Introduction
Navigating the intricate landscape of medical device software testing compliance poses significant challenges and opportunities for organizations committed to healthcare innovation. Regulatory frameworks, such as the FDA’s 21 CFR Part 820 and ISO 13485, establish the standards for safety and efficacy, making it essential for organizations to comprehend these requirements to achieve success.
As the industry continues to evolve, a critical question emerges: how can organizations not only fulfill compliance obligations but also leverage best practices to enhance software quality and ensure patient safety?
This article explores effective strategies for achieving compliance, implementing thorough testing protocols, and cultivating a culture of continuous improvement within medical device software development.
Understand Regulatory Compliance Requirements
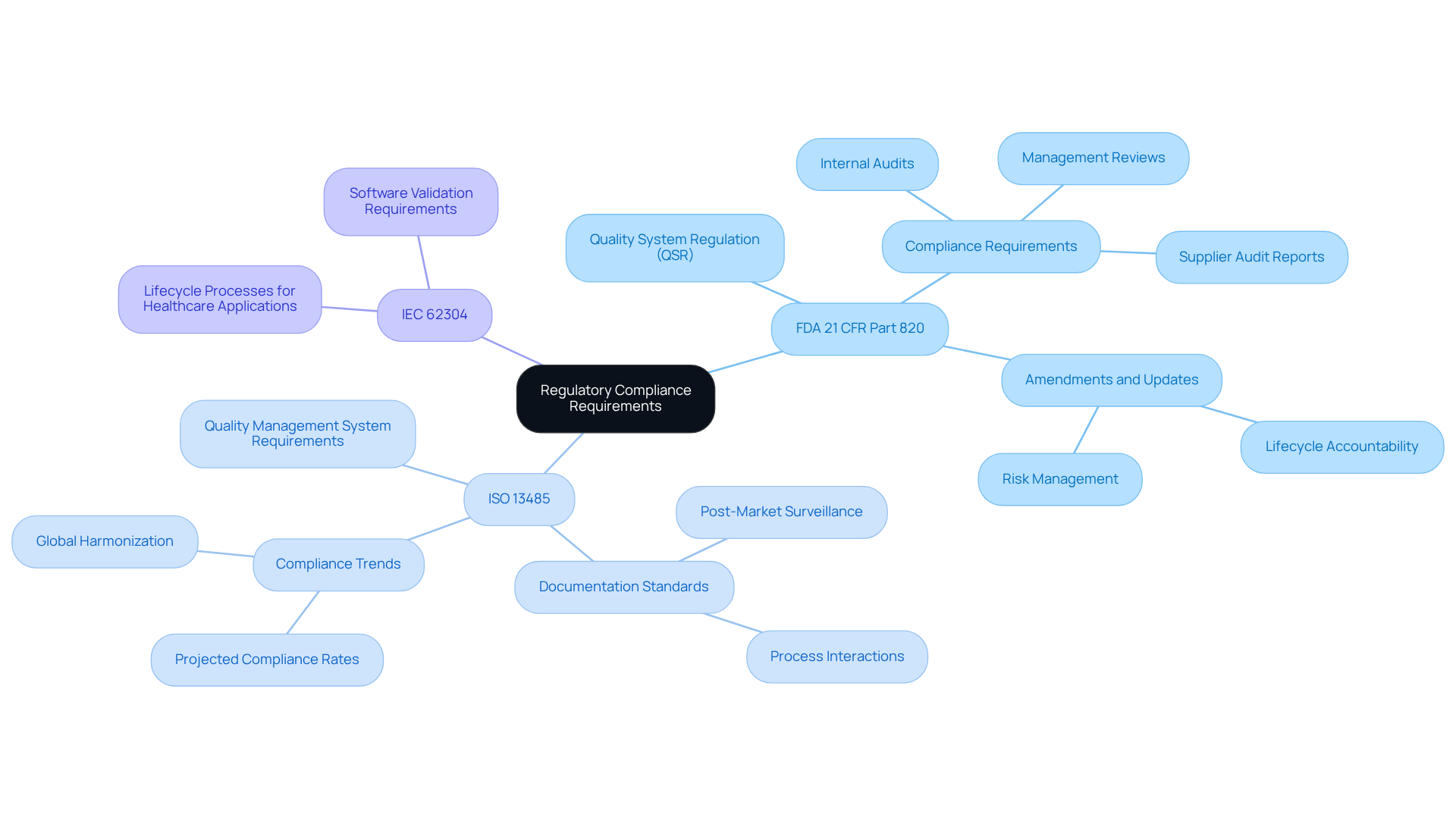
To achieve conformity in healthcare technology testing, organizations must thoroughly understand the relevant . Central to these frameworks are the FDA’s 21 CFR Part 820, which delineates the Quality System Regulation (QSR) for healthcare devices, and ISO 13485, which sets forth the requirements for a quality management system. Additionally, IEC 62304 offers essential guidelines for the lifecycle processes of healthcare applications. Familiarity with these regulations enables organizations to develop robust testing protocols and documentation practices, ensuring that all applications meet safety and efficacy standards.
