Introduction
In the rapidly evolving landscape of healthcare technology, the development of software for medical devices is pivotal to innovation and compliance. Navigating the complexities of regulatory standards, cybersecurity, and effective development processes is essential for ensuring patient safety and product efficacy. This article examines four best practices that not only enhance the quality of medical device software but also streamline the path to regulatory approval. How can developers effectively balance innovation with the stringent requirements that govern this critical field?
Identify Types of Medical Device Software
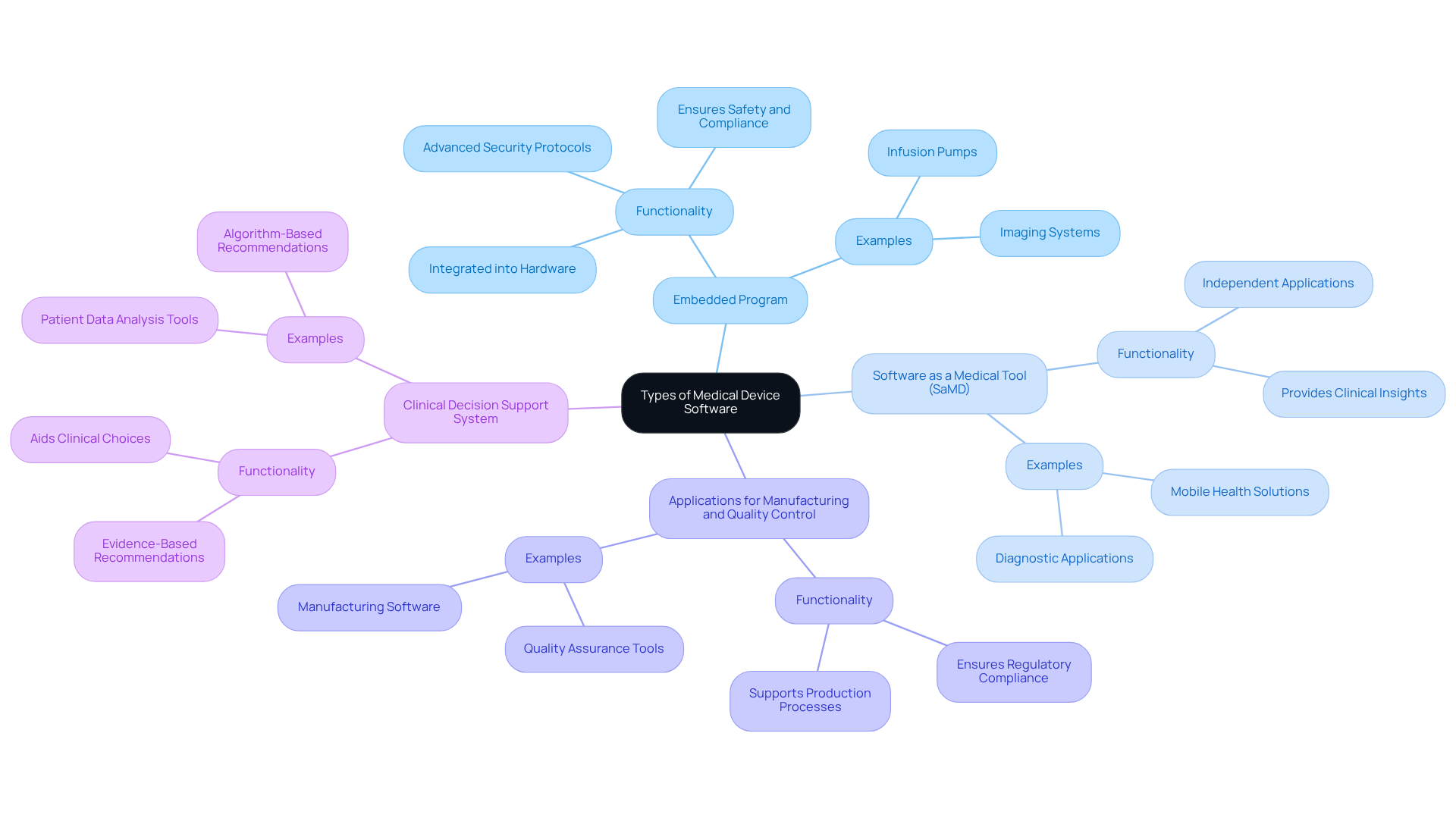
Medical device software can be categorized into several key types, each serving distinct functions and regulatory requirements.
