Introduction
The pharmaceutical industry faces a distinct array of challenges that necessitate precision, compliance, and innovation in software development. With regulatory standards becoming increasingly stringent and the demand for safe, effective products growing, the significance of adhering to best practices in pharmaceutical software development is paramount. This article delves into the essential strategies and frameworks that not only guarantee compliance with rigorous regulations but also bolster operational efficiency and data integrity.
How can pharmaceutical companies effectively navigate the complexities of software development while upholding the highest standards of quality and safety?
Define Pharmaceutical Software Development and Its Importance
The development of specialized applications designed to meet the specific needs of the drug industry encompasses drug discovery, clinical trials, regulatory compliance, and manufacturing processes. This advancement is essential for ensuring that technological solutions adhere to the stringent regulatory standards set by authorities such as the FDA and EMA. By addressing the unique challenges faced by the pharmaceutical sector, developers can create robust applications through pharmaceutical software development that not only facilitate compliance but also enhance operational efficiency and data integrity.
Effective technological solutions streamline workflows, ultimately contributing to improved patient safety and product efficacy. For example, AI-driven validation tools have demonstrated a significant reduction in human errors and a faster audit process, thereby ensuring compliance with quality regulations. Additionally, the integration of insights management platforms fosters standardized and predictable processes for gathering critical data, which is vital for regulatory adherence.
Neutech specializes in various programming skills, including React, Python, and AWS DevOps, which are crucial for developing efficient healthcare applications. In summary, the significance of pharmaceutical software development in creating medical applications lies in their ability to address the complex needs of the industry while enhancing efficiency and safety in drug development and clinical operations.
Identify Key Regulations and Standards for Compliance
Key regulations and standards governing pharmaceutical application development include the FDA’s 21 CFR Part 11. This regulation establishes requirements for electronic records and signatures, ensuring their trustworthiness is on par with traditional paper records. Adherence to these regulations is essential, as they mandate that systems must be validated to uphold data integrity and reliability.
Furthermore, Good Automated Manufacturing Practice (GxP) guidelines play a crucial role in ensuring that system applications conform to industry standards. These guidelines encompass Good Manufacturing Practices (GMP), Good Laboratory Practices (GLP), and Good Clinical Practices (GCP). Non-compliance with these regulations can lead to severe consequences, including product recalls, legal penalties, and loss of market access.
As industry specialists note, “Validation in the drug sector is typically conducted as a part of GAMP’s V-model,” which emphasizes the organized method necessary for compliance. Organizations must ensure that their pharmaceutical software development adheres to these guidelines to avoid operational disruptions and maintain audit readiness.
In pharmaceutical software development, the integration of robust validation processes not only aids in meeting regulatory requirements but also enhances the overall quality and safety of pharmaceutical products. Additionally, systems must produce secure, computer-generated, time-stamped audit trails for electronic records, a critical aspect of adhering to FDA 21 CFR Part 11.
The FDA’s revised guidance released in January 2026 further clarifies these requirements, underscoring the importance of following established frameworks in regulatory efforts.
Leverage Best Practices for Enhanced Efficiency and Compliance
To enhance efficiency and adherence in pharmaceutical software development, implementing a robust Software Development Life Cycle (SDLC) is essential. This SDLC should encompass thorough documentation, regular validation, and effective risk management strategies. Agile methodologies are crucial in this context, as they facilitate iterative processes and continuous feedback, enabling teams to swiftly adapt to evolving regulatory requirements. Neutech, with its extensive engineering services, focuses on programming designed for regulated sectors, including medicine. Their expertise spans various technologies such as React, Python, GoLang, Android Development, and AWS DevOps, ensuring they excel in pharmaceutical software development to meet the specific needs of pharmaceutical applications.
