Introduction
The landscape of medical device software development is undergoing rapid evolution, propelled by technological advancements and a growing reliance on digital health solutions. As healthcare professionals pursue innovative strategies to enhance patient safety and improve outcomes, it becomes essential to grasp the intricacies of this development process. However, the complexities of regulatory compliance and the necessity for rigorous testing present significant challenges.
How can developers effectively navigate these obstacles while fostering innovation in a sector that is vital to patient care?
Define Medical Device Software Development
Healthcare instrument application development encompasses the design, creation, and maintenance of programs intended for clinical use. This includes both standalone applications and those integrated within hardware healthcare instruments. The development process is organized into several key stages:
- Requirements gathering
- Design
- Coding
- Testing
- Regulatory compliance
Each stage is essential for ensuring that the system adheres to the stringent safety and effectiveness standards established by regulatory bodies such as the FDA and EMA, while also adapting to the evolving landscape of healthcare technology.
As of 2026, the FDA has authorized over 520 AI-enabled health instruments, highlighting the rapid advancement and integration of technology in healthcare. With the industry increasingly reliant on sophisticated software solutions, a comprehensive understanding of the complexities involved in healthcare product software development is vital for enhancing patient safety and improving overall health outcomes. Successful projects in this domain illustrate the potential for innovation while strictly adhering to compliance standards, ultimately fostering trust and reliability in healthcare technologies.
At Neutech, we emphasize the importance of intangibles such as work ethic, communication, and leadership when selecting our engineering talent. This focus ensures that our developers not only meet technical requirements but also contribute to successful project outcomes in the healthcare sector. Our flexible month-to-month contracts enable clients to scale their development resources as needed, ensuring optimal project management and responsiveness to changing requirements. Furthermore, RegDesk’s platform simplifies life cycle management, facilitating companies in navigating the complexities of regulatory compliance.
Explore the Evolution of Medical Device Software
The evolution of healthcare equipment programs can be traced back to the early days of health technology, characterized by the use of basic mechanical tools for diagnostics and treatment. A significant turning point occurred in the 1970s with the introduction of computers, which enabled more complex functionalities in healthcare tools. By the 1990s, the medical device software development was influenced by the emergence of applications-driven instruments, leading to the creation of software as a medical device (SaMD). This category of devices operates independently from hardware and is classified into four risk levels: Class I, IIa, IIb, or III.
In recent years, the integration of artificial intelligence (AI) and machine learning (ML) into healthcare tools has enhanced their functionalities, allowing for real-time data analysis. The FDA emphasizes the importance of validating and confirming these programs in the medical device software development to ensure precision, dependability, and functionality, which are essential for maintaining quality standards. This evolution reflects a broader trend towards digital health solutions that prioritize patient-centered care and improved clinical outcomes.
The SaMD market is projected to reach USD 25.87 billion by 2031, with a compound annual growth rate (CAGR) of 37.62% from 2026 to 2031. Additionally, the incorporation of robust cybersecurity protocols into SaMD design and maintenance is crucial to address growing concerns regarding data privacy and security in the technology landscape.
