Introduction
The healthcare landscape is evolving rapidly, with technology playing a crucial role in enhancing patient care and operational efficiency. As the demand for medical applications increases, developers encounter the significant challenge of navigating complex regulatory environments while delivering user-friendly solutions. This article outlines essential best practices for medical application development, emphasizing:
- Regulatory compliance
- User-centric design
- Structured processes
- The importance of feedback loops
How can developers effectively balance innovation and compliance to create impactful healthcare applications that genuinely meet user needs?
Understand Regulatory Compliance in Medical App Development
In the healthcare sector, regulatory compliance is essential. Developers must familiarize themselves with laws such as HIPAA (Health Insurance Portability and Accountability Act) and FDA regulations that are essential for medical application development. These regulations dictate how patient data is collected, stored, and shared, ensuring user privacy is upheld and that programs are safe for public use. To achieve compliance, developers should take the following steps:
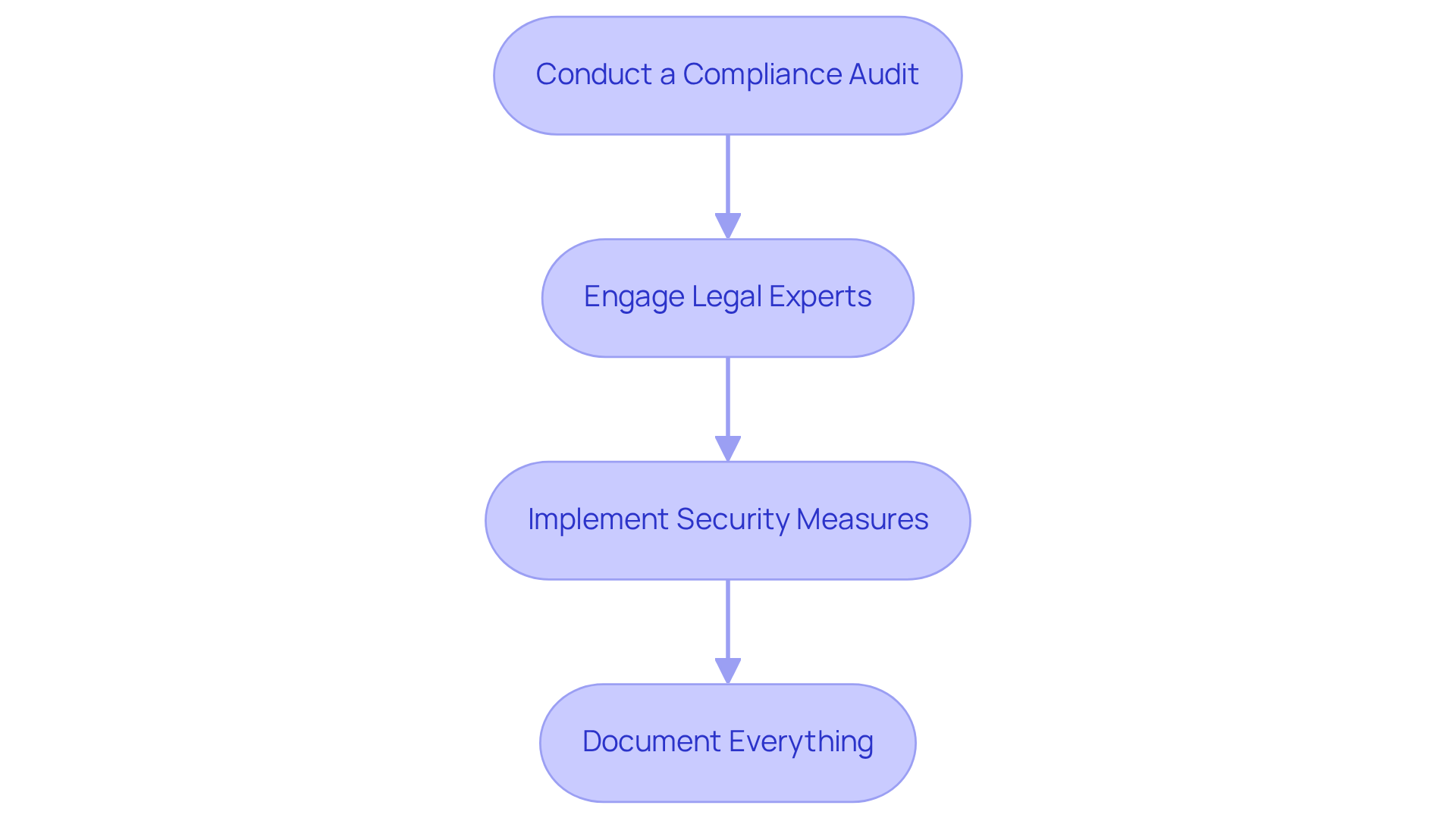
- Conduct a Compliance Audit: Assess existing processes and identify gaps in compliance with relevant regulations.
