user-centered-design-healthtech-2026-how-ux-drives-safety-compliance-aj4pf)
- Top 5 UX Trends Driving Digital Healthcare in 2026 – Onething Design (https://onething.design/post/top-healthcare-ux-trends)
- The Healthcare Experience Reckoning: What 2026 Demands of Leaders. My thoughts. (https://linkedin.com/pulse/healthcare-experience-reckoning-what-2026-demands-leaders-al-krueger-yg8uc)
- Healthcare UX Design Guide: Best Practices for Clinical Products 2026 (https://fuselabcreative.com/healthcare-ux-design-best-practices-guide)
- Elevating Healthcare Access: The Impact of Intelligent Virtual Solutions (https://healthcaretechoutlook.com/news/elevating-healthcare-access-the-impact-of-intelligent-virtual-solutions-nid-4555.html)
- Conduct Comprehensive Testing and Validation Procedures
- Latest Software Testing Statistics (2026 Edition) (https://testgrid.io/blog/software-testing-statistics)
- FDA Adapts with the Times on Digital Health: Updated Guidances on General Wellness Products and Clinical Decision Support Software | Insights | Ropes & Gray LLP (https://ropesgray.com/en/insights/alerts/2026/01/fda-adapts-with-the-times-on-digital-health-updated-guidances-on-general-wellness-products)
- For 2026, FDA signals shifts in digital health framework | Nixon Peabody LLP (https://nixonpeabody.com/insights/alerts/2026/01/27/for-2026-fda-signals-shifts-in-digital-health-framework)
- FDA releases new guidance on software validation for medical devices | Marcelo Trevino posted on the topic | LinkedIn (https://linkedin.com/posts/marcelo-trevino-medicaldevices_computer-software-assurance-for-production-activity-7376400250091143168-acFI)
- FDA Digital Health Guidance: 2026 Requirements Overview | IntuitionLabs (https://intuitionlabs.ai/articles/fda-digital-health-technology-guidance-requirements)
- Leverage Advanced Technologies for Compliance and Quality Improvement
- FDA Digital Health Guidance: 2026 Requirements Overview | IntuitionLabs (https://intuitionlabs.ai/articles/fda-digital-health-technology-guidance-requirements)
- Healthcare’s Regulatory Reckoning: Navigating AI and Data Privacy Overhauls in 2026 (https://linkedin.com/pulse/healthcares-regulatory-reckoning-navigating-ai-data-privacy-conaway-avl9e)
- Pharma’s AI Adoption: Learning From Other Technology Adoptions (https://clinicalresearchnewsonline.com/news/2026/02/13/pharma-s-ai-adoption–learning-from-other-technology-adoptions)
- IEC 62304 Update 2026: Key Changes & Compliance Tips (https://lfhregulatory.co.uk/iec-62304-update-2026)
- For 2026, FDA signals shifts in digital health framework | Nixon Peabody LLP (https://nixonpeabody.com/insights/alerts/2026/01/27/for-2026-fda-signals-shifts-in-digital-health-framework)