For instance, firms like Pfizer and Novartis have effectively incorporated Agile practices, showcasing significant enhancements in their production processes. The integration of automated testing and validation tools further streamlines compliance processes, ensuring applications adhere to necessary standards prior to deployment. Real-world applications, such as the implementation of Continuous Integration/Continuous Deployment (CI/CD) pipelines in leading medical firms, exemplify how these practices can significantly reduce delivery times and enhance program quality. This ultimately leads to more efficient and compliant production cycles. However, it is important to acknowledge the challenges associated with Agile adoption in regulated environments, including cultural resistance and siloed structures, which can impede progress. Addressing these challenges is crucial for leveraging Agile methodologies effectively.
Implement Strategies for Effective Software Development
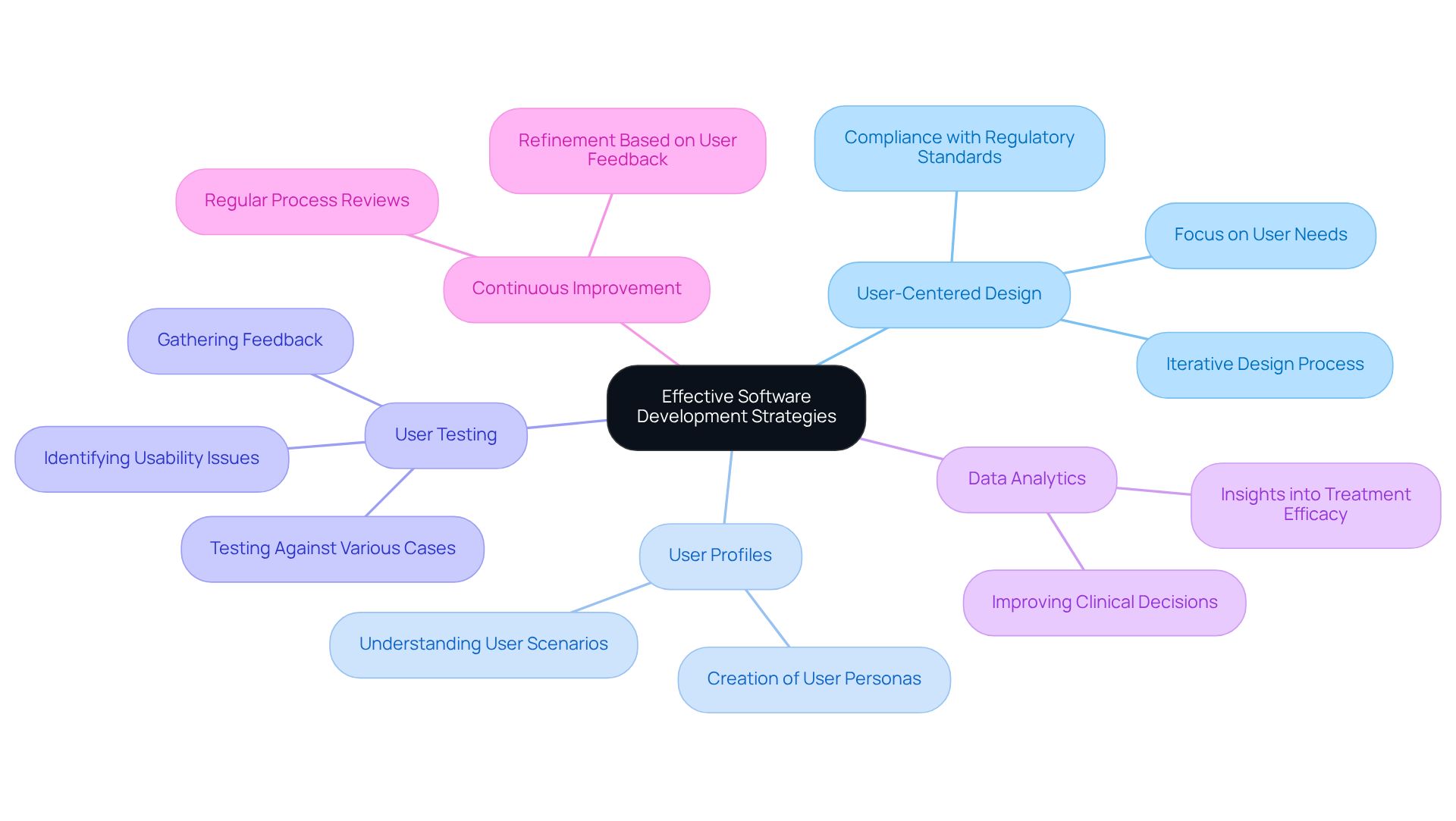
To implement effective pharmaceutical software development strategies in the pharmaceutical sector, teams must prioritize user-centered design (UCD) principles. This approach ensures that applications are intuitive and tailored to the specific needs of healthcare professionals and patients.
