Introduction
Creating software for medical devices presents a complex challenge that requires not only technical expertise but also a thorough understanding of regulatory compliance. As the medical device software landscape continues to evolve, developers must navigate intricate guidelines while prioritizing safety and usability. This article explores best practices designed to empower developers in effectively meeting regulatory standards, enhancing product quality, and ultimately improving patient outcomes.
What key strategies can transform compliance from a daunting requirement into a streamlined process that promotes innovation and user satisfaction?
Understand Regulatory Frameworks for Medical Device Software
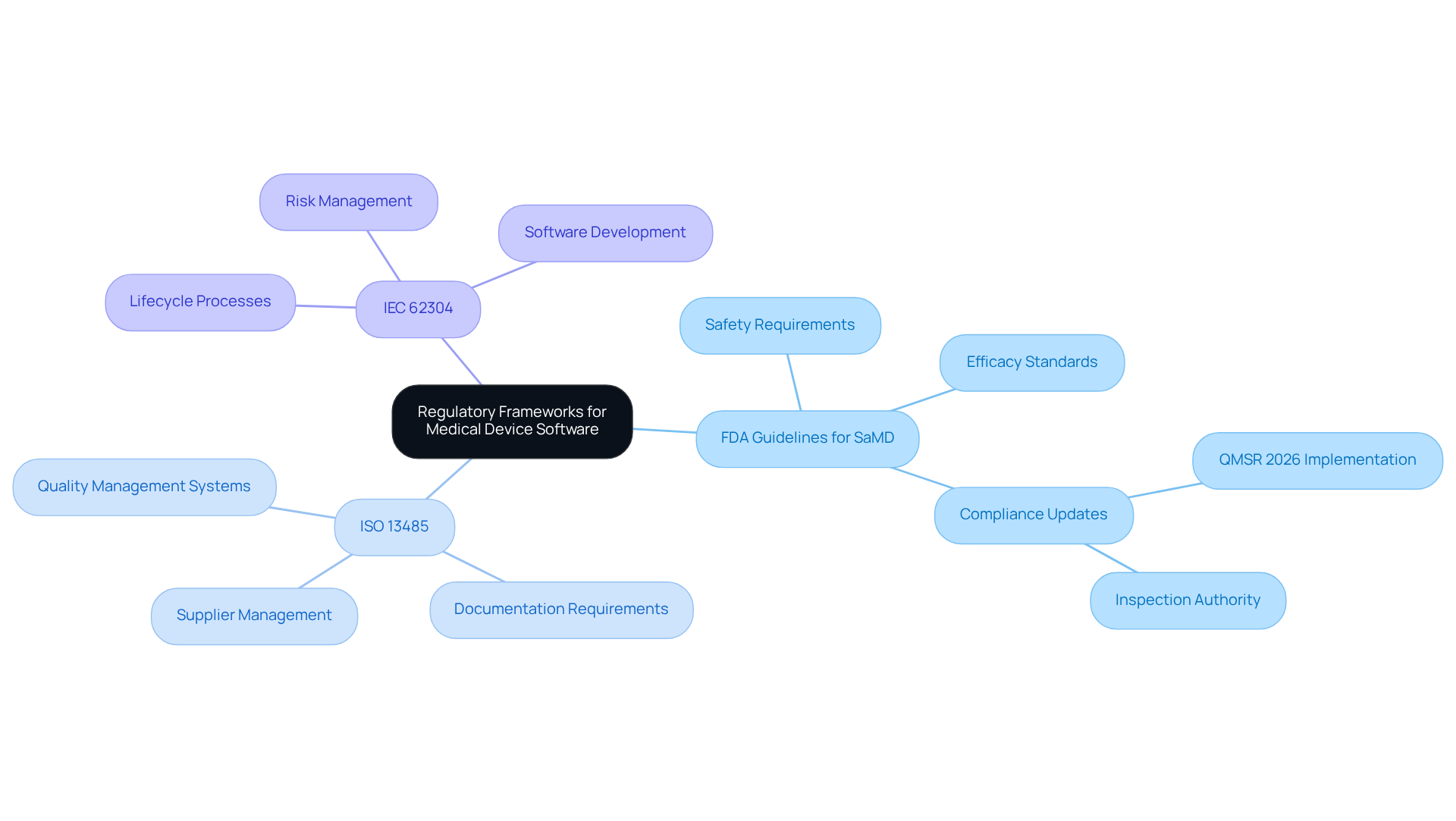
Creating applications for necessitates a thorough understanding of the that govern this sector. Central to this are the , , and . Familiarity with these standards is crucial, as it ensures that the system not only meets but also aligns with market expectations.