- Engage Legal Experts: Consult with legal professionals specializing in medical services to ensure all elements of the submission meet regulatory standards.
- Implement Security Measures: Utilize encryption, secure access controls, and regular security audits to protect sensitive patient information.
- Document Everything: Maintain thorough documentation of compliance efforts, which can be crucial during audits or inspections.
By prioritizing compliance from the outset, developers in medical application development can avoid costly penalties and enhance the reliability of their software.
Implement User-Centric Design for Enhanced Engagement
User-focused design is essential for medical application development that prioritizes the needs and preferences of individuals, particularly in medical environments. This approach aims to create intuitive and accessible interfaces for both patients and healthcare professionals. To effectively implement user-centric design, consider the following strategies:
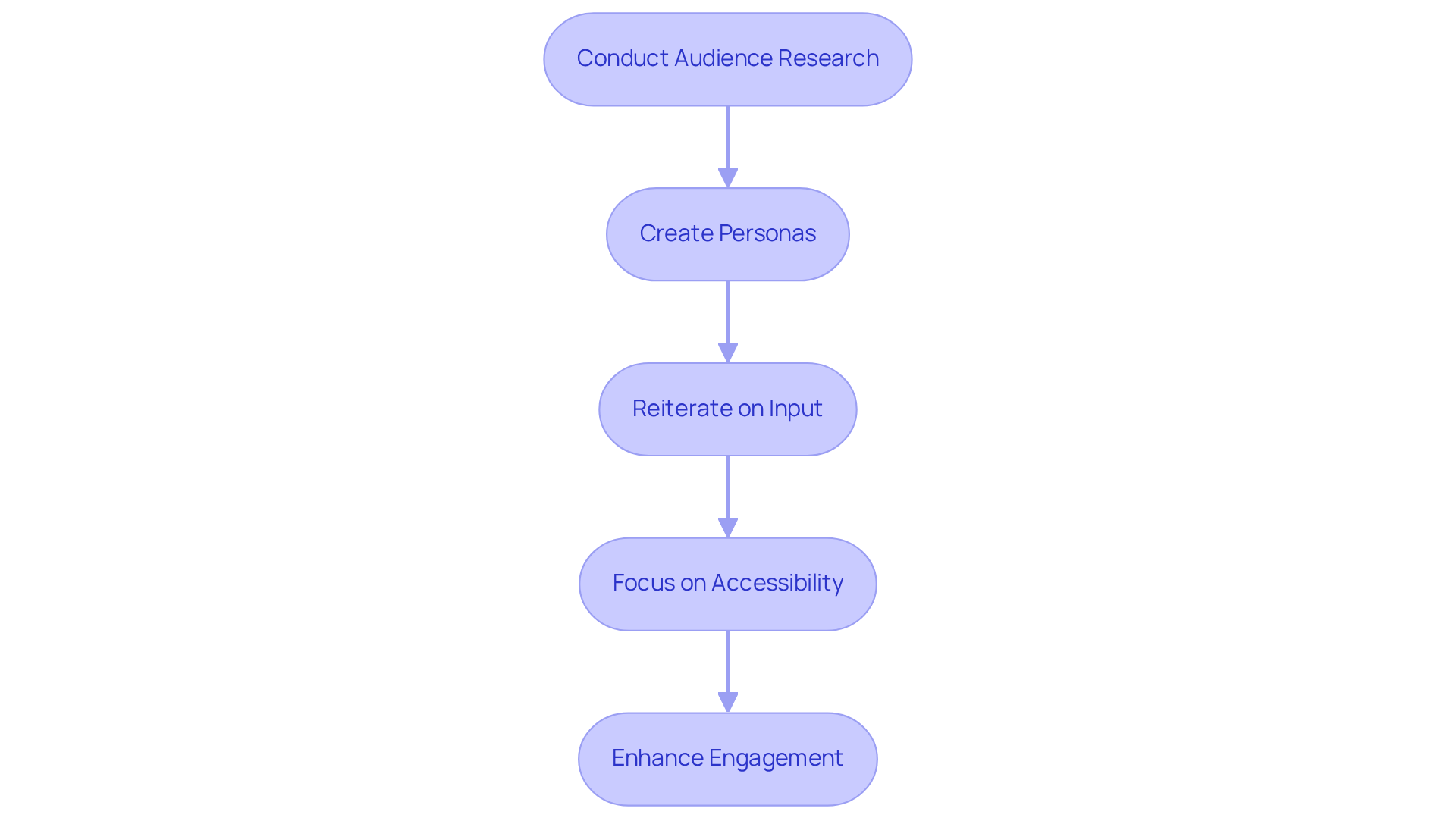
- Conduct Audience Research: Engage potential participants through surveys, interviews, and usability testing to gather valuable insights into their needs and preferences. This foundational step ensures that the application aligns with audience expectations. Continuous research on individuals is vital to adapt to evolving expectations regarding healthcare technology.
- Create Personas: Develop personas that represent various segments of your target audience. These personas serve as a reference for design decisions, aiding in customizing the software to meet diverse needs.
