Introduction
The rapid evolution of technology in healthcare has given rise to Software as a Medical Device (SaMD), fundamentally changing how medical applications function independently of traditional hardware. As developers and manufacturers navigate the complex landscape of regulatory compliance, grasping the nuances of SaMD is essential for ensuring product safety and efficacy. However, with regulations continuously evolving and the integration of advanced technologies such as AI and machine learning, organizations face the challenge of effectively balancing innovation with stringent compliance requirements.
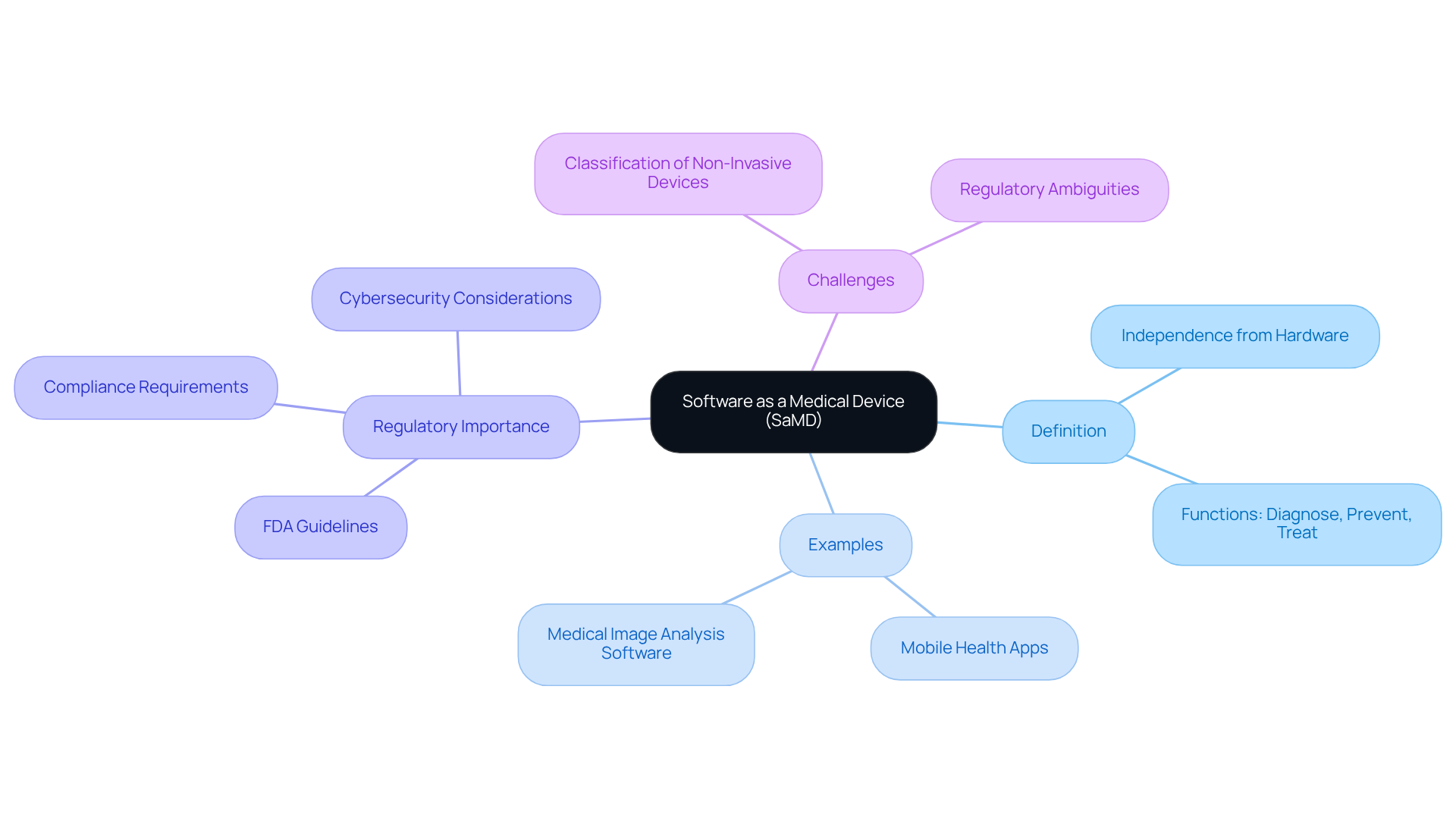
Define Software as a Medical Device (SaMD)
Applications as a Medical Device (SaMD) refers to software programs designed for medical purposes that fulfill these roles independently of any hardware medical device. According to the FDA and the International Medical Device Regulators Forum (IMDRF), SaMD encompasses applications that diagnose, prevent, or treat diseases, functioning autonomously on general-purpose computing platforms. Notable examples include mobile applications that monitor health metrics, such as heart rate or glucose levels, and software that analyzes medical images for diagnostic purposes.
