Introduction
In the rapidly evolving landscape of medical technology, the development of software solutions stands as a pivotal factor in enhancing patient care and healthcare delivery. As the MedTech industry continues to innovate, it becomes essential for stakeholders to understand best practices in software development to create effective and compliant applications. Given the myriad of regulatory standards and user expectations, developers face the challenge of navigating this complex terrain. They must ensure that their solutions not only meet industry requirements but also deliver exceptional user experiences.
Identify Key Types of MedTech Software
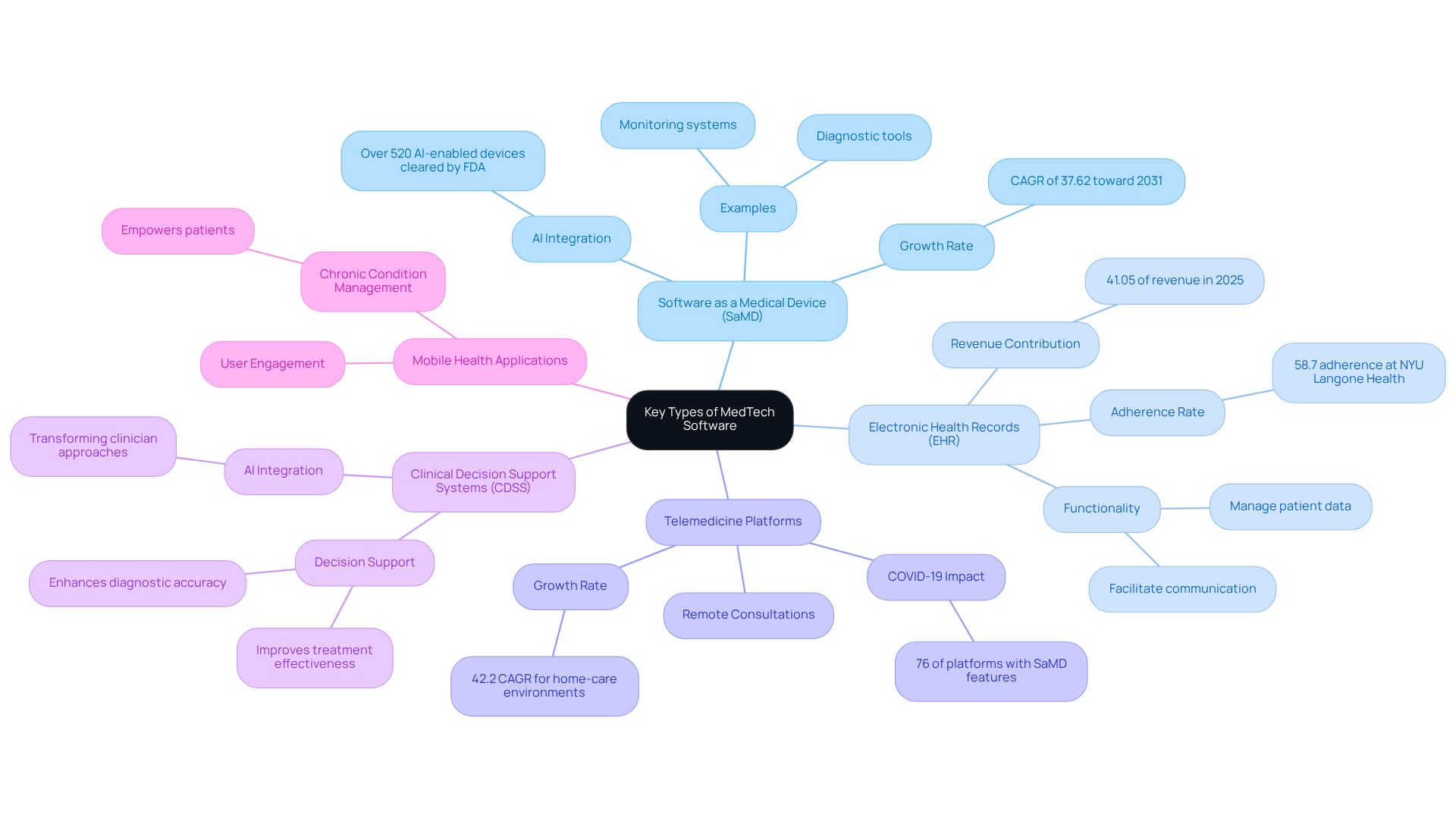
In the MedTech sector, software can be categorized into several key types, each serving distinct purposes:
-
Software as a Medical Device (SaMD): This category includes applications that autonomously perform medical functions, such as diagnostic tools and monitoring systems for individuals. The SaMD market is projected to experience significant growth, with a forecasted CAGR of 37.62% toward 2031, driven by advancements in AI and machine learning. Notably, the FDA has cleared over 520 AI-enabled medical devices to date, underscoring the critical role of AI in this sector.