Developers are encouraged to stay informed about the . Engaging with can also be beneficial, as it aids in navigating the complexities inherent in these frameworks.
Implement Robust Quality Assurance Processes
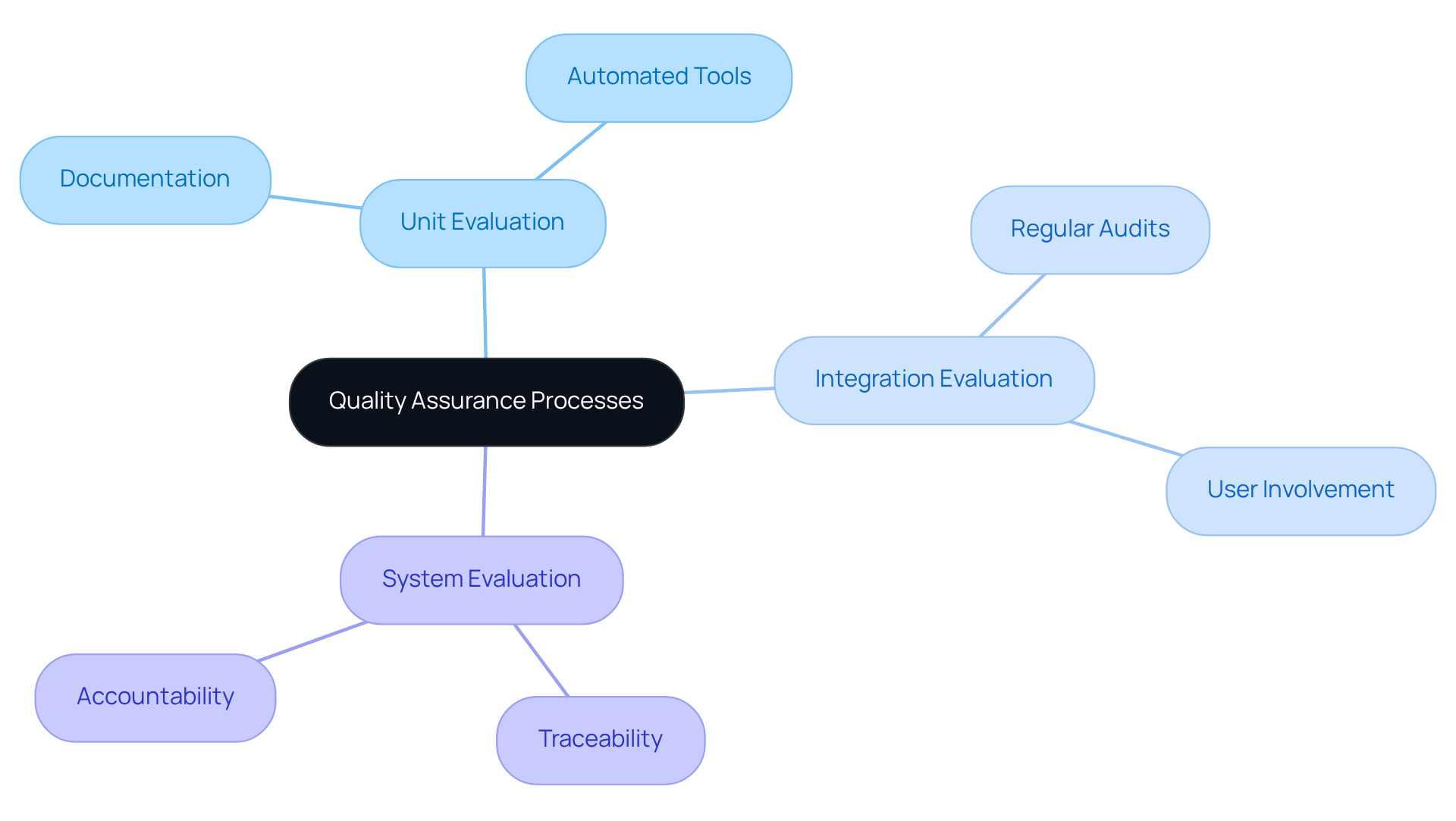
are essential in the software design for medical devices. A structured QA framework necessitates the definition of clear , which include:
- Unit evaluation
- Integration evaluation
- System evaluation
Documenting all examination procedures and results is crucial for providing . Additionally, the use of can significantly enhance both efficiency and precision. Regular audits and reviews of the QA processes are imperative to identify areas for improvement and ensure . Furthermore, involving end-users during the can yield valuable insights into user-friendliness and functionality.