- Embedded Program: This application is integrated directly into the hardware of medical instruments, such as infusion pumps and imaging systems. Its functionality is critical for ensuring safety, compliance, and effective operation, as it often includes advanced security protocols to protect sensitive patient data from cyber threats.
- Software as a Medical Tool (SaMD): This category includes independent applications that carry out medical functions without hardware components. Examples include diagnostic applications and mobile health solutions that provide clinical insights without being tied to specific medical equipment.
- Applications for Manufacturing and Quality Control: This kind of application supports the production and quality assurance processes of medical devices, ensuring adherence to stringent regulatory standards. It plays a vital role in maintaining the integrity and safety of medical products throughout their lifecycle. Neutech’s expertise in application development, including proficiency in languages like Python, GoLang, React, and AWS DevOps, positions them well to deliver robust solutions in this area.
- Clinical Decision Support System: This system aids healthcare professionals in making informed clinical choices based on patient data and algorithms. It enhances the decision-making process by providing evidence-based recommendations, thereby improving patient outcomes. Neutech’s comprehensive engineering services ensure that such applications are developed with the highest quality standards in mind.
By distinctly recognizing these categories, developers can synchronize their processes with the particular legal standards and user expectations linked to each type, ultimately promoting innovation while ensuring adherence and safety in the medical equipment landscape. Furthermore, the incorporation of medical equipment programs with electronic health records (EHRs) and telehealth systems is crucial for smooth data transfer, further improving the functionality and effectiveness of these tools. Neutech is committed to supporting startups and established companies alike in navigating these complexities.
Navigate Regulatory Standards and Compliance
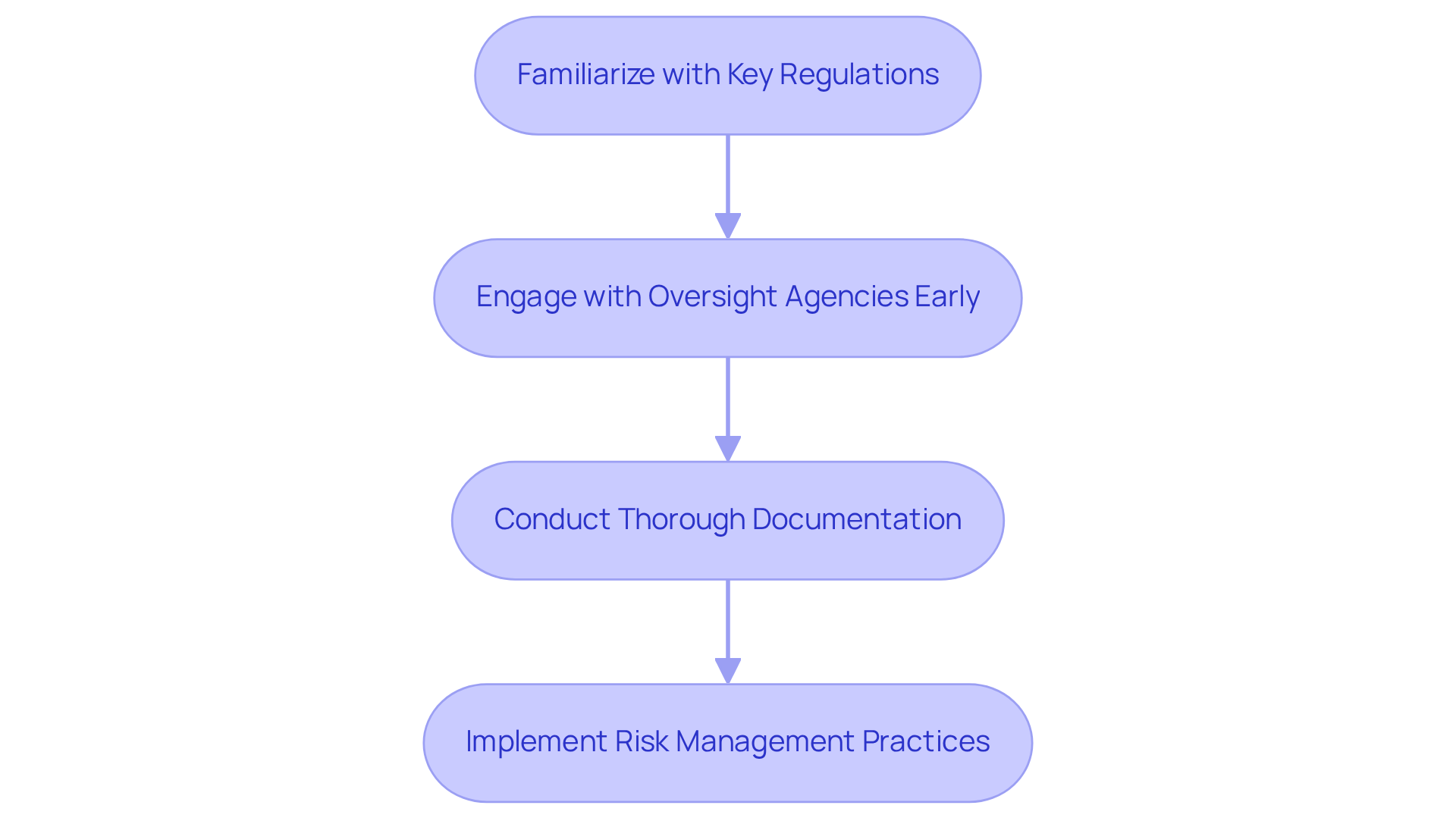
