Introduction
In an era where healthcare technology is rapidly advancing, effective management of medical device inventory is paramount for operational success and regulatory compliance. We will explore ten essential features that enhance medical inventory management while improving patient care and operational efficiency. Organizations are struggling to keep pace with regulatory demands and the necessity for timely data access. Which features are essential for overcoming these challenges and optimizing inventory management in healthcare?
Neutech’s Tailored Engineering Talent for Medical Device Inventory Management
In an industry marked by stringent regulations and rapid technological advancements, Neutech, Inc. specializes in delivering skilled engineering talent tailored for the medical equipment sector. Through its innovative Residency Program, the company transforms junior engineers into senior-level experts, equipping them with deep technical skills and advanced domain knowledge essential for success in medical equipment oversight.
Finding qualified engineering talent in the medical equipment sector is increasingly challenging due to stringent regulatory requirements. This program addresses this challenge, particularly in an industry where precision and regulatory compliance are critical. Neutech places a strong emphasis on qualities like work ethic, communication, and leadership, ensuring its engineers possess both technical skills and the reliability needed in high-stakes environments.
Neutech’s engineers are proficient in merging intricate systems, ensuring that the medical device inventory management software complies with the rigorous standards necessary in medical settings. Recent trends indicate a growing demand for such expertise, as the FDA approved 33% more novel technologies in 2023 compared to the previous year. This growing demand underscores the necessity for robust engineering talent in the field.
Furthermore, strong interoperability and proactive cybersecurity measures are vital for maintaining patient safety and trust, making Neutech’s approach not only timely but essential for the evolving landscape of medical technology. With a flexible month-to-month contract model, Neutech enables clients to adjust their engineering resources according to project needs, offering a seamless integration of talent that adapts to the dynamic requirements of the medical equipment industry.
Neutech’s tailored engineering talent provision process begins with a thorough assessment of client needs, followed by the supply of specialized developers and designers who are best suited to meet those requirements. This strategic approach not only addresses immediate project needs but also positions clients for long-term success in a competitive market.
Real-Time Tracking Capabilities for Enhanced Efficiency
Real-time tracking features are crucial for effective medical equipment management, allowing healthcare providers to monitor the status and location of tools continuously. Without real-time tracking, healthcare providers risk running out of essential equipment, which can hinder patient care.
Utilizing technologies like RFID and barcode scanning enables medical facilities to streamline stock management, resulting in enhanced operational efficiency and significant cost savings. Ultimately, this capability not only enhances patient care but also optimizes resource allocation within healthcare facilities.
![]()
Comprehensive Compliance Support for Regulatory Adherence
Medical device inventory management software plays a crucial role in ensuring compliance with stringent FDA and ISO standards. Key features encompass:
- Tracking Unique Device Identifiers (UDIs)
- Automated reporting for audits
- Thorough documentation oversight
These functionalities assist healthcare organizations in navigating complex regulatory landscapes and reducing the risk of costly penalties for non-compliance. Moreover, adhering to these standards greatly improves patient safety, underscoring that compliance assistance is vital for effective medical device inventory management software solutions. Organizations that have prioritized compliance often see significant improvements in operational quality. Insights from regulatory experts emphasize the importance of staying updated with the latest FDA regulations and ISO 13485 requirements, particularly in light of the new compliance program titled ‘Inspection of Medical Device Manufacturers Compliance Program 7382.850,’ which reflects the FDA’s commitment to a more risk-based inspection approach.
Advanced Analytics and Reporting Features for Data-Driven Decisions
Effective stock management in medical devices hinges on advanced analytics and reporting capabilities offered by medical device inventory management software, which empower organizations to make informed decisions. These tools help analyze usage patterns and forecast demand, enabling providers to identify trends that shape procurement strategies within their medical device inventory management software.
Medical device inventory management software enables medical organizations to improve stock levels, minimize waste, and prepare effectively for changes in demand. This proactive approach enhances operational efficiency and significantly improves patient care outcomes with the implementation of medical device inventory management software.
AI-driven models are increasingly used to enhance the accuracy of demand forecasting, with studies indicating that these applications can reduce forecasting errors for medical supplies by up to 30%. Moreover, cooperation among medical service providers and suppliers is essential for efficient stock control through medical device inventory management software, ensuring that purchasing strategies align with current demand.