Understanding this definition is crucial for developers and manufacturers to ensure compliance with regulatory standards. In 2026, the FDA approved a range of SaMD applications, highlighting the increasing integration of software solutions within healthcare. As emphasized by the FDA, “the guidance aims for consistency in how FDA evaluates cybersecurity aspects, ensuring devices remain ‘sufficiently resilient to cybersecurity threats.'” Adhering to the latest FDA guidelines on SaMD, updated in January 2026, is vital for maintaining product quality and ensuring patient safety.
Moreover, challenges arise in classifying non-invasive blood glucose monitors as general wellness devices, underscoring the importance of strict compliance with regulatory requirements.
Understand Regulatory Compliance for SaMD
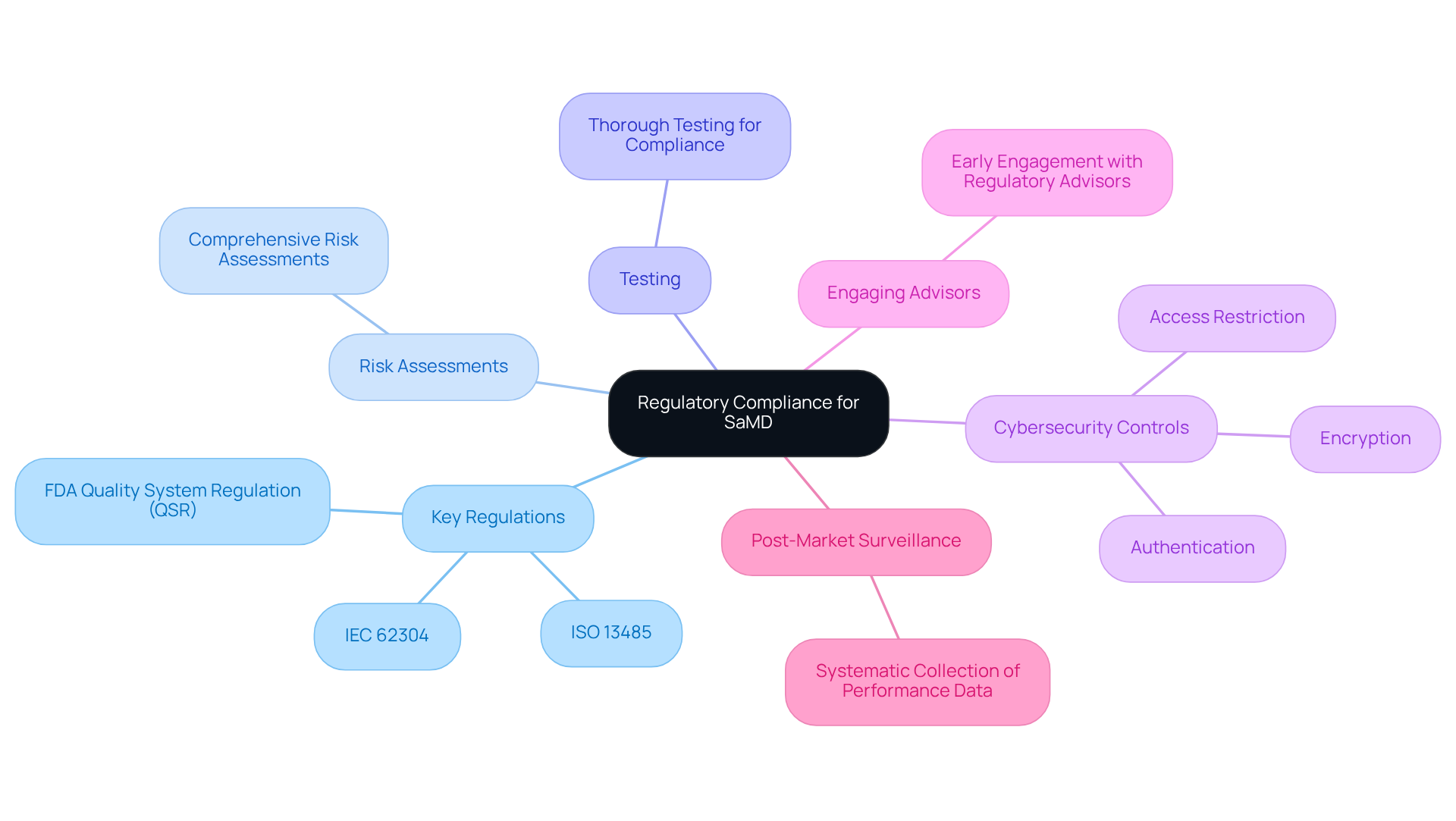
Regulatory compliance for software for medical devices is crucial for ensuring safety and efficacy in healthcare applications. Adhering to guidelines established by regulatory organizations such as the FDA and the European Medicines Agency (EMA) is imperative. Key regulations include:
- The FDA’s Quality System Regulation (QSR)
- ISO 13485 for quality management systems
- IEC 62304, which delineates lifecycle processes for applications
The FDA’s 2026 guidance broadens pathways for low-risk digital health products, underscoring the necessity for caution in product development.
Companies must perform comprehensive risk assessments and maintain meticulous documentation throughout the development process. Thorough testing is essential to demonstrate compliance, ensuring that the software meets both safety and performance standards. Furthermore, mandatory cybersecurity controls – such as authentication, access restriction, and encryption – must be integrated during development to safeguard sensitive data.