To effectively navigate the regulatory landscape for medical device software, developers should consider several best practices:
- Familiarize with Key Regulations: Understanding relevant regulations, including FDA guidelines, IEC 62304, and ISO 13485, is crucial. These regulations outline requirements for software development medical devices, risk management, and quality assurance, ensuring that products meet safety and efficacy standards. Notably, the revised ISO 13485 compliance criteria for 2026 underscore the importance of aligning quality management systems with international standards, which is essential for regulatory adherence.
- Engage with Oversight Agencies Early: Establishing communication with oversight agencies at the beginning of the development process is vital. This proactive engagement clarifies requirements and expectations, reducing the likelihood of misunderstandings and facilitating a smoother approval process. Research indicates that approximately 70% of medical device application firms that interact early with oversight organizations report improved outcomes in their submissions.
- Conduct Thorough Documentation: Maintaining comprehensive documentation throughout the application development lifecycle is essential. This includes detailed design specifications, testing protocols, and proof of adherence, all of which are critical for successful regulatory submissions. The FDA emphasizes that thorough documentation supports clarity and consistency in the review process, as outlined in their latest guidance documents.
- Implement Risk Management Practices: Adopting a robust risk management framework is necessary to identify, assess, and mitigate risks associated with the application. This practice not only ensures compliance but also prioritizes patient safety, aligning with the updated ISO 13485 compliance requirements for 2026.
By adhering to these best practices, developers involved in software development medical devices can significantly enhance their chances of receiving compliance approval and ensure that their applications meet industry standards. Compliance experts note, “Engaging with the FDA early can significantly streamline the approval process and improve the likelihood of successful submissions.