- Reiterate on Input: Utilize insights from testing to consistently enhance the software. This iterative process is crucial for improving the and ensuring the software effectively meets expectations. Empathy plays a significant role in developing human-centered healthcare products, and integrating feedback from users is a key aspect of this.
- Focus on Accessibility: Ensure the software is usable for individuals with disabilities by adhering to established accessibility guidelines, such as the Web Content Accessibility Guidelines (WCAG). This commitment to inclusivity not only broadens the user base but also enhances overall user satisfaction. As noted, ‘90% of the global population that is the poorest has historically been deprived of design ingenuity,’ underscoring the importance of inclusive design in healthcare solutions.
By adopting a user-centric approach, developers can create software in medical application development that not only complies with regulatory standards but also significantly enhances patient engagement and satisfaction. As Dr. Ralf Speth wisely stated, “If you think good design is expensive, you should look at the cost of bad design,” reinforcing the argument for investing in user-centric design.
Follow a Structured Development Process: From Concept to Launch
A structured development process is essential for medical application development to build medical software systematically and efficiently. The following approach is recommended:
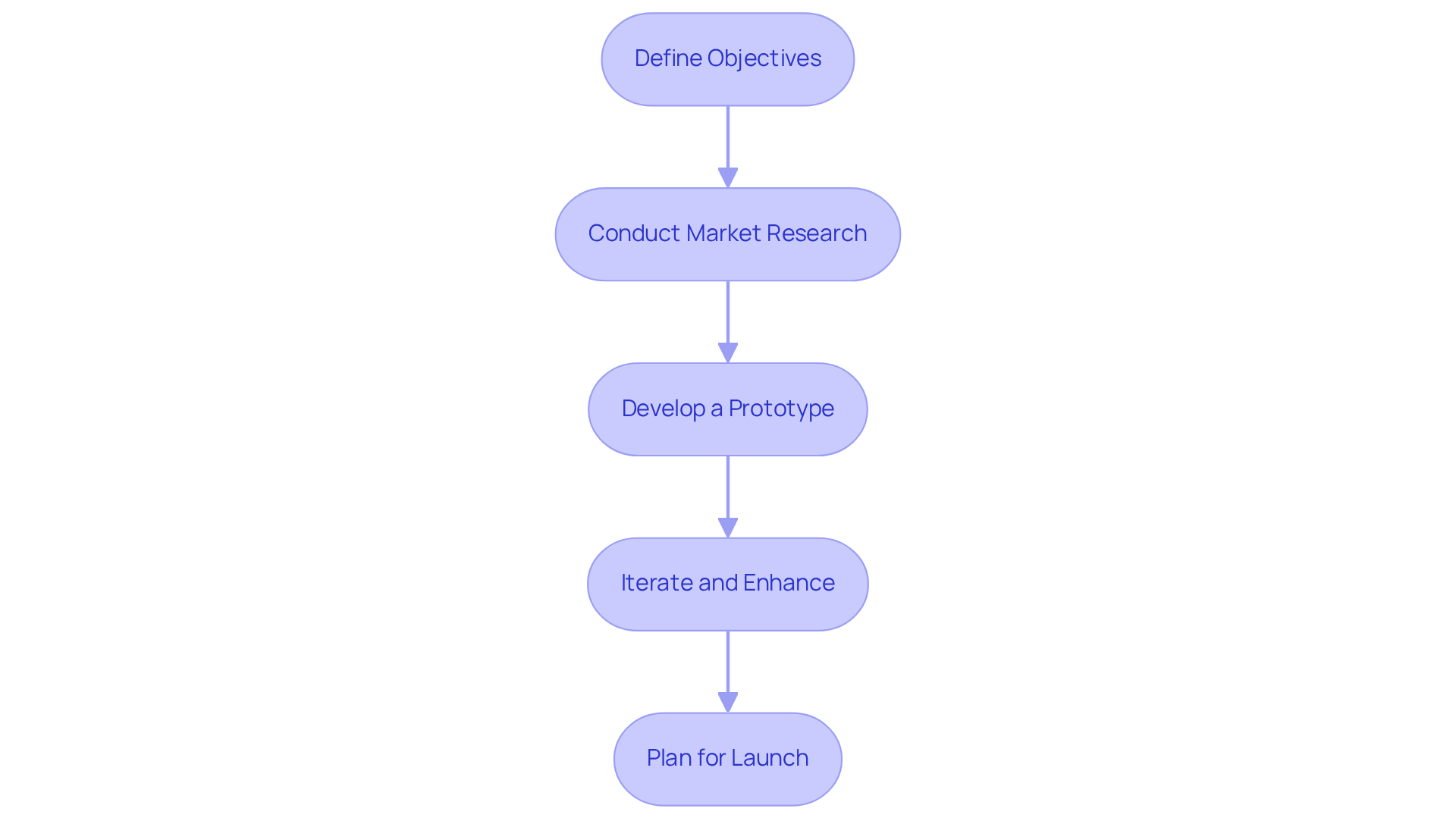
- Define Objectives: Clearly outline the goals of the software, including the target audience and key functionalities. This step is crucial as it aligns the development team with the requirements of the medical field, ensuring that the software addresses specific challenges encountered by individuals.
- Conduct Market Research: Analyze competitors and identify gaps in the market to inform your development strategy. Understanding current trends, such as the shift towards – demonstrated to enhance participant engagement by over 45 percent compared to generic solutions – and the necessity for interoperability, which is now a required feature, can guide the design and functionality of your application.
- Develop a Prototype: Create a minimum viable product (MVP) to test core functionalities and gather feedback early in the process. MVPs are vital in medical application development, enabling teams to validate concepts and make necessary adjustments before full-scale development. Successful case studies illustrate that early participant engagement can significantly enhance the final product’s effectiveness.
- Iterate and Enhance: Utilize agile methodologies to iterate on the application based on feedback from individuals and testing outcomes. Ongoing enhancement is crucial in the rapidly changing medical environment, where consumer needs and regulatory demands can shift swiftly.
- Plan for Launch: Develop a comprehensive launch plan that includes marketing strategies, participant training, and support resources. A well-executed launch can boost adoption and satisfaction, which are essential for the long-term success of medical software.