Incorporate Key Design Considerations for Safety and Usability
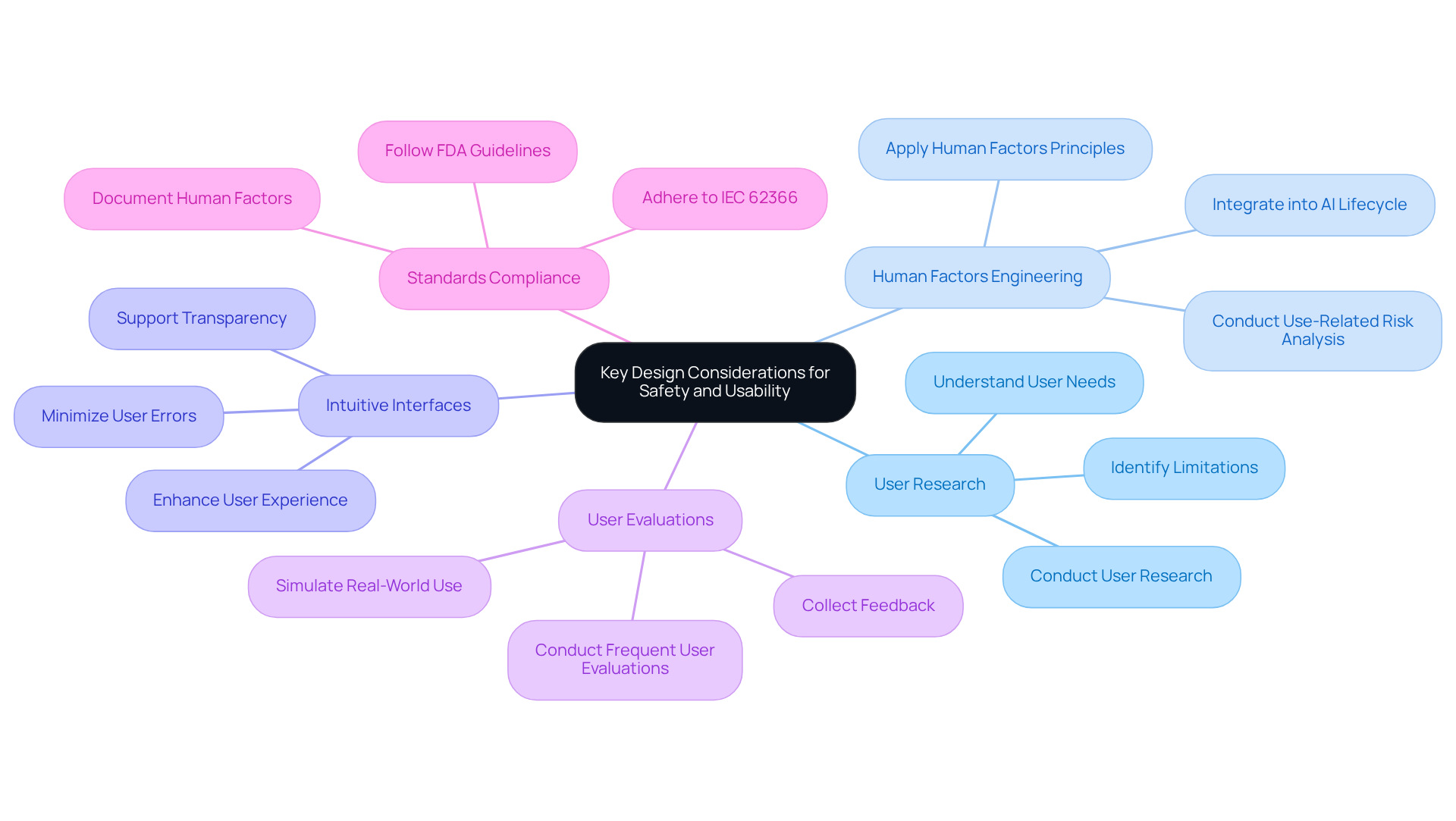
Creating requires a strong emphasis on , particularly regarding safety and usability. A foundational step involves to understand their needs and limitations, which directly informs design decisions. By applying , manufacturers can identify potential risks associated with user interactions, ensuring that the software accommodates real-world usage scenarios.
are essential; they should be designed to minimize user errors and enhance the overall user experience. Frequent user evaluations are critical, as they enable the collection of feedback that fosters incremental improvements. provides a guiding framework for integrating user-friendliness into the design process.
As emphasized by , prioritizing user-friendly designs not only enhances but also improves clinician efficiency, ultimately leading to better patient care outcomes. Furthermore, incorporating cognitive load assessments and perceptual understanding evaluations during can significantly mitigate risks associated with AI outputs, ensuring that users can effectively interpret and act on the information provided by medical devices.
Establish Continuous Monitoring and Maintenance Protocols
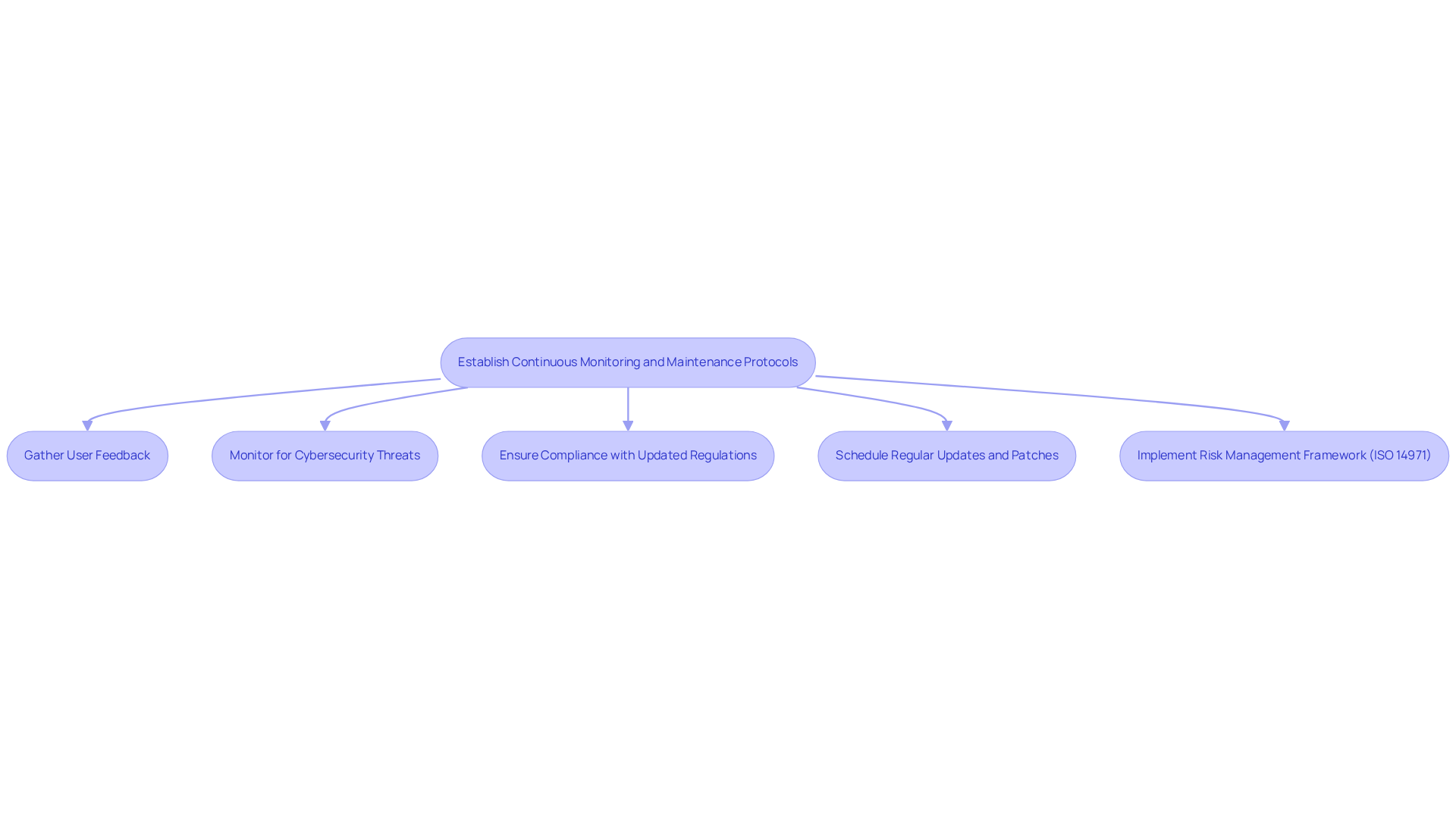