Organizations that implement medical device inventory management software report a 25% decrease in stockouts. This illustrates the tangible benefits of data-driven decision-making in managing resources through medical device inventory management software. Ultimately, leveraging data-driven insights not only streamlines operations but also enhances the quality of patient care delivered.
User-Friendly Interfaces for Improved Usability
A user-friendly interface is critical for medical device inventory management software, as it directly impacts the efficiency of healthcare personnel. Intuitive design reduces the learning curve and minimizes mistakes. This allows users to concentrate on efficient stock management rather than grappling with complex software.
For instance, Tobey Hospital saw an 83% reduction in manual stock management effort after introducing the Wireless PAR Weighing Bin, highlighting how user-friendly systems can significantly boost operational efficiency. Customizable dashboards, straightforward navigation, and clear labeling enhance usability. These features facilitate efficient task performance.
Industry leaders emphasize that investing in intelligent infrastructure that adapts to workflows can lead to substantial operational improvements, ultimately benefiting patient care. As Or Lomnitz from IDENTI states, “It’s not just a smarter way to manage stock – it’s a strategic advantage for hospitals that want to run leaner, respond faster, and reduce waste without compromising care.”
As the medical environment evolves in 2026, the importance of intuitive design in supply management systems becomes clearer. This ensures staff can manage resources efficiently and confidently. Moreover, continuous assistance after the launch from HIMS tool providers is crucial for user training and adjustment to new software, tackling potential challenges staff may encounter during onboarding due to limited computer skills.
Ultimately, the right medical device inventory management software can transform stock management into a streamlined process that improves patient outcomes and operational efficiency.
Mobile Access for On-the-Go Inventory Management
Mobile access fundamentally transforms how medical equipment stock is managed, allowing healthcare professionals to oversee supplies with unprecedented ease. With mobile features, staff can update stock levels, track equipment locations, and receive notifications directly on their smartphones or tablets. This flexibility enhances responsiveness and ensures real-time stock oversight, no matter where the user is located.
As a result, operational efficiency improves significantly, with research indicating that medical organizations using mobile tools for stock management can increase work efficiency by up to 30%. Moreover, nearly 90% of medical practitioners utilize mobile devices to interact with patients, according to a 2015 HIMSS Mobile Technology Survey, underscoring the growing reliance on mobile technology in medical environments.
The incorporation of mobile stock solutions and medical device inventory management software is transforming the management of medical equipment, resulting in improved patient care and more efficient operations. As indicated by medical professionals, these solutions not only enable real-time updates but also contribute to reducing readmission rates, as demonstrated in a pilot study at Hahnemann Hospital, which resulted in a 10% decline in 30-day readmissions.
Integration Capabilities for Seamless Operations
Integration features are critical for optimizing medical device stock control software, linking it effectively with Electronic Health Records (EHR) and supply chain systems. Effective integration is crucial as it facilitates seamless data flow, thereby reducing errors and enhancing operational efficiency. By enabling real-time data sharing, healthcare organizations can significantly improve their stock oversight processes, ensuring that all stakeholders have access to precise and timely information. Recent analyses highlight a significant trend towards EHR integration, which is essential for improving resource management and aligning supply chain operations with clinical needs. The partnership between Movemedical and Advantus Health Partners exemplifies how integrated systems can enhance stock control, reduce costs, and improve patient care outcomes. As we approach 2026, the combination of EHR with stock management systems will be vital for medical organizations seeking to enhance efficiency and responsiveness in their supply chains, particularly as the Data Integration Market is projected to reach USD 4.5 billion by 2026.
Lot and Serial Number Tracking for Enhanced Traceability
Effective tracking of lot and serial numbers is crucial for ensuring compliance and enhancing patient safety in medical equipment management. Without effective tracking, medical organizations risk non-compliance and jeopardize patient safety. This capability is essential for managing recalls effectively. By maintaining detailed records of lot and serial numbers, medical organizations can quickly identify affected products. This proactive approach not only safeguards patients but also mitigates the repercussions of product recalls. Ultimately, robust tracking systems are vital for maintaining trust and accountability in healthcare delivery.
![]()
Customizable Alerts and Notifications for Proactive Management
In the realm of medical device inventory management software, the lack of timely alerts can result in critical shortages that jeopardize patient care. Customizable alerts and notifications are essential for proactive oversight within medical device inventory management software systems. These features empower users to use medical device inventory management software to establish thresholds for stock levels, expiration dates, and other critical metrics, ensuring timely notifications before potential issues arise. This proactive approach is vital.
