Introduction
Navigating the complex landscape of medical equipment software compliance poses significant challenges for developers and organizations. With numerous regulatory requirements from entities such as the FDA and ISO, grasping the nuances of these guidelines is essential for ensuring safety and efficacy in medical software. This article explores four best practices that not only streamline compliance efforts but also improve the overall quality of medical applications.
How can organizations effectively balance strict adherence to regulations with the necessity for innovation in a rapidly evolving healthcare environment?
Understand Regulatory Requirements for Medical Software
Compliance in medical equipment applications relies heavily on a comprehensive understanding of the regulatory landscape, which includes FDA guidelines, ISO standards, and HIPAA requirements. Each regulation specifies criteria that programs must meet to ensure safety and efficacy. For example, the FDA requires that programs classified as medical devices undergo thorough premarket evaluation. In contrast, ISO standards provide frameworks for quality management systems, encompassing risk management and clinical investigation protocols.
The updates to ISO 14155 in 2026 enhance clarity for producers by aligning more closely with ICH guidelines, thereby facilitating adherence to legal limits. To keep development teams informed about these evolving regulations, frequent training sessions and workshops are crucial. This ensures that software not only complies with regulatory standards but is also market-ready.
Companies like Veranex exemplify effective navigation of FDA guidelines by integrating comprehensive trial data into compliance strategies, which supports both approval and market adoption. Statistics indicate that adherence to ISO standards can significantly reduce the risk of regulatory scrutiny. For instance, the rate of Adverse Device Effects (ADEs) during clinical investigations can differ markedly from post-market use, underscoring the necessity of stringent adherence practices.
Implement Rigorous Testing Protocols
To achieve adherence, medical equipment software must undergo a comprehensive range of evaluation protocols, which include:
- Unit evaluation
- Integration evaluation
- System evaluation
- User acceptance evaluation (UAT)
Each phase necessitates meticulous documentation to substantiate compliance efforts. Unit verification focuses on ensuring that individual components function correctly, while integration assessment verifies that these components interact seamlessly. The integration of automated assessment tools is essential for enhancing both efficiency and accuracy throughout the evaluation process. For example, automated regression evaluation is increasingly utilized in healthcare applications, showcasing its effectiveness in minimizing errors and improving system reliability. Current trends indicate that medical equipment software, especially automated testing tools, are becoming indispensable in healthcare application development, with a significant shift towards their adoption anticipated in 2026. This reflects the industry’s commitment to maintaining high regulatory standards and enhancing overall application quality.