For example, software applications intended for Class II health products are required to undergo stringent validation procedures to confirm their intended performance and mitigate potential risks to patients. Recent data indicates that by 2026, compliance rates with ISO 13485 among healthcare product companies are projected to exceed 80%, reflecting a significant industry shift towards standardized quality management practices.
Organizations must remain vigilant regarding regulatory updates, as changes can directly influence compliance strategies and evaluation methodologies. The FDA’s recent amendments to Part 820 underscore the significance of lifecycle accountability and risk management, aligning U.S. regulations with international standards such as ISO 13485:2016. This alignment not only streamlines compliance efforts but also bolsters the credibility of manufacturers in the global market.
Implement Comprehensive Testing Strategies
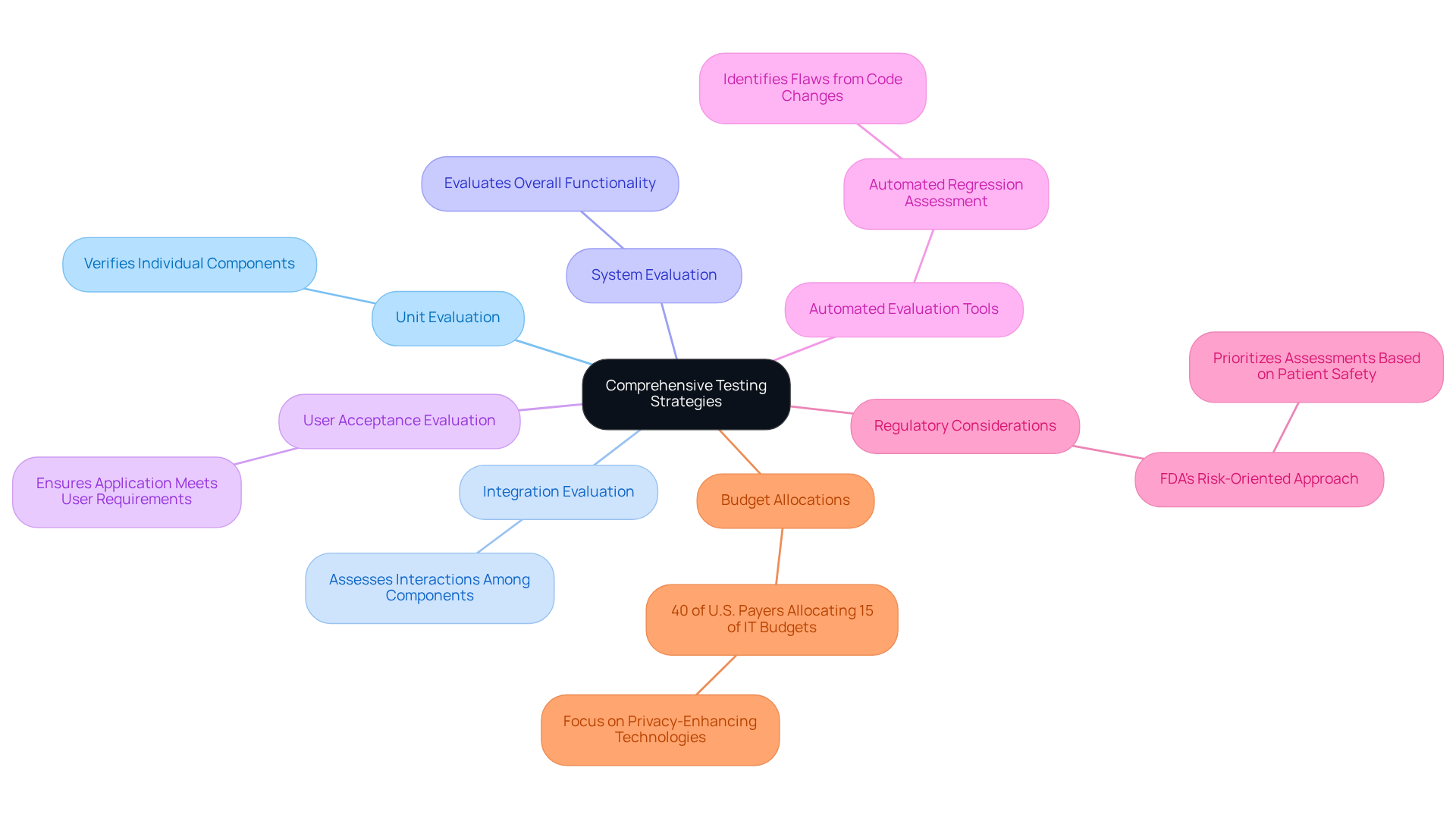
A robust evaluation strategy for medical device software testing must encompass various assessment types, including:
- Unit evaluation
- Integration evaluation
- System evaluation
- User acceptance evaluation
Each type serves a vital function: unit evaluation focuses on verifying individual components, integration evaluation assesses the interactions among these components, system evaluation evaluates the overall functionality of the complete system, and user acceptance evaluation ensures that the application meets user requirements.