Research indicates that utilizing medical device inventory management software for effective inventory oversight can reduce stockout occurrences by as much as 30%. This improvement significantly enhances the availability of essential medical equipment when it is most needed. Timely alerts from medical device inventory management software enable healthcare providers to swiftly replenish stock and tackle emerging issues, thus preventing stockouts. This method is crucial, as it allows healthcare providers to take necessary actions before problems escalate.
Additionally, the FDA’s Early Alert Communications Program, which seeks to enhance the promptness of communications concerning high-risk medical product removals or corrections, highlights the significance of proactive oversight in ensuring the availability of medical device inventory management software. Furthermore, the FDA’s revised recommendations on safety procedures for charging medical equipment emphasize the necessity for operational efficiency and risk oversight in healthcare environments.
Expert insights further emphasize that implementing medical device inventory management software not only enhances operational efficiency but also contributes to improved patient outcomes by ensuring that essential equipment is consistently available. Ultimately, the integration of customizable alerts is essential for safeguarding patient health and ensuring operational excellence in healthcare settings.
Robust Security Features for Data Protection
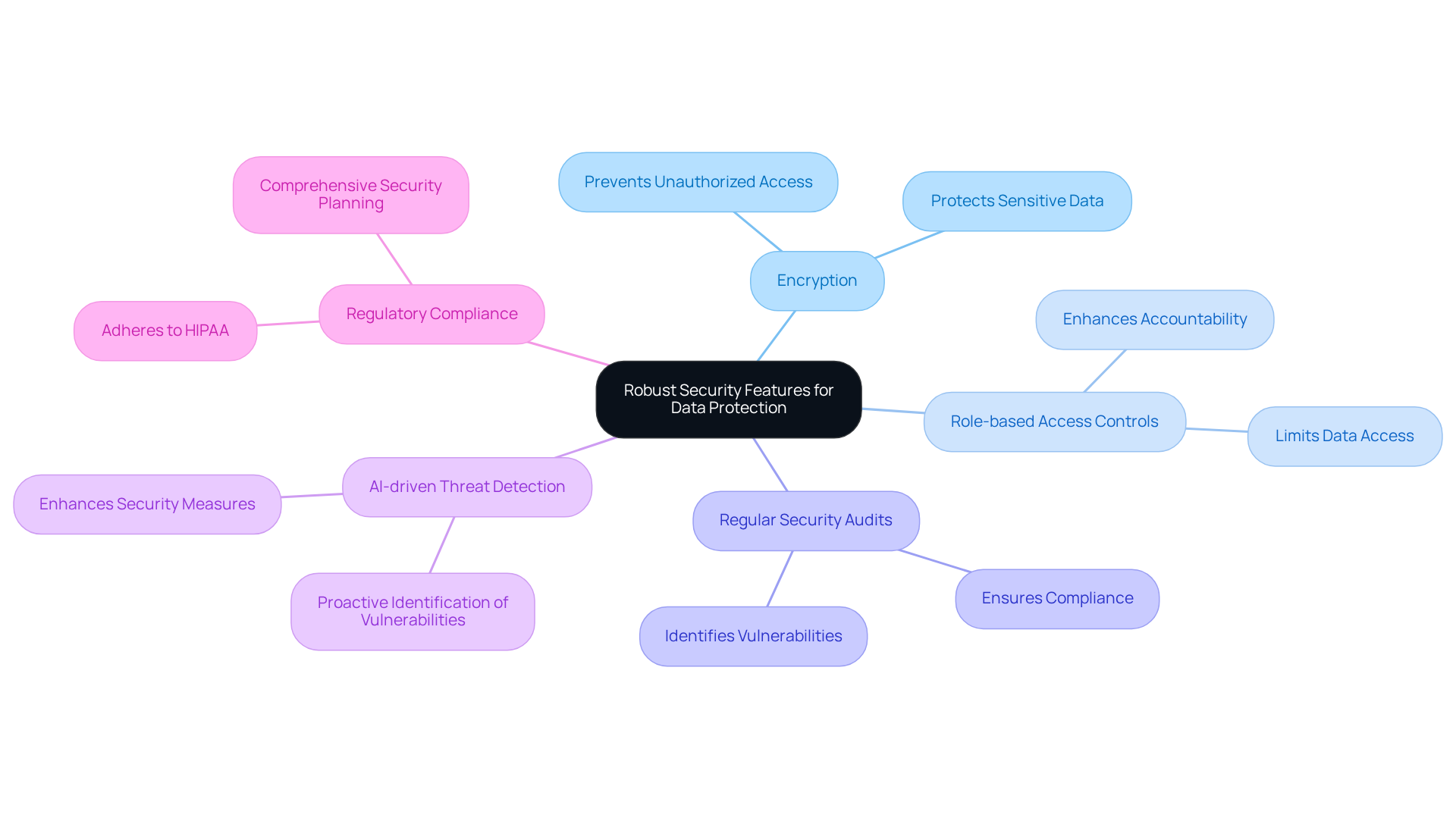
In an era where cyber threats are rampant, the security of medical device inventory management software is non-negotiable, as it safeguards sensitive patient and inventory data. The prevalence of cyberattacks poses a serious threat to the integrity of medical data management, making the prioritization of security protocols critical. Key measures include:
- Encryption
- Role-based access controls
- Regular security audits
All designed to prevent unauthorized access and data breaches. With 92% of medical organizations experiencing at least one cyberattack in 2024, these security protocols must be prioritized. Furthermore, regulations require comprehensive security planning throughout the device lifecycle, ensuring that medical organizations not only protect patient information but also adhere to stringent regulations like HIPAA. Incorporating AI-driven threat detection enhances these security measures, allowing for proactive identification of vulnerabilities. By committing to data protection, organizations not only foster trust among patients and stakeholders but also reinforce the integrity of healthcare operations in a digital age. As the landscape of cybersecurity continues to shift, organizations must adapt their strategies to ensure patient safety and compliance with evolving regulations.
Conclusion
In the competitive landscape of healthcare, effective medical device inventory management is crucial for operational success. This article has explored ten critical functionalities that streamline inventory management and position healthcare organizations to meet the evolving demands of the medical landscape. By leveraging these features, organizations can transform their inventory processes into a strategic advantage, which is vital for staying competitive in the evolving healthcare landscape.