Establish Continuous Monitoring and Feedback Mechanisms
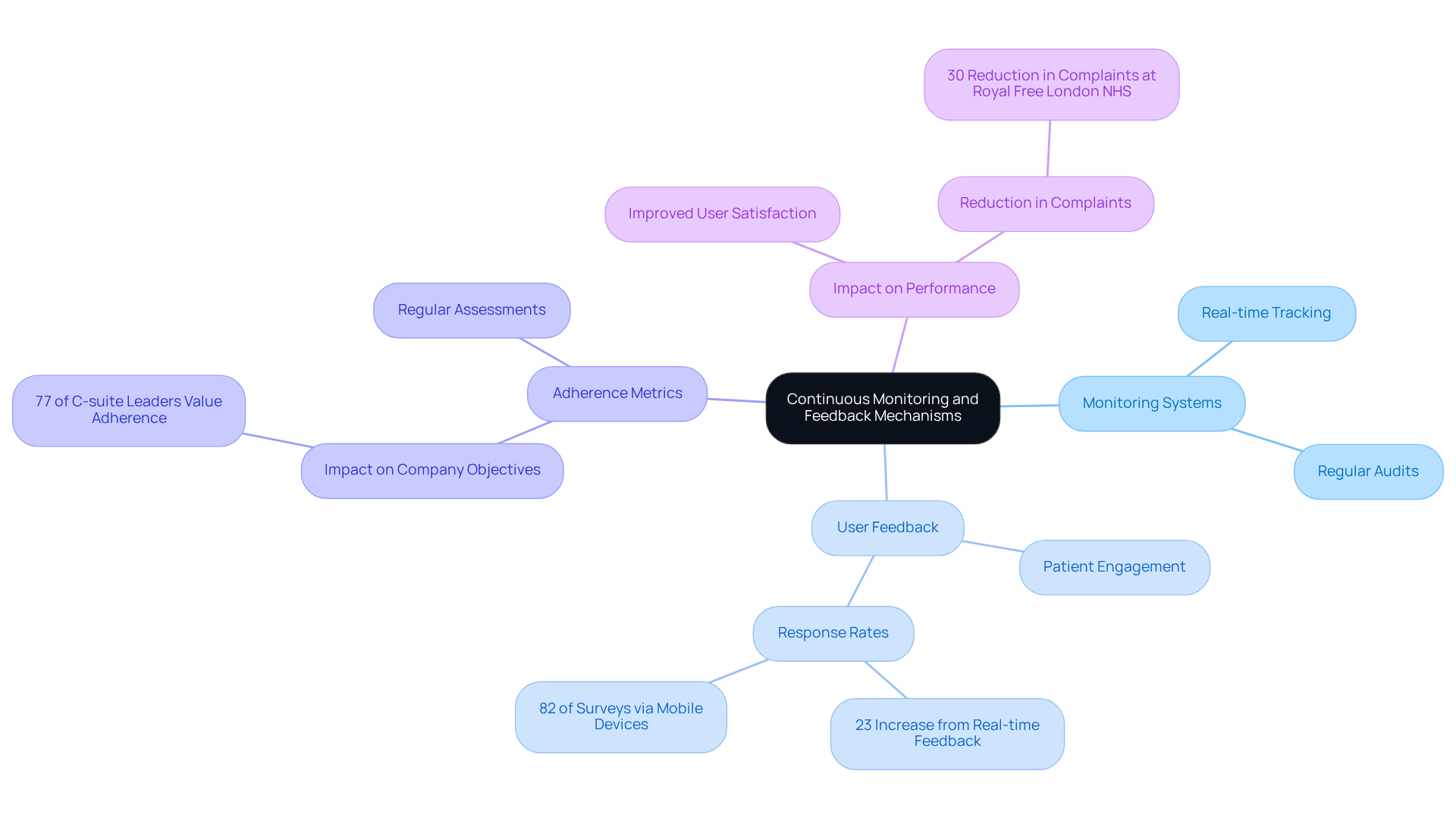
Ongoing monitoring and feedback systems are essential for maintaining regulatory standards in healthcare applications. A robust monitoring system enables organizations to track application performance and user feedback in real-time, allowing for prompt responses to any deviations from expected performance metrics or regulatory standards. For example, healthcare organizations that actively engage with patient feedback to enhance software features often experience increased user satisfaction and improved adherence outcomes. Data indicates that employing real-time feedback techniques can lead to a 23% increase in response rates, with 82% of surveys submitted via mobile devices, underscoring the significance of user involvement in adherence initiatives.
Moreover, 77% of global C-suite leaders assert that adherence plays a significant or moderate role in achieving company objectives, further emphasizing its critical importance in healthcare technology investments. Regular audits and assessments of adherence metrics are vital to ensure that the system consistently aligns with legal requirements, thereby fostering a culture of accountability and continuous improvement. Delayed responses to feedback can exacerbate patient concerns, making timely actions crucial. For instance, the Royal Free London NHS Foundation Trust implemented real-time feedback systems, resulting in a 30% reduction in formal complaints within six months, illustrating the effectiveness of such mechanisms.
Foster Cross-Functional Collaboration for Compliance
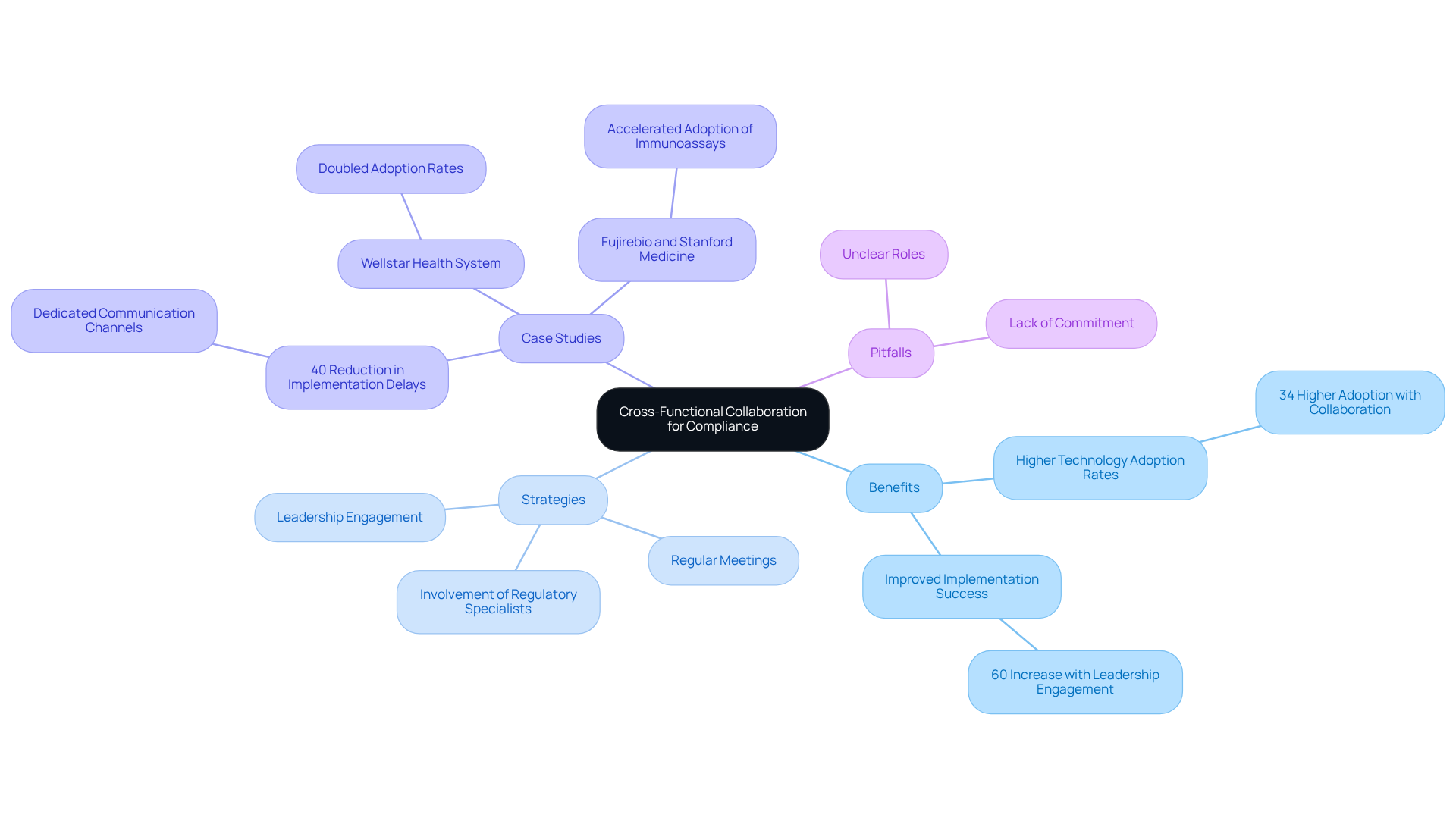
Encouraging cross-functional teamwork is essential for achieving adherence in medical equipment software development. By integrating knowledge from various divisions – such as engineering, oversight affairs, and quality assurance – organizations can formulate a more comprehensive strategy for adherence. Regular meetings and collaborative platforms enhance communication, ensuring that all team members are aligned on regulatory objectives.
Involving regulatory specialists early in the development process helps identify potential adherence issues before they arise, effectively mitigating risks. Notably, organizations that utilize cross-functional teams experience technology adoption rates that are 34% higher than those employing siloed approaches, underscoring the significance of collaboration. Moreover, the involvement of C-suite executives in these initiatives can boost implementation success rates by nearly 60%, highlighting the critical role of leadership engagement.