To enhance efficiency and accuracy, organizations should implement , such as automated regression assessment, which swiftly identifies flaws introduced by new code changes. Furthermore, the FDA’s adoption of a risk-oriented approach to medical device software testing underscores the importance of prioritizing assessments based on the potential impact of system failures on patient safety. By ensuring that critical functionalities undergo comprehensive validation prior to launch, organizations can significantly mitigate risks associated with healthcare technology.
Additionally, with 40% of U.S. payers expected to allocate at least 15% of their IT budgets to privacy-enhancing technologies by 2026, the integration of automated evaluation tools is becoming increasingly essential within the industry.
Adopt Continuous Improvement Practices
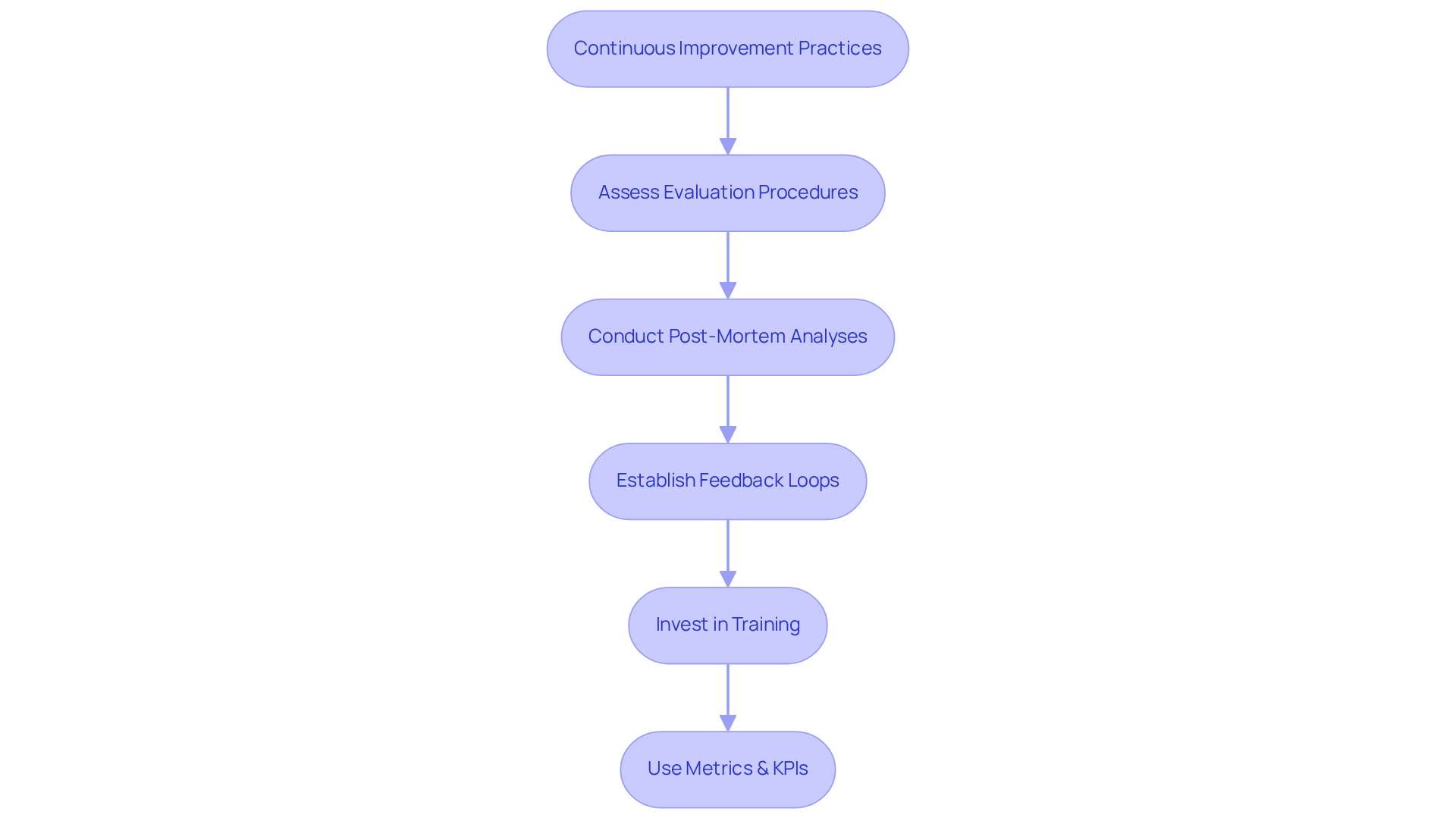
To foster a culture of continuous improvement in healthcare product evaluation, organizations must regularly assess their evaluation procedures and outcomes. This includes conducting to pinpoint areas for enhancement and establishing feedback loops that integrate insights from users and stakeholders. Notably, 75% of medical device producers have experienced a product recall in the past five years, underscoring the critical need for effective evaluation practices.
Moreover, organizations should invest in training and development for their evaluation teams, ensuring they remain updated on the latest assessment methodologies and regulatory changes. The use of metrics and key performance indicators (KPIs) can further assist organizations in tracking their progress and identifying trends over time. For instance, measuring defect rates and time-to-resolution can yield valuable insights into the effectiveness of testing strategies and reveal areas that require attention.
As David Isaacson, Vice President of Product Marketing at ETQ, articulates, “Recalls carry an enormous financial cost with severe brand and regulatory consequences, including costly remediation plans, not to mention the most important issue – patient safety.” By embracing a philosophy of continuous improvement, organizations can enhance software quality through medical device software testing, ensure ongoing compliance, and ultimately deliver safer medical devices to the market.
Conclusion
Achieving compliance in medical device software testing is crucial for ensuring safety, efficacy, and market readiness. Organizations must prioritize a comprehensive understanding of regulatory frameworks, such as the FDA’s 21 CFR Part 820 and ISO 13485, which lay the groundwork for developing effective testing protocols. By aligning with these regulations, manufacturers can enhance their credibility and streamline compliance efforts, ultimately fostering trust in their products.
Key strategies for successful software testing encompass:
- Implementing comprehensive evaluation methods
- Utilizing automated testing tools
- Embracing a culture of continuous improvement
Each testing type – from unit evaluations to user acceptance testing – plays a critical role in validating software performance and mitigating risks. Staying informed about regulatory changes and adopting a risk-oriented approach to testing can significantly enhance patient safety and product reliability.