Identify Key Characteristics and Types of Medical Device Software
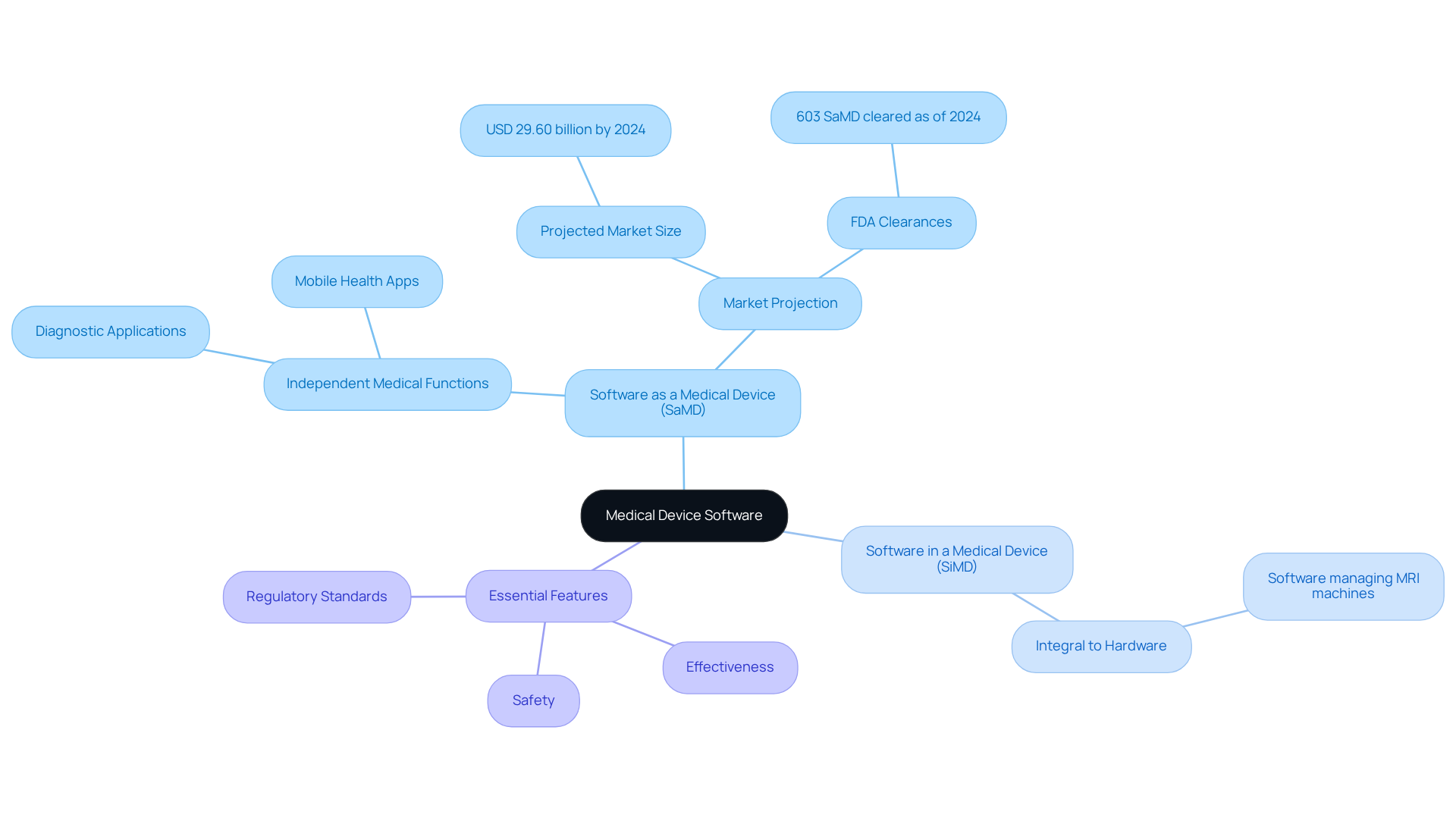
Healthcare equipment applications encompass various types, notably:
- Software as a medical device (SaMD)
- Software in a medical device (SiMD)
- Applications that support healthcare tool functions
The essential features of these applications include:
- Safety
- Effectiveness
- Adherence to regulatory standards
SaMD refers to programs that independently perform medical functions, such as diagnostic applications or mobile health apps. The global SaMD market is projected to reach USD 29.60 billion by 2024. Conversely, SiMD is a program integral to the operation of a hardware tool, exemplified by software that manages an MRI machine. As of 2024, approximately 603 SaMD have received FDA clearance, highlighting the increasing importance of this category.
Understanding these distinctions is crucial for developers and manufacturers in the medical device software development to effectively navigate the regulatory landscape and ensure compliance with safety and efficacy standards. Regulatory specialists emphasize that precise definitions of SaMD and SiMD are vital for fostering innovation while ensuring safety, as the FDA continues to refine its guidance on these classifications.
Neutech offers comprehensive engineering services tailored for the healthcare sector, focusing on application development areas such as React Development and Python Development, ensuring compliance and enhancing patient safety.