Engaging regulatory advisors early in the development stage can significantly streamline adherence efforts, ultimately reducing time to market while enhancing product reliability and user trust. Additionally, post-market surveillance is vital, involving the systematic collection and analysis of performance data post-launch to ensure ongoing compliance and safety.
Understanding the distinction between applications that assist healthcare providers’ decision-making and those that substitute or guide it is also critical for navigating regulatory classifications. Incorporating these elements provides a comprehensive overview of the regulatory requirements for software for medical devices.
Implement Quality Assurance Practices
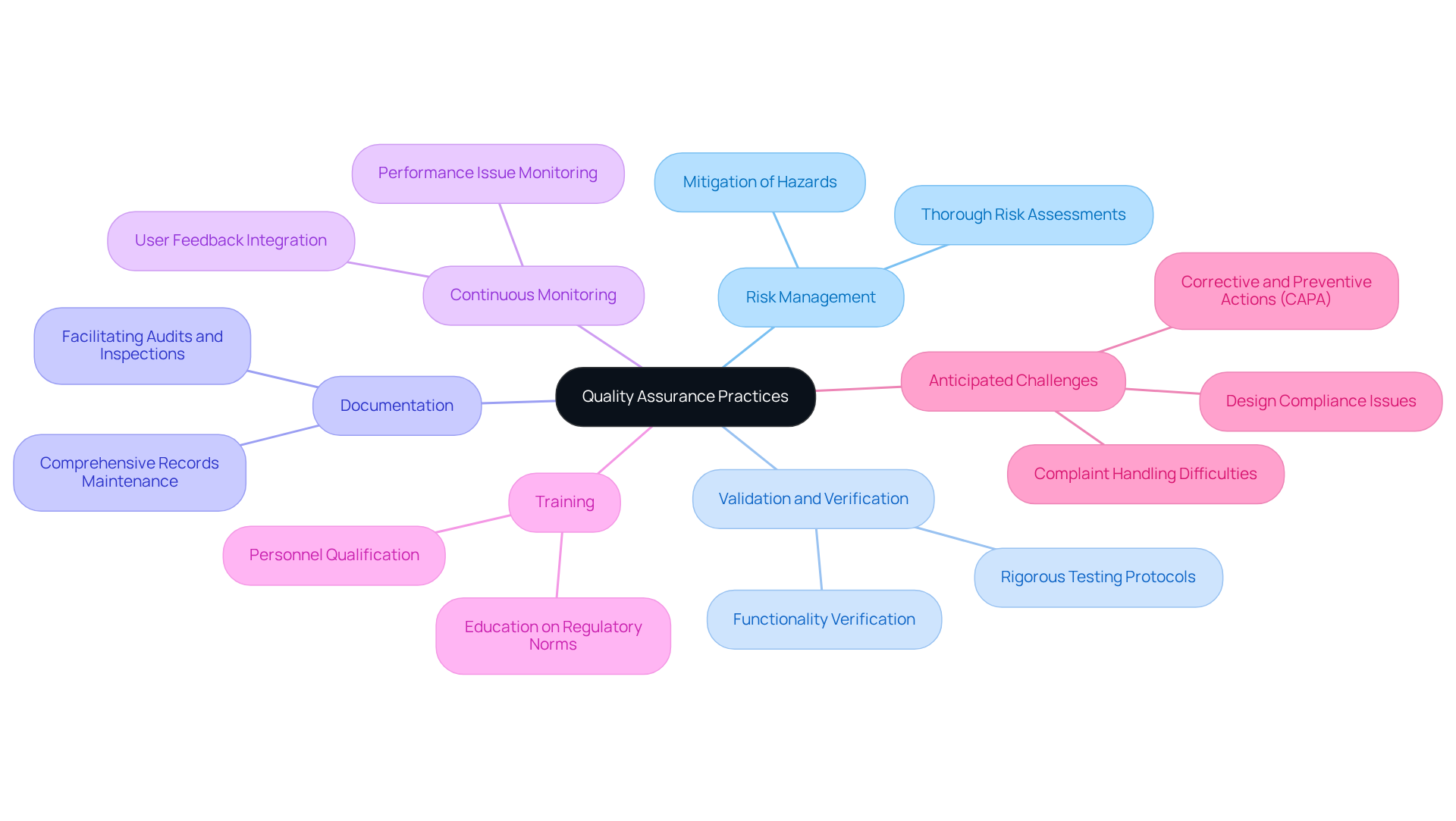
Quality assurance (QA) practices for software for medical devices as a service (SaMD) must be seamlessly integrated throughout the application development lifecycle. Establishing a Quality Management System (QMS) that adheres to ISO 13485 standards is essential for ensuring conformity and safety, especially in light of the new FDA regulation effective February 2, 2026, which aligns U.S. medical device quality management standards, including those for software for medical devices, with global requirements.
Key QA practices include:
- Risk Management: Conducting thorough risk assessments is crucial for identifying and mitigating potential hazards associated with the application. This proactive approach safeguards patient safety and ensures that the software for medical devices complies with regulations.
- Validation and Verification: Implementing rigorous testing protocols is necessary to validate that the system meets its intended use and to verify its functionality under various conditions. This step is vital for demonstrating the reliability and effectiveness of the software for medical devices.
- Documentation: Maintaining comprehensive records of all QA processes, test results, and regulatory activities is essential. This documentation facilitates audits and inspections, ensuring transparency and accountability throughout the development process of software for medical devices.
- Continuous Monitoring: After deployment, it is imperative to continuously monitor the software for performance issues and user feedback. This ongoing vigilance ensures sustained adherence and safety in the software for medical devices, allowing for timely updates and improvements as necessary.
- Training: Ensuring that all personnel involved in the QA process are adequately educated in regulatory norms and practices is critical. Training is a fundamental component of the quality system in regulated industries, especially in the context of software for medical devices, helping to uphold high standards of quality and safety.
- Anticipated Challenges: It is important to recognize that, despite the introduction of the QMSR, common adherence issues related to design, Corrective and Preventive Actions (CAPA), and complaint handling are likely to persist. Proactively addressing these challenges related to software for medical devices can mitigate risks associated with regulatory scrutiny.