By adhering to this organized medical application development process, teams can mitigate risks, ensure compliance with healthcare regulations, and deliver high-quality solutions that effectively meet the needs of individuals. The global mobile health market is projected to exceed $300 billion by 2026, underscoring the importance of a well-defined development strategy in a rapidly expanding market.
Establish Feedback Loops for Continuous Improvement and Compliance
Feedback loops are crucial for the ongoing success of medical tools, enabling developers to gather insights from users and implement necessary modifications. As Peter Drucker observed, “The quality of care is ultimately determined by the patient’s perception and experience, not just by the provider’s intentions.” To establish effective feedback loops, consider the following strategies:
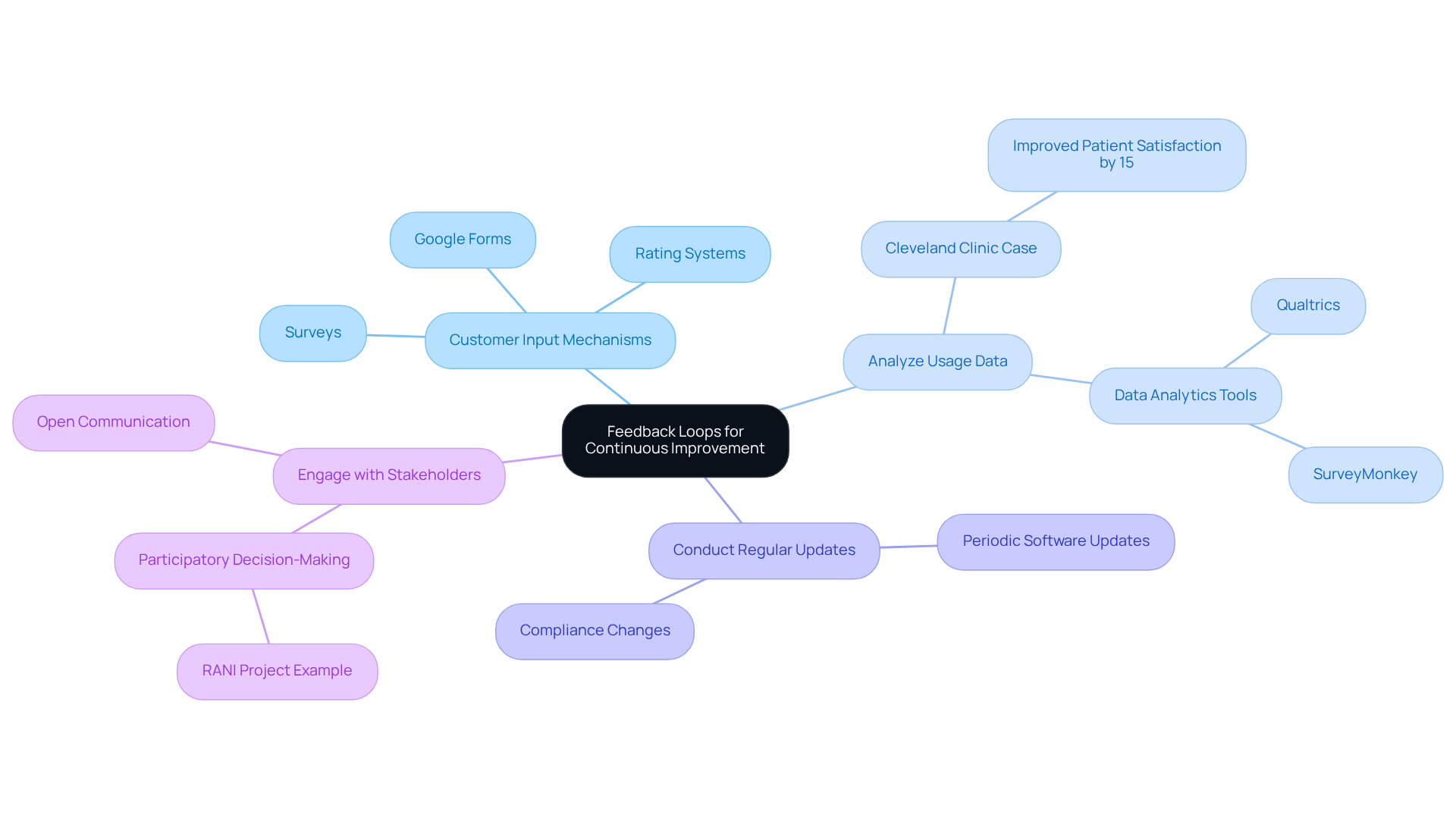
- Implement : Integrate tools within the software that facilitate participant responses, such as surveys or rating systems, to capture real-time insights.