Understand the Regulatory Landscape and Compliance Requirements
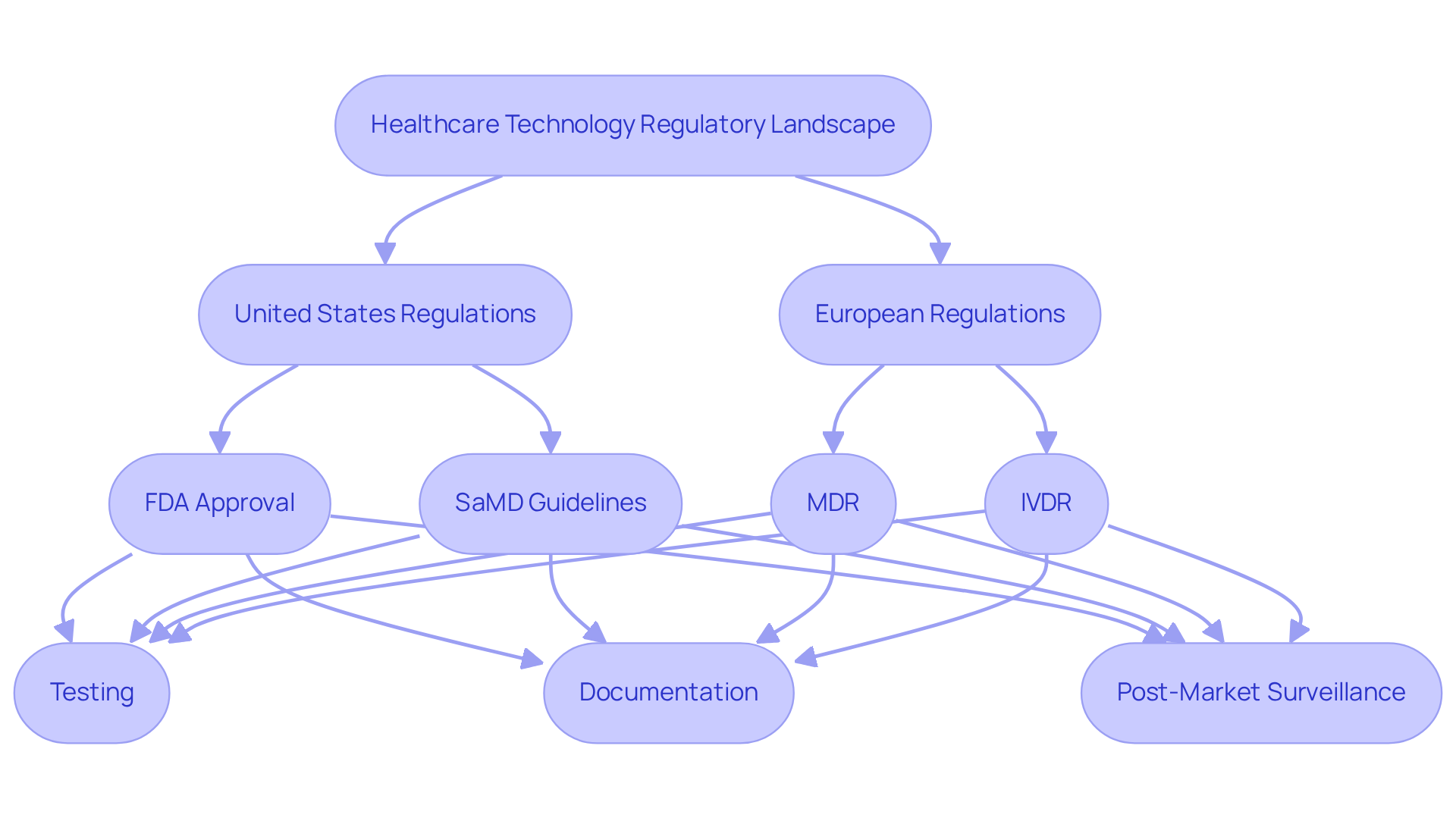
The regulatory environment for healthcare technology applications is complex and varies by region. In the United States, the FDA oversees the approval and regulation of health-related devices, including applications. In the medical device software development, developers are required to adhere to guidelines such as the FDA’s Software as a Health Device (SaMD) framework, which specifies the requirements for programs intended for health-related purposes.