Implement Structured Software Development Processes
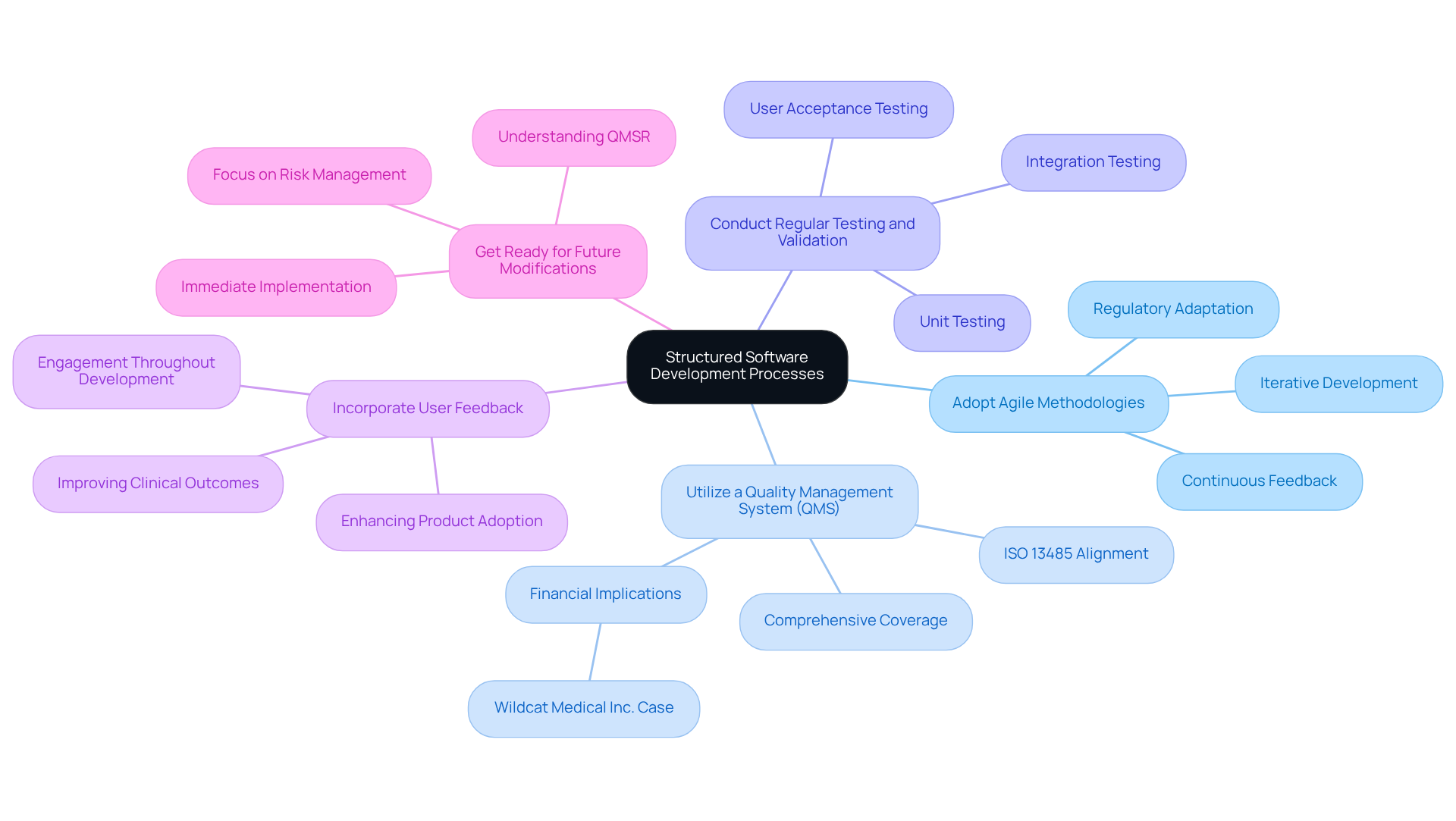
To implement effective software development processes for medical devices, consider the following best practices:
- Adopt Agile Methodologies: Agile practices facilitate iterative development and continuous feedback, enhancing responsiveness to changes and improving product quality. Adapting Agile methodologies in software development medical devices to satisfy regulatory requirements is essential for adherence and efficiency.
- Utilize a Quality Management System (QMS): Implement a QMS that aligns with ISO 13485 standards, which serves as the foundation for U.S. quality system requirements. This system should encompass all aspects of the software development medical devices process, from design to post-market surveillance, ensuring thorough adherence and quality assurance. Notably, Wildcat Medical Inc. spent approximately $100K to update their QMS after the QMSR, highlighting the financial implications of compliance.
- Conduct regular testing and validation in the software development medical devices process to establish a rigorous method that ensures the system meets all functional and regulatory requirements. This should include unit testing, integration testing, and user acceptance testing, which are critical for identifying potential issues early in the development lifecycle.
- Incorporate User Feedback: Engage end-users throughout the development process to gather feedback and ensure that the software meets their needs and expectations. This user-centric approach in software development medical devices not only enhances product adoption but also increases overall satisfaction, leading to better outcomes in clinical settings.