-
Electronic Health Records (EHR): EHR systems manage patient data and facilitate communication among healthcare providers, playing a crucial role in enhancing care coordination and outcomes. Successful implementations have demonstrated improved data submission adherence, as evidenced by programs like NYU Langone Health’s, which achieved a 58.7% adherence rate. Hospitals and clinics contributed 41.05% of revenue in 2025, highlighting the significance of EHR systems in the MedTech landscape.
-
Telemedicine Platforms: These software solutions enable remote consultations and monitoring, thereby expanding access to healthcare services. The COVID-19 pandemic accelerated their adoption, with 76% of telehealth platforms incorporating at least one SaMD feature, emphasizing their importance in modern healthcare delivery. The home-care environments are projected to grow at a CAGR of 42.2%, reflecting the increasing demand for remote healthcare solutions.
-
Clinical Decision Support Systems (CDSS): CDSS tools assist healthcare professionals in making informed decisions based on individual data and clinical guidelines, thereby enhancing diagnostic accuracy and treatment effectiveness. The integration of AI in these systems is transforming how clinicians approach patient care.
-
Mobile Health Applications: Designed for user engagement, health monitoring, and wellness management, these applications are gaining popularity among consumers. They empower patients to take an active role in their health, contributing to better management of chronic conditions.
Understanding these categories enables developers and stakeholders to focus their efforts on the specific requirements and regulatory considerations relevant to each type, ultimately resulting in more effective technological solutions.
Ensure Regulatory Compliance and Standards
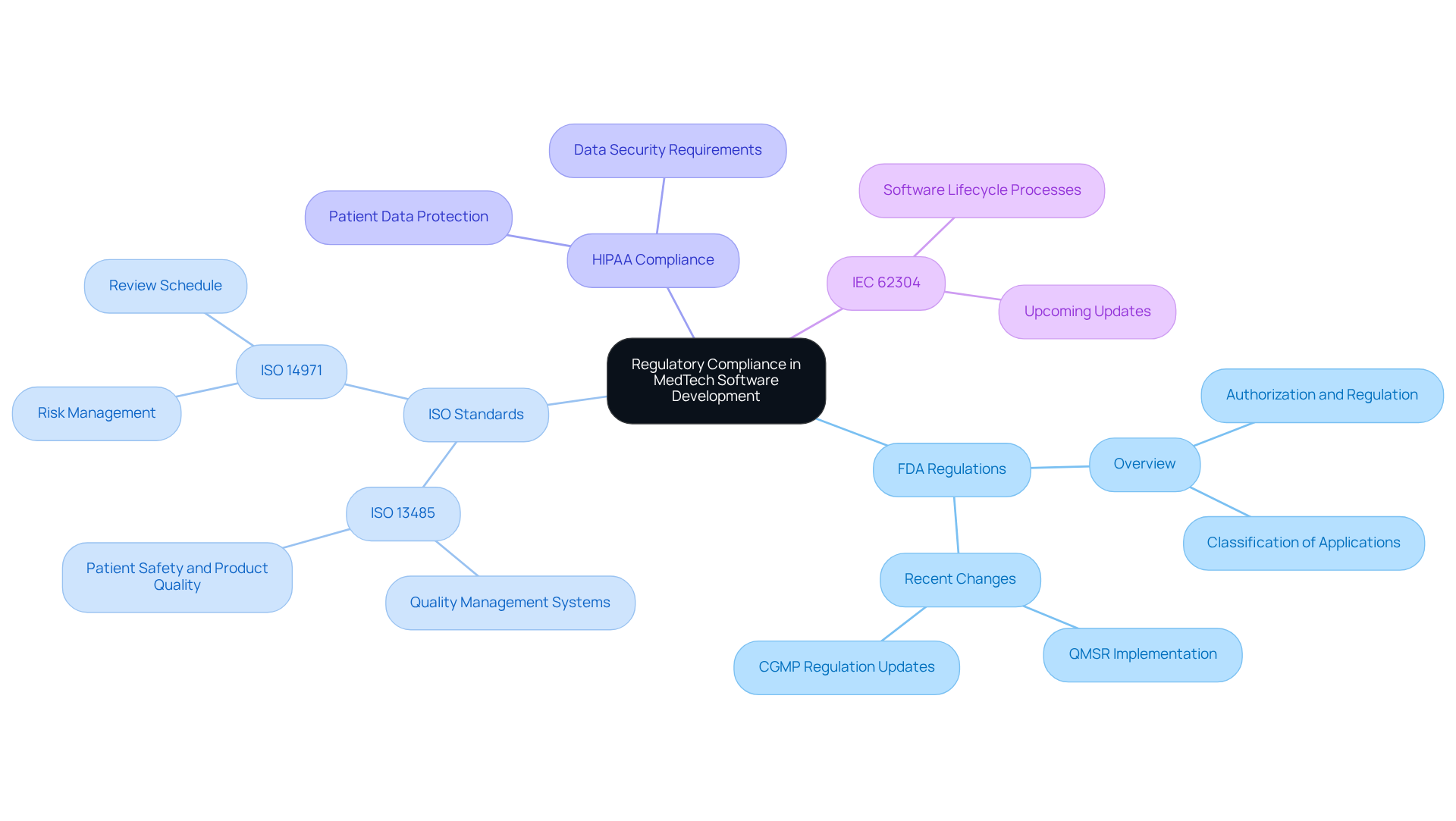
Regulatory compliance plays a vital role in medtech software development for MedTech applications. Key standards and regulations that developers must adhere to include:
- FDA Regulations: In the United States, the FDA oversees the authorization and regulation of medical devices, including applications. It is imperative for developers to comprehend the classification of their applications and to comply with the relevant guidelines.
- ISO Standards: Compliance with ISO 13485, which pertains to quality management systems, and ISO 14971, which focuses on risk management, is essential for ensuring the safety and effectiveness of products.
- HIPAA Compliance: For applications that handle patient data, adherence to HIPAA regulations is crucial to protect patient privacy and ensure data security.
- IEC 62304: This standard outlines the lifecycle processes for medical device programs, ensuring that creation, maintenance, and risk management are systematically addressed.
By integrating these compliance measures into medtech software development, companies can mitigate risks and enhance the reliability of their solutions.
Implement Best Practices in the Development Process
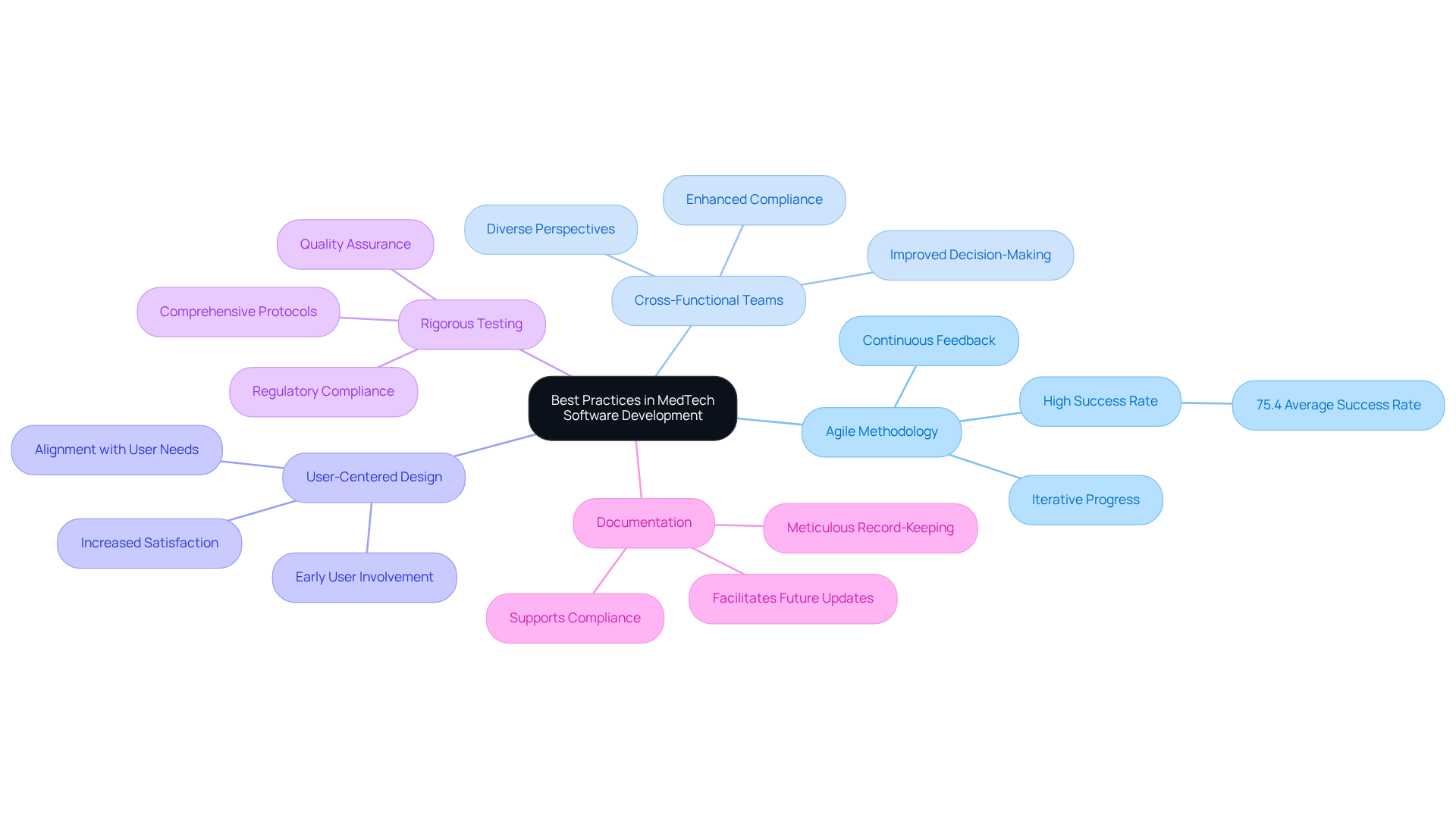
To ensure successful MedTech software development, consider the following best practices:
-
Agile Methodology: Embrace Agile practices to facilitate iterative progress, enabling continuous feedback and swift adaptation to evolving requirements. Agile methodologies have been shown to enhance project success rates, with organizations reporting an average success rate of 75.4%.
-
Cross-Functional Teams: Form teams that encompass developers, regulatory experts, and healthcare professionals. This diversity ensures that various perspectives and expertise are integrated into the development process, leading to more innovative solutions. Successful implementations have demonstrated that cross-functional collaboration significantly improves decision-making and compliance.
-
User-Centered Design: Involve end-users early in the design process to gather valuable insights and feedback. This engagement guarantees that the application aligns with needs and expectations, ultimately enhancing satisfaction and product effectiveness.
-
Rigorous Testing: Establish comprehensive testing protocols, including unit testing, integration testing, and user acceptance testing. These measures are crucial for identifying and addressing issues before deployment, thereby maintaining high quality and compliance standards. Neutech’s dedication to quality ensures that all digital solutions undergo thorough testing to meet industry standards.
-
Documentation: Maintain meticulous documentation throughout the development process. This practice supports compliance efforts and facilitates future updates or audits, ensuring that all regulatory requirements are met.
By following these best practices and utilizing Neutech’s extensive engineering services, developers can significantly enhance the quality and reliability of their medtech software development solutions, ultimately resulting in improved patient outcomes and adherence to industry standards.