Integrating these practices not only enhances adherence but also fosters a culture of quality within the organization, ultimately leading to improved patient outcomes.
Leverage AI and Machine Learning in Compliance
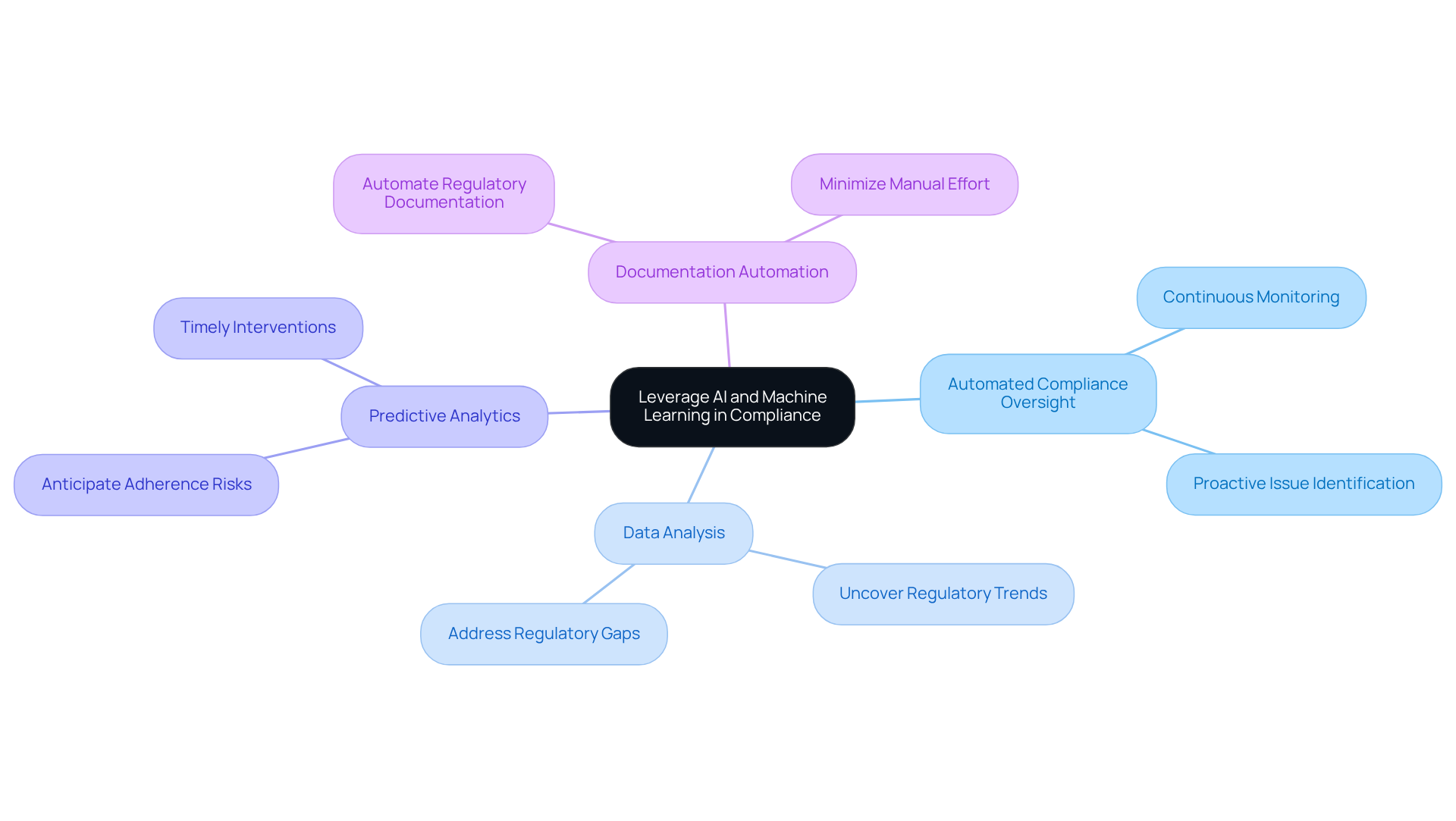
Incorporating AI and machine learning into the regulatory processes for software for medical devices can significantly enhance both efficiency and accuracy. This integration offers several key practices that organizations should consider:
- Automated Compliance Oversight: AI algorithms can be employed to continuously monitor software performance and ensure conformity to regulatory standards. This proactive approach allows for the identification of potential issues before they escalate.