- Get Ready for Future Modifications: With the QMSR scheduled to be implemented on February 2, 2026, it is vital for developers to grasp the new regulatory environment. The FDA’s focus on risk management and adherence in the QMS highlights the significance of implementing these best practices immediately.
By adhering to these organized procedures, developers involved in software development medical devices can greatly improve the quality and compliance of their medical technology, ultimately aiding in enhanced patient safety and regulatory compliance.
Prioritize Cybersecurity and Data Protection
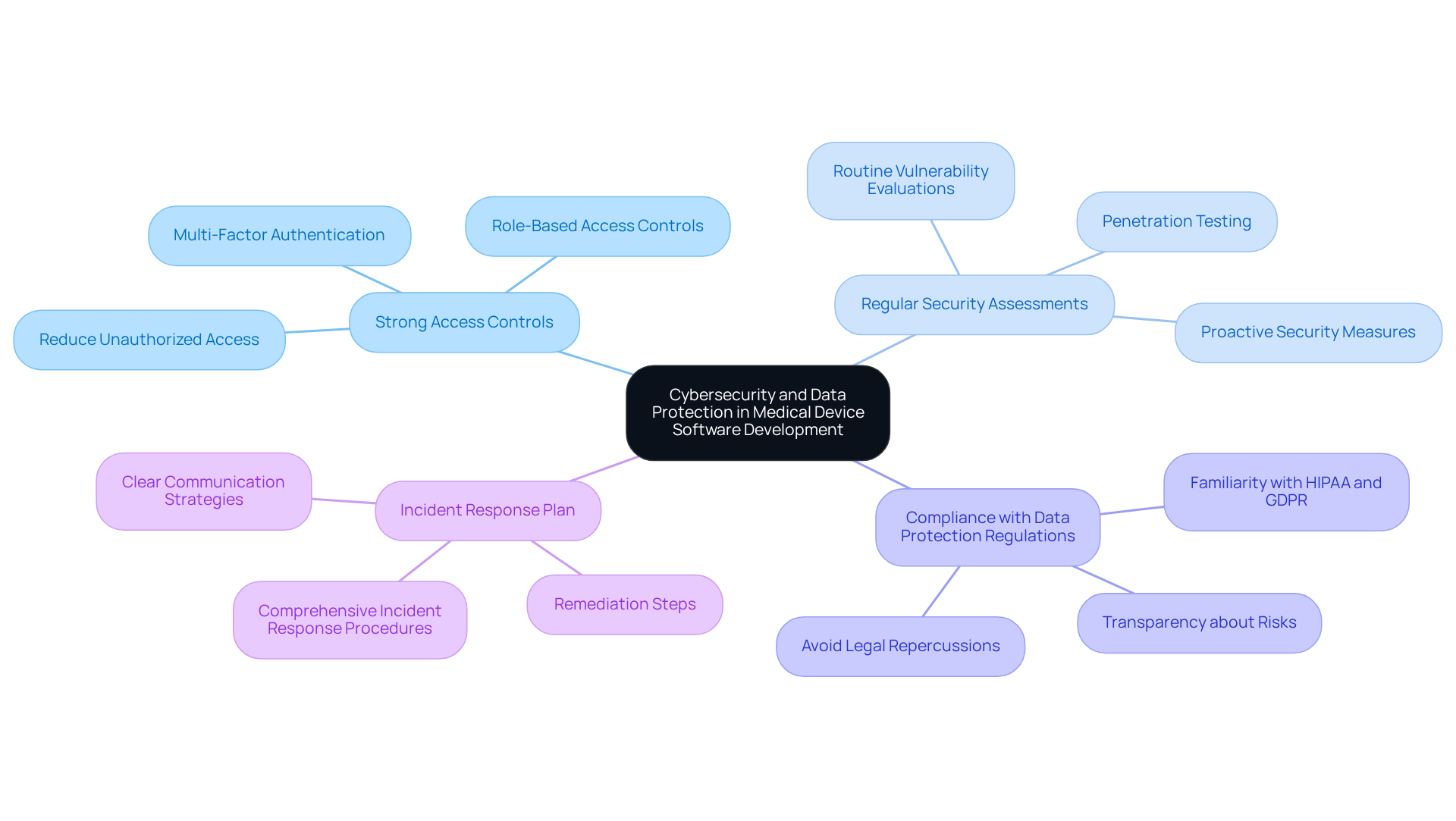
To effectively prioritize cybersecurity and data protection in medical device software development, consider the following strategies:
- Implement Strong Access Controls: Utilize multi-factor authentication and role-based access controls to restrict access to sensitive data and functionalities. This approach significantly reduces the risk of unauthorized access and potential data breaches, which are critical in maintaining patient safety. Phil Englert, Director of Medical Equipment Security, emphasizes that “Medical instruments account for between 5% and 11% of the endpoints, while your Internet of Things and operational technology population represents about 30%.” This underscores the importance of securing these devices to protect patient safety.