Prioritize User Experience and Usability
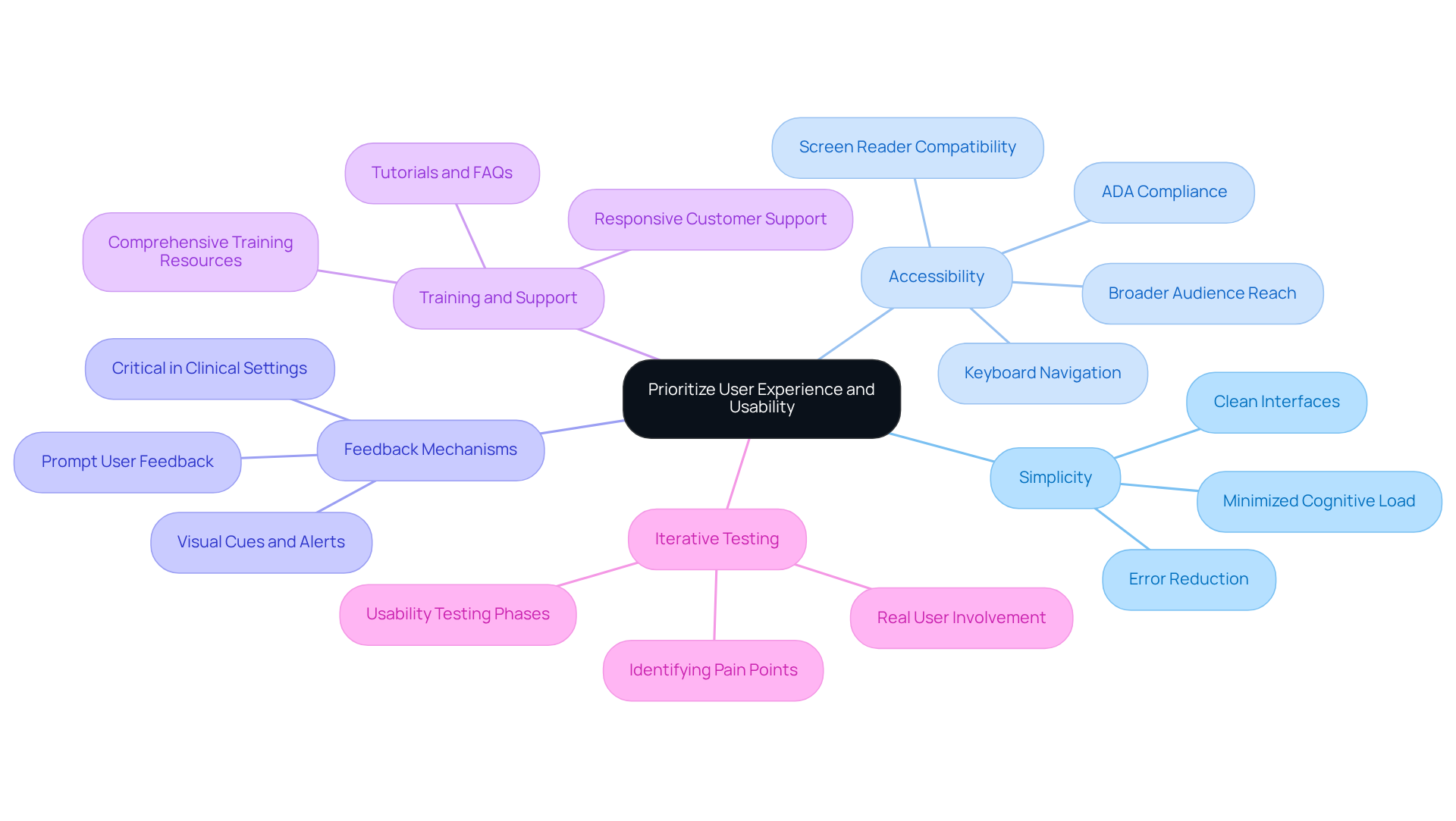
User experience (UX) and usability are essential elements of effective medtech software development. To enhance UX, consider the following strategies:
-
Simplicity: Design interfaces that are clean and straightforward, minimizing cognitive load for users who may be under stress. A simple design can significantly improve client satisfaction and reduce errors, particularly in high-pressure environments like healthcare.