- Data Analysis: Machine learning can analyze extensive datasets to uncover regulatory trends. This capability assists organizations in proactively addressing possible regulatory gaps, thereby enhancing compliance.
- Predictive Analytics: By implementing predictive analytics, organizations can anticipate adherence risks based on historical data. This foresight enables timely interventions, reducing the likelihood of compliance failures.
- Documentation Automation: AI tools can automate the generation of regulatory documentation, which minimizes manual effort and reduces the potential for errors.
By adopting software for medical devices, organizations can streamline their compliance processes and significantly enhance their overall quality management efforts.
Conclusion
The integration of software into medical devices signifies a critical shift in healthcare, where adherence to compliance and quality standards is essential. By defining Software as a Medical Device (SaMD) and comprehending its regulatory landscape, developers and manufacturers can ensure their products not only meet safety requirements but also improve patient outcomes. The emphasis on adhering to FDA guidelines and the significance of a comprehensive Quality Management System (QMS) are vital, as these components are crucial for maintaining the integrity and effectiveness of medical software.
Key practices have been underscored throughout this discussion, including the necessity for rigorous risk assessments, thorough documentation, and continuous monitoring to uphold quality assurance. Furthermore, the application of AI and machine learning can significantly streamline compliance processes, allowing organizations to proactively address potential regulatory issues and enhance overall product quality. These insights illustrate the multifaceted approach required to successfully navigate the complexities of software in medical devices.
In conclusion, the journey toward achieving compliance and quality in medical device software is intricate yet indispensable. Organizations must adopt best practices, invest in robust quality systems, and leverage technological advancements to remain competitive in a rapidly evolving landscape. Ultimately, prioritizing these elements will not only ensure regulatory adherence but also cultivate trust and safety in healthcare applications, paving the way for innovation and improved patient care.
Frequently Asked Questions
What is Software as a Medical Device (SaMD)?
Software as a Medical Device (SaMD) refers to software programs designed for medical purposes that can operate independently of any hardware medical device. It includes applications that diagnose, prevent, or treat diseases using general-purpose computing platforms.
What are some examples of SaMD?
Notable examples of SaMD include mobile applications that monitor health metrics such as heart rate or glucose levels, as well as software that analyzes medical images for diagnostic purposes.
Why is the definition of SaMD important for developers and manufacturers?
Understanding the definition of SaMD is crucial for developers and manufacturers to ensure compliance with regulatory standards and maintain product quality and patient safety.
What recent developments have occurred regarding SaMD applications?
In 2026, the FDA approved a range of SaMD applications, indicating an increasing integration of software solutions within healthcare.
What does the FDA emphasize about SaMD and cybersecurity?
The FDA emphasizes that the guidance aims for consistency in evaluating cybersecurity aspects of SaMD, ensuring that devices remain sufficiently resilient to cybersecurity threats.
When were the latest FDA guidelines on SaMD updated?
The latest FDA guidelines on SaMD were updated in January 2026.
What challenges exist in classifying certain medical devices?
Challenges arise in classifying non-invasive blood glucose monitors as general wellness devices, highlighting the importance of strict compliance with regulatory requirements.