- Conduct Regular Security Assessments: Routine vulnerability evaluations and penetration testing are crucial for identifying and addressing potential security weaknesses in the system. These proactive measures help maintain a robust security posture, ensuring that any vulnerabilities are addressed before they can be exploited. With cybercrime projected to cost the world $23 trillion by 2027, the stakes for maintaining security are higher than ever.
- Ensure Compliance with Data Protection Regulations: Familiarize yourself with relevant data protection regulations such as HIPAA and GDPR. Compliance with these regulations is essential for protecting patient data and avoiding legal repercussions, as non-compliance can lead to severe penalties and damage to reputation. Overlooking cybersecurity can result in significant consequences, including compliance issues and risks to patient safety.
- Develop a Robust Incident Response Plan: Establish a comprehensive incident response plan that outlines procedures for addressing security breaches or data leaks. This plan should include clear communication strategies and remediation steps to minimize the impact of any incidents, ensuring a swift recovery and maintaining trust with stakeholders. The average number of cyberattacks per organization per year has increased by 25%, making it essential to be prepared for potential incidents.
By prioritizing these cybersecurity and data protection strategies, developers can significantly enhance the safety and reliability of software development medical devices, ultimately contributing to better patient outcomes.
Conclusion
In the field of medical device software development, adhering to best practices is essential for ensuring safety, compliance, and innovation. By categorizing medical device software into distinct types, developers can tailor their approaches to meet specific regulatory requirements and user expectations. This clarity fosters adherence to safety standards and encourages the integration of advanced technologies that enhance patient care.
Key insights emphasize the importance of navigating regulatory standards, implementing structured development processes, and prioritizing cybersecurity. Understanding regulations such as FDA guidelines and ISO standards is crucial for compliance. Additionally, adopting agile methodologies and a robust quality management system can significantly improve product quality. The urgency of cybersecurity measures and data protection is vital, as they safeguard sensitive patient information and maintain trust in medical technologies.
As the landscape of medical device software evolves, it is imperative for developers to remain vigilant and proactive. Embracing these best practices aids in achieving regulatory compliance and enhances the overall quality and safety of medical devices. By prioritizing patient safety and leveraging innovative solutions, developers can contribute to a future where medical technology meets and exceeds the expectations of healthcare professionals and patients alike.
Frequently Asked Questions
What are the main categories of medical device software?
Medical device software can be categorized into four main types: Embedded Program, Software as a Medical Tool (SaMD), Applications for Manufacturing and Quality Control, and Clinical Decision Support System.
What is an Embedded Program in medical device software?
An Embedded Program is integrated directly into the hardware of medical instruments, such as infusion pumps and imaging systems. It is critical for ensuring safety, compliance, and effective operation, often including advanced security protocols to protect sensitive patient data.
What does Software as a Medical Tool (SaMD) refer to?
Software as a Medical Tool (SaMD) refers to independent applications that perform medical functions without being tied to hardware components. Examples include diagnostic applications and mobile health solutions that provide clinical insights.
What role do Applications for Manufacturing and Quality Control play in medical devices?
Applications for Manufacturing and Quality Control support the production and quality assurance processes of medical devices, ensuring adherence to regulatory standards and maintaining the integrity and safety of medical products throughout their lifecycle.
How does a Clinical Decision Support System function?
A Clinical Decision Support System aids healthcare professionals by providing evidence-based recommendations based on patient data and algorithms, thereby improving the decision-making process and patient outcomes.
Why is it important for developers to recognize different types of medical device software?
Recognizing different types allows developers to align their processes with specific legal standards and user expectations, promoting innovation while ensuring adherence and safety in the medical equipment landscape.