-
Accessibility: Ensure the application is accessible to individuals with diverse abilities by adhering to standards such as the Americans with Disabilities Act (ADA). Features like screen reader compatibility and keyboard navigation can enhance usability for users with disabilities, ultimately broadening the audience and improving adoption rates. A case study on blindness and low vision highlights the importance of enhanced web accessibility for individuals with visual impairments, demonstrating how accessibility features can positively impact application adoption rates.
-
Feedback Mechanisms: Integrate features that provide users with prompt feedback on their actions, helping them understand the system’s responses. This may include visual cues or alerts that confirm successful actions, which are critical in clinical settings where timely information is essential.
-
Training and Support: Offer comprehensive training and support resources to assist users in navigating the software effectively. Providing tutorials, FAQs, and responsive customer support can significantly alleviate frustration and enhance confidence, leading to higher adoption rates.
-
Iterative Testing: Conduct usability testing throughout the development process to identify pain points and areas for improvement. Involving real users in testing phases allows developers to gather valuable insights and ensure the final product meets expectations and requirements.
By prioritizing UX and usability, developers can create medtech software development that not only complies with regulatory standards but also enhances the overall user experience. It is noteworthy that poor UX costs businesses an estimated $1.4 trillion annually, highlighting the financial implications of overlooking these critical components. Furthermore, developers should be mindful of common pitfalls, such as the tendency to overcomplicate designs in the quest for simplicity, which can ultimately hinder usability.
Conclusion
In the field of MedTech software development, it is crucial to understand the various categories of software and adhere to established best practices for achieving success. By concentrating on the distinct functions of:
- Software as a Medical Device
- Electronic Health Records
- Telemedicine Platforms
- Clinical Decision Support Systems
- Mobile Health Applications
stakeholders can effectively tailor their development efforts to address specific regulatory and user requirements, thereby enhancing healthcare delivery.
Key insights throughout this article underscore the significance of:
- Regulatory compliance
- The integration of Agile methodologies
- The prioritization of user experience
Developers are urged to implement rigorous testing protocols, maintain meticulous documentation, and promote cross-functional collaboration to ensure that their solutions are not only compliant but also innovative and effective. The financial repercussions of inadequate user experience further emphasize the necessity of designing intuitive and accessible software that caters to the needs of diverse users.
As the MedTech landscape continues to evolve, embracing these best practices will be essential for developers striving to create impactful software solutions. By prioritizing regulatory adherence, user-centered design, and continuous improvement, stakeholders can contribute to a future where technology enhances patient outcomes and transforms the healthcare experience. The call to action is clear: invest in quality development processes today to pave the way for a healthier tomorrow.
Frequently Asked Questions
What are the key types of MedTech software?
The key types of MedTech software include Software as a Medical Device (SaMD), Electronic Health Records (EHR), Telemedicine Platforms, Clinical Decision Support Systems (CDSS), and Mobile Health Applications.
What is Software as a Medical Device (SaMD)?
SaMD refers to applications that autonomously perform medical functions, such as diagnostic tools and monitoring systems for individuals. The market for SaMD is expected to grow significantly, with a projected CAGR of 37.62% toward 2031, largely due to advancements in AI and machine learning.
How do Electronic Health Records (EHR) systems benefit healthcare?
EHR systems manage patient data and facilitate communication among healthcare providers, which enhances care coordination and outcomes. Successful implementations, such as that of NYU Langone Health, have shown improved data submission adherence rates.
What role did the COVID-19 pandemic play in the adoption of Telemedicine Platforms?
The COVID-19 pandemic accelerated the adoption of telemedicine platforms, with 76% incorporating at least one SaMD feature. This highlights their importance in expanding access to healthcare services.
What are Clinical Decision Support Systems (CDSS) and their significance?
CDSS tools assist healthcare professionals in making informed decisions based on individual data and clinical guidelines, enhancing diagnostic accuracy and treatment effectiveness. The integration of AI is transforming how clinicians approach patient care.
What are Mobile Health Applications and their purpose?
Mobile Health Applications are designed for user engagement, health monitoring, and wellness management. They empower patients to take an active role in their health, contributing to better management of chronic conditions.
Why is understanding these categories of MedTech software important for developers and stakeholders?
Understanding these categories helps developers and stakeholders focus their efforts on the specific requirements and regulatory considerations relevant to each type, leading to more effective technological solutions.