Creating user profiles or personas assists design teams in empathizing with users, allowing for a more targeted design process. Conducting thorough user testing is essential; it enables teams to gather valuable feedback and identify potential issues early in the development process. For instance, user testing can unveil critical insights into how medical personnel engage with applications, leading to enhancements that improve usability and reduce errors.
Incorporating data analytics capabilities into application solutions further enhances decision-making and patient outcomes. By leveraging analytics, healthcare providers can gain insights into treatment efficacy and patient behavior, ultimately leading to more informed clinical decisions.
Establishing a culture of continuous improvement is vital, where teams regularly review and refine their processes based on user feedback and performance metrics. This iterative approach fosters innovation and ensures that pharmaceutical software development remains compliant with regulatory standards, including expectations for patient-centric medical device design, effectively meeting the evolving needs of the healthcare sector.
Additionally, integrating UCD into Quality Management Systems (QMS) addresses challenges in agile practices, ensuring that user needs are consistently prioritized throughout the development lifecycle.
Conclusion
The development of pharmaceutical software plays a crucial role in ensuring that the drug industry adheres to stringent regulatory standards while enhancing operational efficiency. By concentrating on specialized applications that address the unique challenges of drug discovery, clinical trials, and manufacturing processes, organizations can significantly bolster compliance and patient safety. This underscores the necessity of integrating robust validation practices and complying with key regulations, such as the FDA’s 21 CFR Part 11 and GxP guidelines, which are vital for maintaining data integrity and reliability in pharmaceutical applications.
Key insights reveal the importance of implementing a comprehensive Software Development Life Cycle (SDLC) that incorporates Agile methodologies, user-centered design principles, and continuous improvement. These strategies not only streamline compliance processes but also enable rapid adaptation to evolving regulatory requirements. Real-world examples from leading firms illustrate that adopting automated testing, CI/CD pipelines, and thorough user testing can enhance the quality and efficiency of pharmaceutical software development, ultimately resulting in improved patient outcomes and reduced operational risks.
In conclusion, the importance of adhering to best practices in pharmaceutical software development is paramount. As the industry continues to evolve, embracing innovative technologies and methodologies will be essential in addressing compliance challenges and enhancing the overall quality of healthcare solutions. Stakeholders in the pharmaceutical sector must prioritize these strategies to ensure that their software development processes align with regulatory expectations while effectively meeting the needs of healthcare professionals and patients alike.
Frequently Asked Questions
What is pharmaceutical software development?
Pharmaceutical software development involves creating specialized applications tailored to meet the specific needs of the drug industry, including areas such as drug discovery, clinical trials, regulatory compliance, and manufacturing processes.
Why is pharmaceutical software development important?
It is essential for ensuring that technological solutions comply with stringent regulatory standards set by authorities like the FDA and EMA, while also enhancing operational efficiency and data integrity within the pharmaceutical sector.
How do technological solutions in pharmaceutical software development improve patient safety?
Effective technological solutions streamline workflows, which contributes to improved patient safety by reducing errors and ensuring compliance with quality regulations.
What role do AI-driven validation tools play in pharmaceutical software development?
AI-driven validation tools help reduce human errors and expedite the audit process, ensuring compliance with quality regulations in the pharmaceutical industry.
What are insights management platforms, and how do they contribute to pharmaceutical software development?
Insights management platforms foster standardized and predictable processes for gathering critical data, which is vital for adhering to regulatory requirements in the pharmaceutical sector.