How do medical device software programs integrate with electronic health records (EHRs) and telehealth systems?
The incorporation of medical device software with EHRs and telehealth systems is crucial for smooth data transfer, enhancing the functionality and effectiveness of these tools.
What support does Neutech provide to companies in the medical device software space?
Neutech supports both startups and established companies in navigating the complexities of medical device software development, ensuring adherence to regulations and quality standards.
List of Sources
- Identify Types of Medical Device Software
- How Embedded Software is transforming Healthcare devices (https://exaud.com/blog/embedded-software-solutions-in-healthcare-devices)
- For 2026, FDA signals shifts in digital health framework | Nixon Peabody LLP (https://nixonpeabody.com/insights/alerts/2026/01/27/for-2026-fda-signals-shifts-in-digital-health-framework)
- Five Major Trends Shaping Medical Device Software (https://aami.org/news/five-major-trends-shaping-medical-device-software)
- AI-Enabled Medical Devices (https://fda.gov/medical-devices/software-medical-device-samd/artificial-intelligence-enabled-medical-devices)
- FDA Digital Health Guidance: 2026 Requirements Overview | IntuitionLabs (https://intuitionlabs.ai/articles/fda-digital-health-technology-guidance-requirements)
- Navigate Regulatory Standards and Compliance
- What FDA Guidance Means For The Future Of Health Software (https://cov.com/en/news-and-insights/insights/2026/02/what-fda-guidance-means-for-the-future-of-health-software)
- FDA’s 2026 Guidance Expands Pathway for Low-Risk Digital Health Products—But Caution Remains Essential | Berkley Lifesciences (https://berkleyls.com/blog/fdas-2026-guidance-expands-pathway-low-risk-digital-health-products-caution-remains-essential)
- For 2026, FDA signals shifts in digital health framework | Nixon Peabody LLP (https://nixonpeabody.com/insights/alerts/2026/01/27/for-2026-fda-signals-shifts-in-digital-health-framework)
- FDA Implements Quality Management System Regulation for Medical Devices (https://aabb.org/news-resources/news/article/2026/02/04/fda-implements-quality-management-system-regulation-for-medical-devices)
- FDA Digital Health Guidance: 2026 Requirements Overview | IntuitionLabs (https://intuitionlabs.ai/articles/fda-digital-health-technology-guidance-requirements)
- Implement Structured Software Development Processes
- The QMSR Goes Live and FDA Implements a New Medical Device Inspection Technique | JD Supra (https://jdsupra.com/legalnews/the-qmsr-goes-live-and-fda-implements-a-8697798)
- FDA QMSR & ISO 13485: Key Changes Effective 2026 | IntuitionLabs (https://intuitionlabs.ai/articles/fda-qmsr-iso-13485-changes-2026)
- MedTech Product Development Trends in 2026 (https://enlil.com/blog/medtech-product-development-trends-in-2026)
- Quality Management System Regulation: Final Rule – FAQ (https://fda.gov/medical-devices/quality-management-system-regulation-qmsr/quality-management-system-regulation-frequently-asked-questions)
- What the FDA’s New Quality Requirements Mean for U.S. Medical Device Companies (https://amtivo.com/us/resources/insights/fdas-new-quality-requirements-mean-for-us-medical-device-companies)
- Prioritize Cybersecurity and Data Protection
- FDA Tightens Its Medical Device Cybersecurity Guidance (https://fedtechmagazine.com/article/2026/03/fda-tightens-its-medical-device-cybersecurity-guidance-perfcon)
- Key Cyber Security Statistics for 2026 (https://sentinelone.com/cybersecurity-101/cybersecurity/cyber-security-statistics)
- Cybersecurity: Quality System Considerations and Premarket Submissions (https://fda.gov/regulatory-information/search-fda-guidance-documents/cybersecurity-medical-devices-quality-management-system-considerations-and-content-premarket)
- Why many existing medical devices fall short of the FDA’s new cybersecurity standards (https://todaysmedicaldevelopments.com/news/why-many-existing-medical-devices-fall-short-fda-new-cybersecurity-standards)
- FDA Cybersecurity Guidance 2026: What Changed? (https://hattrick-it.com/blog/cybersecurityguidanceupdate)