Continuous monitoring and maintenance are crucial for ensuring the ongoing safety and efficacy of . Developers must establish protocols for to effectively monitor the application’s performance and identify any issues that may arise after deployment. This process includes:
- Gathering user feedback
- Monitoring for
- Ensuring compliance with
Regular updates and patches should be scheduled to address . Furthermore, implementing a , such as ISO 14971, is essential for assessing and mitigating risks throughout the software’s lifecycle.
Conclusion
Navigating the intricate landscape of software design for medical devices necessitates a thorough understanding of regulatory frameworks, quality assurance processes, user-centered design, and ongoing maintenance protocols. By adhering to these established practices, developers can ensure that their applications not only comply with standards but also prioritize safety and usability, thereby enhancing patient care and clinician efficiency.
Key insights from the article emphasize the necessity of staying informed about regulatory guidelines, implementing structured quality assurance frameworks, and conducting comprehensive user research to guide design decisions. Continuous monitoring and maintenance further bolster the reliability of medical device software, addressing emerging issues and ensuring that applications remain compliant with evolving regulations.
The importance of these practices is paramount; they are essential to the development of safe, effective, and user-friendly medical devices. As the field of medical technology continues to advance, embracing these best practices will be crucial for developers striving to deliver high-quality solutions that meet the needs of both patients and healthcare professionals. Prioritizing compliance and user experience today will lay the groundwork for a safer, more efficient healthcare environment in the future.
Frequently Asked Questions
What is the importance of understanding regulatory frameworks for medical device software?
Understanding regulatory frameworks is crucial for ensuring that medical device applications meet safety and efficacy requirements and align with market expectations.
What are the key regulatory guidelines for Software as a Medical Device (SaMD)?
The key regulatory guidelines for SaMD include the FDA’s guidelines, ISO 13485 for quality management systems, and IEC 62304, which outlines lifecycle processes for applications.
Why is it important for developers to stay informed about regulatory updates?
Staying informed about regulatory updates helps developers ensure compliance with the latest standards and guidelines, which is essential for the safety and effectiveness of medical device software.
How can engaging with compliance experts benefit developers of medical device software?