What programming skills are essential for developing healthcare applications in pharmaceutical software development?
Key programming skills include React, Python, and AWS DevOps, which are crucial for creating efficient healthcare applications.
How does pharmaceutical software development enhance efficiency in drug development and clinical operations?
By addressing the complex needs of the industry and creating robust applications, pharmaceutical software development enhances efficiency and safety throughout the drug development and clinical operations processes.
List of Sources
- Define Pharmaceutical Software Development and Its Importance
- How pharma can transform compliance (https://fastcompany.com/91475687/how-pharma-can-transform-compliance)
- Pharma compliance software | Within3 (https://within3.com/blog/pharma-compliance-software)
- Pharma Software – GxP and Regulatory Compliance Guide (https://emorphis.health/blogs/pharma-software-gxp-regulatory-compliance)
- The Need for Software Validation in Pharmaceutical Manufacturing (https://nestorbird.com/need-for-software-validation-in-pharmaceutical-manufacturing)
- AI is Revolutionizing Compliance in the Pharmaceutical Industry (https://pharmatechoutlook.com/news/ai-is-revolutionizing-compliance-in-the-pharmaceutical-industry-nwid-3469.html)
- Identify Key Regulations and Standards for Compliance
- Navigating GxP Compliance in Software and Data Services Development: What Is It and What To Be Aware Of? (https://fptsoftware.com/resource-center/blogs/navigating-gxp-compliance-in-software-and-data-services-development)
- FDA Implements Quality Management System Regulation for Medical Devices (https://aabb.org/news-resources/news/article/2026/02/04/fda-implements-quality-management-system-regulation-for-medical-devices)
- FDA’s 2026 Guidance Expands Pathway for Low-Risk Digital Health Products—But Caution Remains Essential | Berkley Lifesciences (https://berkleyls.com/blog/fdas-2026-guidance-expands-pathway-low-risk-digital-health-products-caution-remains-essential)
- Software development for the pharmaceutical industry (https://biosistemika.com/blog/software-development-for-the-pharmaceutical-industry-2)
- Low-Code Pharma Platforms: Building GxP-Compliant Systems | IntuitionLabs (https://intuitionlabs.ai/articles/low-code-pharma-gxp-mes-qms)
- Leverage Best Practices for Enhanced Efficiency and Compliance
- Agile Methodologies in Pharma. Moving Beyond Traditional Drug Development Pipelines (https://pharmanow.live/leadership/agile-in-the-pharmaceutical-industry-from-rigid-pipelines-to-adaptive-innovation)
- Top 10 Pharma Companies Using Agile Methodology to Drive Innovation (https://pharmanow.live/leadership/top-pharma-companies-using-agile-methodology)
- Unlocking Agility: Driving Innovation in Pharma with Agile Planning (https://uptakestrategies.com/unlocking-agility-driving-innovation-in-pharma-with-agile-planning)
- Agile Regulatory Strategies for a Dynamic Pharma Landscape (https://freyrsolutions.com/blog/agile-regulatory-strategies-for-a-dynamic-pharmaceutical-landscape)
- The statistical software revolution in pharmaceutical development: challenges and opportunities in open source – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC12989278)
- Implement Strategies for Effective Software Development
- Developing software for a pharmaceutical company? Here’s why user testing is critical. — SciMed (https://scimed.io/news/developing-software-for-a-pharmaceutical-company-heres-why-user-testing-is-critical)
- User-centred development of medical software: The key role of human-centered Design (https://adesso.de/en/news/blog/user-centred-development-of-medical-software-the-key-role-of-human-centered-design.jsp)
- News & Insights | iO Life Science (https://iolifescience.com/news/the-critical-role-of-user-centered-design-in-medtech-innovation)
- The Importance of User-Centered Design in Developing Medical Software (https://kandasoft.com/blog/user-centered-design-in-developing-medical-software)
- htdhealth.com (https://htdhealth.com/insights/software-as-medical-device-guide-to-implementing-user-centered-design)