In Europe, the Medical Device Regulation (MDR) and In Vitro Diagnostic Regulation (IVDR) dictate the approval process. Compliance with these regulations necessitates rigorous testing, comprehensive documentation, and post-market surveillance to ensure ongoing safety and effectiveness.
Understanding these requirements is essential for developers aiming to successfully launch their products while prioritizing user safety.
Examine the Impact of Medical Device Software on Healthcare
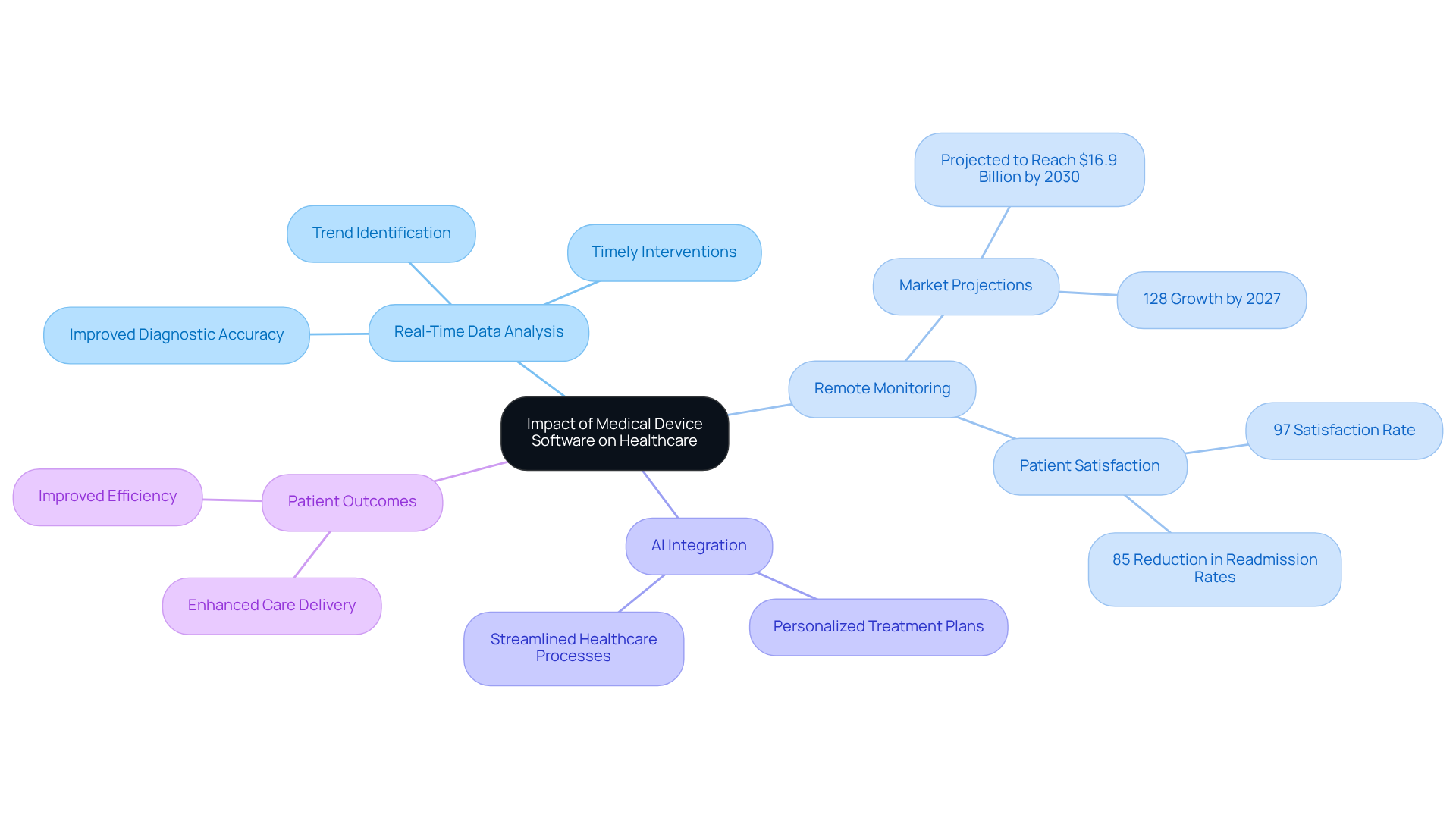
Medical instrument applications are transforming healthcare by enhancing the methods through which medical professionals diagnose, treat, and manage patients. These applications facilitate real-time data analysis and remote monitoring, which significantly improves diagnostic accuracy and enables timely interventions. For example, applications that analyze patient data can identify trends and alert healthcare providers to potential issues before they escalate, thereby preventing complications.