In conclusion, the medical device industry must acknowledge the significance of rigorous software testing and compliance practices. By investing in training, employing metrics for tracking progress, and fostering feedback loops, organizations can not only refine their evaluation processes but also contribute to the overall safety of healthcare technology. Embracing these best practices is essential for delivering high-quality medical devices that meet regulatory standards and ultimately serve the well-being of patients.
Frequently Asked Questions
What are the key regulatory frameworks for healthcare technology testing?
The key regulatory frameworks include the FDA’s 21 CFR Part 820, which outlines the Quality System Regulation (QSR) for healthcare devices, ISO 13485 for quality management systems, and IEC 62304 for lifecycle processes of healthcare applications.
Why is understanding these regulations important for organizations?
Understanding these regulations is crucial for organizations to develop effective testing protocols and documentation practices, ensuring that applications meet safety and efficacy standards.
What are the validation requirements for software applications intended for Class II health products?
Software applications intended for Class II health products must undergo stringent validation procedures to confirm their intended performance and mitigate potential risks to patients.
What is the projected compliance rate with ISO 13485 among healthcare product companies by 2026?
Compliance rates with ISO 13485 among healthcare product companies are projected to exceed 80% by 2026.
How do regulatory updates affect compliance strategies?
Regulatory updates can directly influence compliance strategies and evaluation methodologies, making it essential for organizations to remain vigilant regarding such changes.
What recent amendments have been made to the FDA’s Part 820?
Recent amendments to Part 820 emphasize the importance of lifecycle accountability and risk management, aligning U.S. regulations with international standards like ISO 13485:2016.
What are the benefits of aligning U.S. regulations with international standards?
Aligning U.S. regulations with international standards streamlines compliance efforts and enhances the credibility of manufacturers in the global market.
List of Sources
- Understand Regulatory Compliance Requirements
- Case Study: Implementing a QMS in a Small Medical Device Company (https://cloudtheapp.com/case-study-implementing-a-qms-in-a-small-medical-device-company)
- FDA QMSR & ISO 13485: Key Changes Effective 2026 | IntuitionLabs (https://intuitionlabs.ai/articles/fda-qmsr-iso-13485-changes-2026)
- FDA Implements Quality Management System Regulation for Medical Devices (https://aabb.org/news-resources/news/article/2026/02/04/fda-implements-quality-management-system-regulation-for-medical-devices)
- QMSR 2026 Explained: FDA Quality Management System Regulation & ISO 13485 Alignment Guide (https://sushvin.com/qmsr-2026-fda-quality-management-system-regulation.html)
- Fda 21 Cfr Part 820 Qsr Medical Device Compliance Checklist | POPProbe Blog (https://popprobe.com/blog/fda-21-cfr-part-820-qsr-medical-device-compliance-checklist)
- Implement Comprehensive Testing Strategies
- Complete guide to software testing in the medical device industry (https://qt.io/quality-assurance/medical-device-software-testing-guide)
- 21 inspirational quotes about software testing (https://testlio.com/blog/21-inspirational-quotes-about-software-testing)
- Medical Device Testing in Transition (https://mpo-mag.com/medical-device-testing-in-transition)
- 50 Inspirational Quotes About Software Testing – QA Madness (https://qamadness.com/inspirational-quotes-about-software-testing)
- The Best Quality Assurance Quotes You Need to Hear (https://rarecrew.com/blog/post/the-best-qa-quotes-you-need-to-hear)
- Adopt Continuous Improvement Practices
- Best Practices for Continuous Improvement in Medical Device Quality (https://cloudtheapp.com/best-practices-for-continuous-improvement-in-medical-device-quality)
- 50 Inspirational Quotes About Software Testing – QA Madness (https://qamadness.com/inspirational-quotes-about-software-testing)
- Medical device manufacturers embrace automated quality management (https://todaysmedicaldevelopments.com/news/medical-device-manufacturers-embrace-automated-quality-management)
- Case Study: Implementing a QMS in a Small Medical Device Company (https://cloudtheapp.com/case-study-implementing-a-qms-in-a-small-medical-device-company)