Case studies from successful medical device companies illustrate that cross-functional collaboration, supported by medical equipment software, not only improves compliance but also accelerates the development timeline and enhances overall product quality. For example, a hospital system achieved a 40% reduction in implementation delays by establishing dedicated communication channels for cross-functional teams, demonstrating the tangible benefits of this approach.
However, organizations must remain vigilant against common pitfalls, such as unclear roles or lack of commitment, which can undermine the effectiveness of cross-functional collaboration.
Conclusion
Understanding and adhering to best practices for medical equipment software compliance is essential for ensuring safety, efficacy, and market readiness. This article underscores the importance of grasping regulatory requirements, implementing rigorous testing protocols, establishing continuous monitoring mechanisms, and fostering cross-functional collaboration. Together, these elements create a robust compliance framework that not only meets legal standards but also enhances the quality of healthcare software solutions.
Key insights discussed include the necessity of a thorough understanding of FDA guidelines, ISO standards, and HIPAA requirements, which form the foundation of compliance. The article emphasizes the significance of rigorous testing throughout the software development lifecycle, the value of real-time feedback systems to improve user satisfaction, and the benefits of cross-departmental collaboration to streamline adherence processes. Each of these practices contributes to reducing risks and improving the overall effectiveness of medical software.
Ultimately, the commitment to best practices in medical equipment software compliance transcends mere regulatory obligation; it represents a strategic advantage that can lead to better patient outcomes and increased operational efficiency. Organizations are encouraged to prioritize these compliance strategies, engage their teams in continuous learning, and embrace collaboration to adapt to the evolving regulatory landscape. By doing so, they not only protect their interests but also enhance the quality of care delivered to patients.
Frequently Asked Questions
What are the key regulatory requirements for medical software?
Key regulatory requirements for medical software include compliance with FDA guidelines, ISO standards, and HIPAA requirements. Each regulation outlines specific criteria to ensure safety and efficacy.
What does the FDA require for programs classified as medical devices?
The FDA requires that programs classified as medical devices undergo thorough premarket evaluation to ensure their safety and effectiveness before they can be approved for market use.