The discussion highlighted the critical role of real-time tracking in enhancing operational efficiency, comprehensive compliance support for regulatory adherence, and advanced analytics that drive informed decision-making. Furthermore, user-friendly interfaces, mobile access, and robust security features are essential for ensuring that healthcare personnel can manage resources effectively while maintaining patient safety. These elements are essential for optimizing medical device inventory management software, reinforcing the need for healthcare providers to adopt these technologies.
As the medical industry continues to advance, embracing innovative inventory management solutions is not just beneficial but essential. Organizations must prioritize the implementation of these features to enhance operational efficiency, safeguard patient data, and ultimately deliver superior care. The commitment to adopting advanced inventory management solutions will define the future of patient care and operational excellence.
Frequently Asked Questions
What does Neutech specialize in?
Neutech specializes in delivering skilled engineering talent tailored for the medical equipment sector, focusing on transforming junior engineers into senior-level experts through its innovative Residency Program.
What is the purpose of Neutech’s Residency Program?
The Residency Program aims to equip junior engineers with deep technical skills and advanced domain knowledge essential for success in medical equipment oversight.
Why is finding qualified engineering talent in the medical equipment sector challenging?
It is challenging due to stringent regulatory requirements and the need for precision and regulatory compliance in the industry.
What qualities does Neutech emphasize in its engineers?
Neutech emphasizes qualities such as work ethic, communication, and leadership, ensuring that engineers possess both technical skills and reliability in high-stakes environments.
How does Neutech ensure compliance in medical device inventory management?
Neutech’s engineers are proficient in merging intricate systems to ensure that medical device inventory management software complies with rigorous standards necessary in medical settings.
What recent trend highlights the growing demand for engineering talent in the medical equipment field?
The FDA approved 33% more novel technologies in 2023 compared to the previous year, indicating a growing demand for robust engineering talent in the field.
What are the key features of Neutech’s engineering talent provision process?
The process begins with a thorough assessment of client needs, followed by supplying specialized developers and designers best suited to meet those requirements, addressing immediate project needs and positioning clients for long-term success.
What role do real-time tracking capabilities play in medical equipment management?
Real-time tracking features allow healthcare providers to continuously monitor the status and location of tools, preventing shortages that could hinder patient care.
How do technologies like RFID and barcode scanning benefit medical facilities?