The integration of AI and machine learning into medical devices further personalizes treatment plans, addressing individual patient needs. This tailored approach not only enhances patient outcomes but also streamlines healthcare processes, leading to more efficient and effective care delivery. Current trends indicate a growing adoption of remote monitoring software, with market projections suggesting it will reach $16.9 billion by 2030 and expand by 128% by 2027.
Moreover, an impressive 97% of individuals participating in remote health monitoring programs reported satisfaction with their experience, highlighting the critical role of real-time data analysis in contemporary healthcare. Additionally, the implementation of remote patient monitoring has resulted in an 85% reduction in hospital readmission rates, underscoring its effectiveness in improving healthcare outcomes.
Conclusion
The landscape of medical device software development is defined by its complex processes and the vital role it plays in improving healthcare delivery. By comprehensively understanding the stages involved – from requirements gathering to regulatory compliance – stakeholders can ensure that their software solutions adhere to safety and efficacy standards while promoting innovation in patient care.
Key insights illustrate the evolution of medical device software, emphasizing the transition from basic mechanical tools to advanced AI-enabled applications. The classification of software into categories such as Software as a Medical Device (SaMD) and Software in a Medical Device (SiMD) highlights the necessity for developers to adeptly navigate intricate regulatory frameworks. As the market for SaMD continues to expand, compliance with FDA and European regulations remains crucial for ensuring user safety and upholding high-quality standards.
Reflecting on the transformative influence of medical device software, it is clear that these innovations significantly enhance diagnostic accuracy and improve patient outcomes. As the industry progresses, embracing contemporary trends and technological advancements will be essential for both developers and healthcare providers. By prioritizing patient-centered solutions and adhering to regulatory guidelines, stakeholders can contribute to a future where medical device software not only meets the demands of modern healthcare but also establishes new benchmarks for excellence in patient care.
Frequently Asked Questions
What is medical device software development?
Medical device software development refers to the design, creation, and maintenance of programs intended for clinical use, including both standalone applications and those integrated within hardware healthcare instruments.
What are the key stages of the medical device software development process?
The key stages include requirements gathering, design, coding, testing, and regulatory compliance.
Why is regulatory compliance important in medical device software development?
Regulatory compliance is essential to ensure that the system adheres to safety and effectiveness standards set by regulatory bodies like the FDA and EMA, thereby enhancing patient safety and improving health outcomes.
How has artificial intelligence impacted medical device software?
The integration of AI and machine learning into healthcare tools has enhanced their functionalities, allowing for real-time data analysis and improving clinical outcomes.
What is a Software as a Medical Device (SaMD)?
SaMD refers to software that operates independently from hardware and is classified into four risk levels: Class I, IIa, IIb, or III.
What is the projected market growth for SaMD?
The SaMD market is projected to reach USD 25.87 billion by 2031, with a compound annual growth rate (CAGR) of 37.62% from 2026 to 2031.
What role does cybersecurity play in medical device software?
Robust cybersecurity protocols are crucial in SaMD design and maintenance to address concerns regarding data privacy and security.
How does Neutech approach the selection of engineering talent for medical device software development?
Neutech emphasizes intangibles such as work ethic, communication, and leadership to ensure that developers contribute to successful project outcomes in the healthcare sector.
What benefits do Neutech’s flexible month-to-month contracts offer clients?
The flexible contracts enable clients to scale their development resources as needed, ensuring optimal project management and responsiveness to changing requirements.
How does RegDesk’s platform assist companies in medical device software development?
RegDesk’s platform simplifies life cycle management, helping companies navigate the complexities of regulatory compliance.