How do ISO standards contribute to medical software compliance?
ISO standards provide frameworks for quality management systems, which include risk management and clinical investigation protocols, helping ensure that medical software meets regulatory requirements.
What changes are expected in ISO 14155 in 2026?
Updates to ISO 14155 in 2026 will enhance clarity for producers by aligning more closely with ICH guidelines, which will facilitate adherence to legal limits in medical software development.
Why are training sessions and workshops important for development teams?
Frequent training sessions and workshops are crucial for keeping development teams informed about evolving regulations, ensuring that their software complies with regulatory standards and is market-ready.
How do companies like Veranex navigate FDA guidelines?
Companies like Veranex effectively navigate FDA guidelines by integrating comprehensive trial data into their compliance strategies, which supports both regulatory approval and market adoption.
What is the impact of adhering to ISO standards on regulatory scrutiny?
Adherence to ISO standards can significantly reduce the risk of regulatory scrutiny, as it ensures that the software meets established quality and safety criteria.
What does the difference in rates of Adverse Device Effects (ADEs) indicate?
The difference in rates of Adverse Device Effects (ADEs) during clinical investigations compared to post-market use underscores the necessity of stringent adherence to regulatory practices to ensure ongoing safety and efficacy.
List of Sources
- Understand Regulatory Requirements for Medical Software
- The ISO 14155:2026 Update Is Here: Top 3 Medical Device Insights (https://veranex.com/blog/iso-14155-2026-is-here-medical-device-insights)
- For 2026, FDA signals shifts in digital health framework | Nixon Peabody LLP (https://nixonpeabody.com/insights/alerts/2026/01/27/for-2026-fda-signals-shifts-in-digital-health-framework)
- FDA’s 2026 Guidance Expands Pathway for Low-Risk Digital Health Products—But Caution Remains Essential | Berkley Lifesciences (https://berkleyls.com/blog/fdas-2026-guidance-expands-pathway-low-risk-digital-health-products-caution-remains-essential)
- FDA Digital Health Guidance: 2026 Requirements Overview | IntuitionLabs (https://intuitionlabs.ai/articles/fda-digital-health-technology-guidance-requirements)
- FDA’s 2026 Expectations: Clinical Decision Support Compliance (https://about.citiprogram.org/blog/clinical-decision-support-compliance-fdas-2026-expectations)
- Implement Rigorous Testing Protocols
- 50 Inspirational Quotes About Software Testing – QA Madness (https://qamadness.com/inspirational-quotes-about-software-testing)
- 51 HIPAA Statistics Every Healthcare Entity Needs to Know in 2026 | UpGuard (https://upguard.com/blog/hipaa-statistics)
- 130+ Compliance Statistics & Trends to Know for 2026 (https://secureframe.com/blog/compliance-statistics)
- Healthcare Application Testing: All You Need to Know in 2026 (https://kms-technology.com/blog/healthcare-application-testing-all-you-need-to-know-in-2026)
- Latest Software Testing Statistics (2026 Edition) (https://testgrid.io/blog/software-testing-statistics)
- Establish Continuous Monitoring and Feedback Mechanisms
- The Power of Real-Time Patient Feedback: Enhancing the Patient Journey in Healthcare | Swell (https://swellcx.com/resources/real-time-patient-feedback-healthcare)
- 130+ Compliance Statistics & Trends to Know for 2026 (https://secureframe.com/blog/compliance-statistics)
- Real-Time Feedback: How Digital Tools Are Closing the Patient Engagement Loop (https://linkedin.com/pulse/real-time-feedback-how-digital-tools-closing-patient-loop-altayyeb-wk3se)
- Use of Daily Web-Based, Real-Time Feedback to Improve Patient and Family Experience – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11005486)
- Smarter Care, Faster: Why Hospitals Are Turning to Real-Time Feedback and Predictive AI to Transform Patient Experience • FeedbackNow (https://feedbacknow.com/blog/smarter-care-faster-why-hospitals-are-turning-to-real-time-feedback-and-predictive-ai-to-transform-patient-experience)
- Foster Cross-Functional Collaboration for Compliance
- Beyond the Operating Room: How MedTech Firms Are Building Multidisciplinary Teams for 2026 (https://mrinetwork.com/hiring-talent-strategy/medtech-multidisciplinary-teams-2026)
- Want to Drive Innovation? You Need Cross-Functional Collaboration (https://americanhealthcareleader.com/2025/drive-innovation-with-cross-functional-collaboration)
- 3 quotes about regulatory affairs that will make you smile | Regulatory Affairs Professionals Society (RAPS) (https://linkedin.com/posts/regulatory-affairs-professionals-society-raps-_3-quotes-about-regulatory-affairs-that-will-activity-7370109430329409536-T7xL)