They streamline stock management, enhance operational efficiency, and result in significant cost savings while optimizing resource allocation within healthcare facilities.
What are the key features of medical device inventory management software for regulatory compliance?
Key features include tracking Unique Device Identifiers (UDIs), automated reporting for audits, and thorough documentation oversight, which assist in navigating regulatory landscapes and improving patient safety.
Why is compliance assistance vital for medical device inventory management?
Compliance assistance helps organizations adhere to FDA and ISO standards, reducing the risk of costly penalties for non-compliance and improving operational quality.
What recent compliance program reflects the FDA’s commitment to risk-based inspections?
The new compliance program titled ‘Inspection of Medical Device Manufacturers Compliance Program 7382.850’ reflects the FDA’s commitment to a more risk-based inspection approach.
List of Sources
- Neutech’s Tailored Engineering Talent for Medical Device Inventory Management
- News Archives (https://medicaldevice-network.com/news)
- Medical device maker Zimmer Biomet adopts cautious outlook amid sales force overhaul (https://reuters.com/business/healthcare-pharmaceuticals/medical-device-maker-zimmer-biomet-raises-annual-profit-forecast-announces-cfo-2026-04-28)
- Can Medical Device Makers Keep Up with Their Talent Needs? (https://mpo-mag.com/can-medical-device-makers-keep-up-with-their-talent-needs)
- Five Major Trends Shaping Medical Device Software (https://aami.org/news/five-major-trends-shaping-medical-device-software)
- Real-Time Tracking Capabilities for Enhanced Efficiency
- Medline taps into automated vending solution to help EMS providers track inventory in real-time – Medline Newsroom (https://newsroom.medline.com/releases/medline-taps-into-automated-vending-solution-to-help-ems-providers-track-inventory-in-real-time)
- RFID Medication Tracking for Pharmaceutical Inventory Management – Bluesight (https://bluesight.com/news/rfid-is-transforming-inventory-management-insights-and-predictions-for-pharma)
- Tackling Hospital Waste: How RFID Improves Medical Inventory Management (https://atlasrfidstore.com/rfid-insider/tackling-hospital-waste-how-rfid-improves-medical-inventory-management?srsltid=AfmBOoonaPfrPrH8wRuw4x5UAzRDI1hsAYaRzLUwR–_saIftJowKNjI)
- Medical device inventory management: Staying compliant in a regulated industry (https://netstock.com/blog/medical-device-inventory-management)
- Comprehensive Compliance Support for Regulatory Adherence
- What the FDA’s New Quality Requirements Mean for U.S. Medical Device Companies (https://amtivo.com/us/resources/insights/fdas-new-quality-requirements-mean-for-us-medical-device-companies)
- Quality Management System Regulation (https://fda.gov/medical-devices/postmarket-requirements-devices/quality-management-system-regulation-qmsr)
- gardner.law (https://gardner.law/news/fda-revised-qmsr-modernizes-device-quality-regulation)
- Healthcare Compliance Software Market Size to Reach USD 9.9 Bn by 2034 (https://dimensionmarketresearch.com/report/healthcare-compliance-software-market)
- FDA QMSR and ISO 13485: Key Changes for Manufacturers | PSC Software (https://pscsoftware.com/resource-center/article/fdas-new-qmsr-rule-what-medical-device-manufacturers-need-to-know)
- Advanced Analytics and Reporting Features for Data-Driven Decisions
- Columbus Global | 5 Use Cases for Analytics in Medical Device Manufacturing (https://columbusglobal.com/insights/articles/5-use-cases-for-analytics-in-medical-device-manufacturing)
- 5 insights driving smarter healthcare procurement in 2026 (https://advisory.com/sponsored/healthcare-procurement-insights)
- Big Data Analytics in Healthcare Market 2026 improving patient outcomes through insights (https://norfolkdailynews.com/online_features/press_releases/big-data-analytics-in-healthcare-market-2026-improving-patient-outcomes-through-insights/article_cd646ba9-1249-5c21-a9a0-537e2dcc4c3a.html)
- Triple A: How Analytics, AI, and Algorithms Are Improving Inventory Management in Healthcare (https://mdpi.com/2305-6290/10/5/103)
- User-Friendly Interfaces for Improved Usability
- Healthcare Inventory Management in 2025: 3 Must-Have AI Products | IDENTI Medical (https://identimedical.com/ai-transforming-healthcare-inventory-management)