List of Sources
- Define Software as a Medical Device (SaMD)
- Software as a Medical Device (SaMD) Market Size, Trends, Growth Report 2032 (https://databridgemarketresearch.com/reports/global-software-as-a-medical-device-samd-market?srsltid=AfmBOoqHPGalOftT8a99SgkgKTq5RL5uCE3-01a70wbMiYonGND-4Tt6)
- FDA Updates Broaden Wellness and Clinical Decision Support Software Devices Under Enforcement (https://emergobyul.com/news/fda-updates-broaden-wellness-and-clinical-decision-support-software-devices-under-enforcement)
- FDA Digital Health Guidance: 2026 Requirements Overview | IntuitionLabs (https://intuitionlabs.ai/articles/fda-digital-health-technology-guidance-requirements)
- FDA’s 2026 Guidance Expands Pathway for Low-Risk Digital Health Products—But Caution Remains Essential | Berkley Lifesciences (https://berkleyls.com/blog/fdas-2026-guidance-expands-pathway-low-risk-digital-health-products-caution-remains-essential)
- FDA’s 2026 Guidance on General Wellness Devices: Policy for Low-Risk Devices— Key Compliance and Regulatory Insights for Digital Health Companies – Kendall PC (https://kendallpc.com/fdas-2026-guidance-on-general-wellness-devices-policy-for-low-risk-devices-key-compliance-and-regulatory-insights-for-digital-health-companies)
- Understand Regulatory Compliance for SaMD
- FDA’s 2026 Guidance Expands Pathway for Low-Risk Digital Health Products—But Caution Remains Essential | Berkley Lifesciences (https://berkleyls.com/blog/fdas-2026-guidance-expands-pathway-low-risk-digital-health-products-caution-remains-essential)
- FDA’s 2026 Guidance on General Wellness Devices: Policy for Low-Risk Devices— Key Compliance and Regulatory Insights for Digital Health Companies – Kendall PC (https://kendallpc.com/fdas-2026-guidance-on-general-wellness-devices-policy-for-low-risk-devices-key-compliance-and-regulatory-insights-for-digital-health-companies)
- For 2026, FDA signals shifts in digital health framework | Nixon Peabody LLP (https://nixonpeabody.com/insights/alerts/2026/01/27/for-2026-fda-signals-shifts-in-digital-health-framework)
- Medical Device Software Development (2026 Guide) – CLEIO (https://cleio.com/insights/blog/medical-device-software-development)
- Implement Quality Assurance Practices
- FDA’s Computer Software Assurance 2026: Changes and What to Do Next | PSC Software (https://pscsoftware.com/resource-center/article/fda-computer-software-assurance-2026-changes)
- FDA’s 2026 Guidance Expands Pathway for Low-Risk Digital Health Products—But Caution Remains Essential | Berkley Lifesciences (https://berkleyls.com/blog/fdas-2026-guidance-expands-pathway-low-risk-digital-health-products-caution-remains-essential)
- A QMSR State of Mind: FDA Adopts New Inspection Approach for Medical Devices as Quality Management System Regulation Takes Effect | Insights | Ropes & Gray LLP (https://ropesgray.com/en/insights/alerts/2026/02/a-qmsr-state-of-mind-fda-adopts-new-inspection-approach-for-medical-devices)
- FDA Aligns U.S. Medical Device Rules with Global Standards (https://mddionline.com/regulatory-quality/medical-device-companies-must-navigate-historic-fda-rule-change)
- Artificial Intelligence-Based Software as a Medical Device (AI-SaMD): A Systematic Review – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11988595)
- Leverage AI and Machine Learning in Compliance
- Tracking How AI Will Impact the Medical Device Industry (https://mddionline.com/artificial-intelligence/tracking-how-ai-will-impact-the-medical-device-industry-healthcare)
- AI Adoption In Healthcare Is Surging: What A New Report Reveals (https://forbes.com/sites/sachinjain/2025/10/21/ai-adoption-in-healthcare-is-surging-what-a-new-report-reveals)
- Post-Market Surveillance for SaMD: Real-World Data, Wearable Integration, Adverse Events & Continuous Monitoring | Freyr – Global Regulatory Solutions and Services Company (https://freyrsolutions.com/blog/post-market-surveillance-for-samd-real-world-data-wearable-integration-adverse-events-continuous-monitoring)
- FDA Updates Broaden Wellness and Clinical Decision Support Software Devices Under Enforcement (https://emergobyul.com/news/fda-updates-broaden-wellness-and-clinical-decision-support-software-devices-under-enforcement)
- AI in Healthcare 2025 Statistics: Market Size, Adoption, Impact (https://ventionteams.com/healthtech/ai/statistics)