Engaging with compliance experts can help developers navigate the complexities of regulatory frameworks, ensuring that their applications meet all necessary requirements.
List of Sources
- Understand Regulatory Frameworks for Medical Device Software
- QMSR 2026 Explained: FDA Quality Management System Regulation & ISO 13485 Alignment Guide (https://sushvin.com/qmsr-2026-fda-quality-management-system-regulation.html)
- FDA QMSR & ISO 13485: Key Changes Effective 2026 | IntuitionLabs (https://intuitionlabs.ai/articles/fda-qmsr-iso-13485-changes-2026)
- A QMSR State of Mind: FDA Adopts New Inspection Approach for Medical Devices as Quality Management System Regulation Takes Effect | Insights | Ropes & Gray LLP (https://ropesgray.com/en/insights/alerts/2026/02/a-qmsr-state-of-mind-fda-adopts-new-inspection-approach-for-medical-devices)
- FDA Implements Quality Management System Regulation for Medical Devices (https://aabb.org/news-resources/news/article/2026/02/04/fda-implements-quality-management-system-regulation-for-medical-devices)
- FDA’s 2026 Guidance Expands Pathway for Low-Risk Digital Health Products—But Caution Remains Essential | Berkley Lifesciences (https://berkleyls.com/blog/fdas-2026-guidance-expands-pathway-low-risk-digital-health-products-caution-remains-essential)
- Implement Robust Quality Assurance Processes
- QMSR 2026 Explained: FDA Quality Management System Regulation & ISO 13485 Alignment Guide (https://sushvin.com/qmsr-2026-fda-quality-management-system-regulation.html)
- FDA’s Computer Software Assurance 2026: Changes and What to Do Next | PSC Software (https://pscsoftware.com/resource-center/article/fda-computer-software-assurance-2026-changes)
- Computer Software Assurance for Production and QMS Software guidance (https://fda.gov/regulatory-information/search-fda-guidance-documents/computer-software-assurance-production-and-quality-management-system-software)
- FDA Aligns U.S. Medical Device Rules with Global Standards (https://mddionline.com/regulatory-quality/medical-device-companies-must-navigate-historic-fda-rule-change)
- Incorporate Key Design Considerations for Safety and Usability
- FDA AI Device Human Factors 2026 Guide | Mavenrs (https://mavenrs.com/blog/fda-ai-enabled-medical-device-human-factors-2026)
- Top 5 Dos and Don’ts – Human Factors Validation Testing of Medical Devices for FDA Market Access (https://emergobyul.com/news/top-5-dos-and-donts-human-factors-validation-testing-medical-devices-fda-market-access)
- Medical Device Design Tips for Usability (https://acenth.com/blog/design-tips-to-make-medical-devices-easier-to-use)
- FDA’s Human Factors Expectations for AI-enabled Software Functions (https://emergobyul.com/news/fdas-human-factors-expectations-ai-enabled-software-functions)
- Enhancing usability in medical devices: A key to MDR compliance and patient safety | Kiwa (https://kiwa.com/en/news/enhancing-usability-in-medical-devices-a-key-to-mdr-compliance-and-patient-safety)
- Establish Continuous Monitoring and Maintenance Protocols
- Tackling Cybersecurity Threats in Healthcare (https://mddionline.com/medical-iot/cybersecurity-threats-to-medical-devices-navigating-the-evolving-threat-landscape)
- Targeted Postmarket Surveillance: The Way Toward Responsible AI Innovation in Health Care (https://paragoninstitute.org/private-health/targeted-postmarket-surveillance-the-way-toward-responsible-ai-innovation-in-health-care)
- Post-Market Surveillance of Software Medical Devices: Evidence from Regulatory Data (https://profiles.wustl.edu/en/publications/post-market-surveillance-of-software-medical-devices-evidence-fro-2)
- FDA Tightens Its Medical Device Cybersecurity Guidance (https://fedtechmagazine.com/article/2026/03/fda-tightens-its-medical-device-cybersecurity-guidance-perfcon)