- Hospital Inventory Management Solutions: A Complete Overview (https://itransition.com/healthcare/hospital-inventory-management)
- The Future of Medical Inventory Management Software for the Best Medical Device Teams | ConnectSx (https://connectsx.com/blog/future-of-medical-inventory-management-software)
- EHR Interface Design Principles, UX, And Usability Challenges (https://fuselabcreative.com/ehr-interface-design-principles-ux-and-usability-challenges)
- Mobile Access for On-the-Go Inventory Management
- BD Launches AI-Enabled Medication Dispensing System to the European Market (https://prnewswire.com/news-releases/bd-launches-ai-enabled-medication-dispensing-system-to-the-european-market-302730648.html)
- 30 Amazing Mobile Health Technology Statistics (https://referralmd.com/mobile-healthcare-technology-statistics)
- Advantus Health Partners, Movemedical Work to Improve Medtech Inventory (https://mpo-mag.com/breaking-news/advantus-health-partners-movemedical-work-to-improve-medtech-inventory)
- Medical Mobile Device Success Case: How to Improve Hospital Efficiency and Safety (https://seuic.us/news-center/product-dynamics/medical-mobile-device-success-case-how-to-improve-hospital-efficiency-and-safety.html)
- Integration Capabilities for Seamless Operations
- Digica and ORtelligence Collaborate with Leading Medical Device Manufacturers to Launch ExecuTorch Application for Inventory Optimization and Revenue Enhancement – Digica | AI powered software (https://digica.com/news/digica-and-ortelligence-collaborate-with-leading-medical-device-manufacturers-to-launch-executorch-application-for-inventory-optimization-and-revenue-enhancement.html)
- Advantus Health Partners, Movemedical Work to Improve Medtech Inventory (https://mpo-mag.com/breaking-news/advantus-health-partners-movemedical-work-to-improve-medtech-inventory)
- Movemedical Partners with Resurgens Technology (https://resurgenstech.com/news/movemedical-partners-with-resurgens-technology-to-expand-innovation-in-the-medical-device-supply-chain)
- Healthcare Data Integration Market Size & Outlook 2035 (https://marketresearchfuture.com/reports/healthcare-data-integration-market-26516)
- Lot and Serial Number Tracking for Enhanced Traceability
- Medical device inventory management: Staying compliant in a regulated industry (https://netstock.com/blog/medical-device-inventory-management)
- How Can Lot and Serialized Tracking Benefit Medical Practices (https://elevateht.com/blog/how-can-lot-and-serialized-tracking-benefit-medical-practices)
- Medical Device Inventory Management Best Practices (https://leandna.com/resource/the-inventory-control-hurdle-whats-next-for-medical-device-manufacturers)
- Enhanced traceability with RFID supports DSCSA compliance (https://healthcareitnews.com/news/enhanced-traceability-rfid-supports-dscsa-compliance)
- Customizable Alerts and Notifications for Proactive Management
- FDA Expands Early Alert Communications for Medical Devices (https://cov.com/en/news-and-insights/insights/2025/10/fda-expands-early-alert-communications-for-medical-devices)
- FDA expands Early Alert program to all medical devices (https://beckershospitalreview.com/supply-chain/fda-expands-early-alert-program-to-all-medical-devices)
- Inventory Report: The Ultimate Guide to Low Stock Alerts: Everything You Need to Know [2023] (https://genie.io/blog-articles/the-ultimate-guide-to-low-stock-alerts-everything-you-need-to-know-2023)
- FDA Expands Early Alert Program to All Medical Devices – Law Offices of Snell & Wilmer (https://swlaw.com/publication/fda-expands-early-alert-program-to-all-medical-devices)
- Robust Security Features for Data Protection
- Five Major Trends Shaping Medical Device Software (https://aami.org/news/five-major-trends-shaping-medical-device-software)
- CISOs, are your medical devices secure? Attackers are watching closely – Help Net Security (https://helpnetsecurity.com/2025/03/13/secure-medical-devices)
- Federal Regulators Weigh Cost vs. Security in HIPAA Security Proposal – ACA International (https://acainternational.org/news/hipaa-security-rule-new-enforceable-standards-set-for-may-2026)
- The Next Wave of Medical Device Security: Proactive Lifecycle Protection (https://mpo-mag.com/exclusives/the-next-wave-of-medical-device-security-proactive-lifecycle-protection)