- Analyze Usage Data: Regularly review analytics to understand interactions with the application, identifying trends and areas for enhancement. For example, the Cleveland Clinic improved patient satisfaction scores by 15% over one year by addressing concerns raised in patient surveys.
- Conduct Regular Updates: Schedule periodic updates based on user insights and compliance changes, ensuring the software remains relevant and effective in a dynamic regulatory environment.
- Engage with Stakeholders: Promote open communication with healthcare providers and patients to gather insights and foster a collaborative development atmosphere. The RANI project exemplifies this approach, successfully employing response cycles to enhance intervention delivery and community involvement.
By establishing robust response mechanisms, developers can ensure that their medical application development consistently meets user needs and complies with evolving regulations, ultimately enhancing patient care and operational efficiency. However, it is vital to recognize potential challenges in implementing these loops, such as resistance to change and data privacy concerns, which may impede the effectiveness of feedback mechanisms.
Conclusion
In healthcare, developing medical applications demands a meticulous approach that emphasizes regulatory compliance, user-centric design, and a structured development process. Understanding the significance of adhering to regulations such as HIPAA and FDA guidelines enables developers to create applications that not only safeguard patient data but also enhance the overall safety and reliability of their software. This foundational step is essential for fostering trust and ensuring that medical applications can be effectively utilized in real-world scenarios.
The article outlines several best practices that contribute to successful medical application development. Key strategies include:
- Engaging legal experts to navigate compliance
- Implementing user-centric design principles to boost patient engagement
- Adhering to a structured development process that encompasses clear objectives, market research, and iterative improvements
Furthermore, establishing feedback loops allows developers to continuously refine their applications based on user insights, ultimately leading to improved patient care and satisfaction.
As the healthcare landscape evolves, the importance of these practices cannot be overstated. Developers are urged to adopt these guidelines not only to meet regulatory requirements but also to create innovative solutions that genuinely address user needs. By prioritizing compliance, user experience, and ongoing improvement, the potential for medical applications to transform healthcare delivery and patient outcomes becomes increasingly attainable. The future of healthcare app development relies on a commitment to excellence, and the time to act is now.
Frequently Asked Questions
Why is regulatory compliance important in medical app development?
Regulatory compliance is essential in medical app development to ensure that patient data is collected, stored, and shared in a manner that upholds user privacy and guarantees the safety of the applications for public use.
What are some key regulations developers should be aware of?
Developers should familiarize themselves with laws such as HIPAA (Health Insurance Portability and Accountability Act) and FDA regulations that govern medical application development.
What steps should developers take to achieve compliance?
Developers should conduct a compliance audit, engage legal experts, implement security measures, and maintain thorough documentation of their compliance efforts.
What is involved in conducting a compliance audit?
A compliance audit involves assessing existing processes and identifying any gaps in compliance with relevant regulations.
Why is it important to engage legal experts in the development process?
Engaging legal experts ensures that all elements of the submission meet regulatory standards and helps navigate the complexities of medical regulations.
What security measures should be implemented to protect patient information?
Security measures include utilizing encryption, secure access controls, and conducting regular security audits.
How important is documentation in the compliance process?
Thorough documentation of compliance efforts is crucial, as it can be essential during audits or inspections to demonstrate adherence to regulations.
What are the consequences of not prioritizing compliance in medical app development?
Failing to prioritize compliance can lead to costly penalties and undermine the reliability of the software developed.
