Introduction
In the rapidly evolving landscape of the pharmaceutical industry, the integration of effective software solutions is essential for ensuring compliance and operational efficiency. Organizations have a unique opportunity to enhance their processes by adopting best practices in pharmaceutical software management. However, navigating the complex regulatory requirements presents a significant challenge. Companies must select tools that not only meet compliance standards but also optimize performance.
To address these challenges, what strategies can organizations employ to streamline their software implementation and training processes while ensuring adherence to stringent regulations?
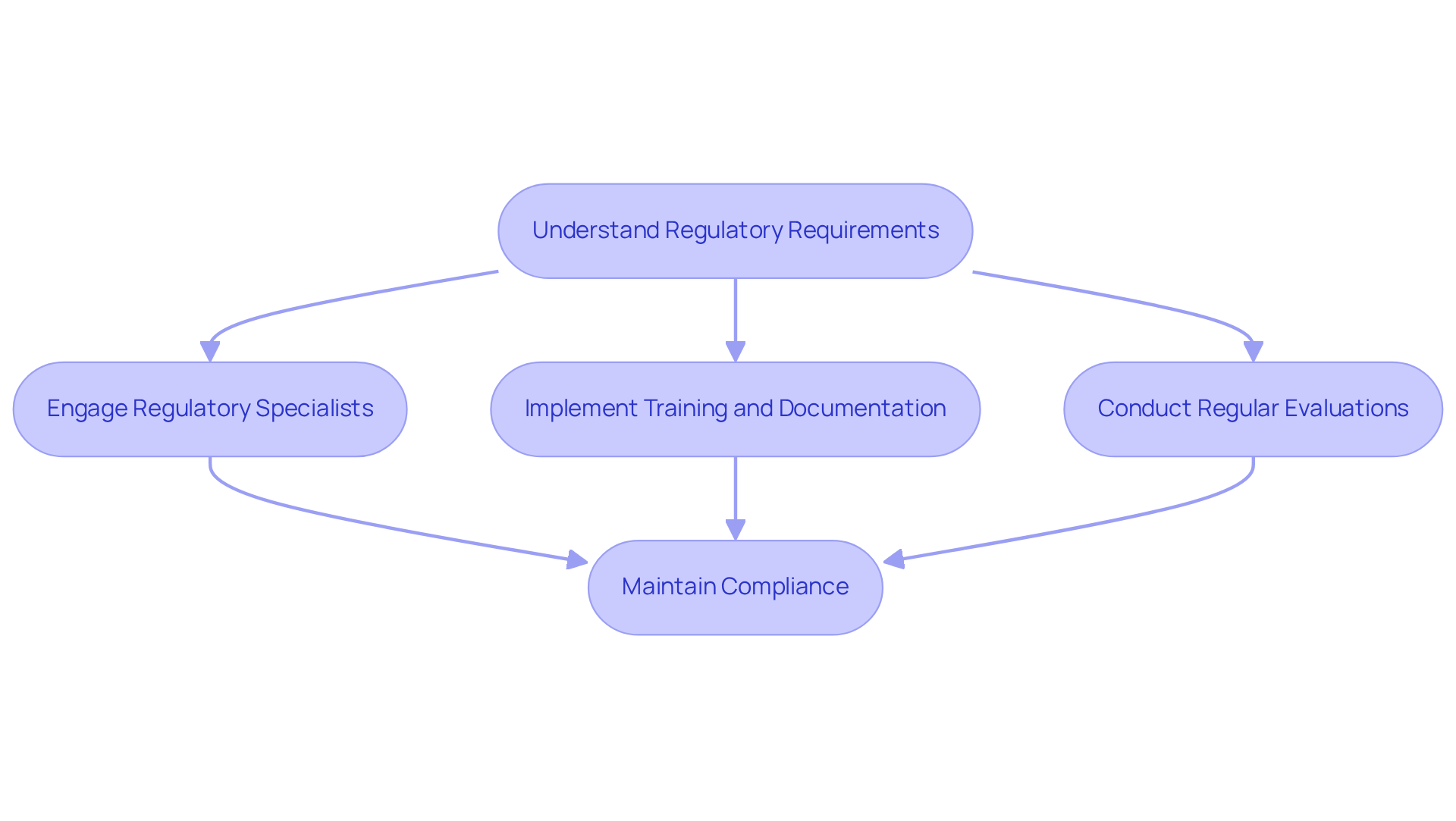
Understand Regulatory Compliance Requirements
must comply with a variety of regulations, including FDA guidelines, EU directives, and . Companies should begin by thoroughly understanding the specific regulations pertinent to their technological solutions. This entails a comprehensive grasp of the requirements for as outlined in , which mandates that electronic signatures be unique to individuals and linked to their corresponding records to prevent unauthorized alterations. Furthermore, specified in EU Annex 11 must be taken into account, emphasizing the necessity for robust controls to safeguard electronic records from unauthorized access, alteration, or deletion.
Engaging with early in the application development process is crucial for identifying potential pitfalls and ensuring that the system is designed with compliance in mind. As highlighted by quality management experts, “A compliant system is only effective if your team knows how to use it correctly,” underscoring the significance of . Regular evaluations and updates to the system are also essential to maintain , with proactive serving as a vital tool for remaining inspection-ready. By implementing these best practices, companies can effectively navigate the complexities of regulatory adherence, safeguarding data integrity and ensuring patient safety.
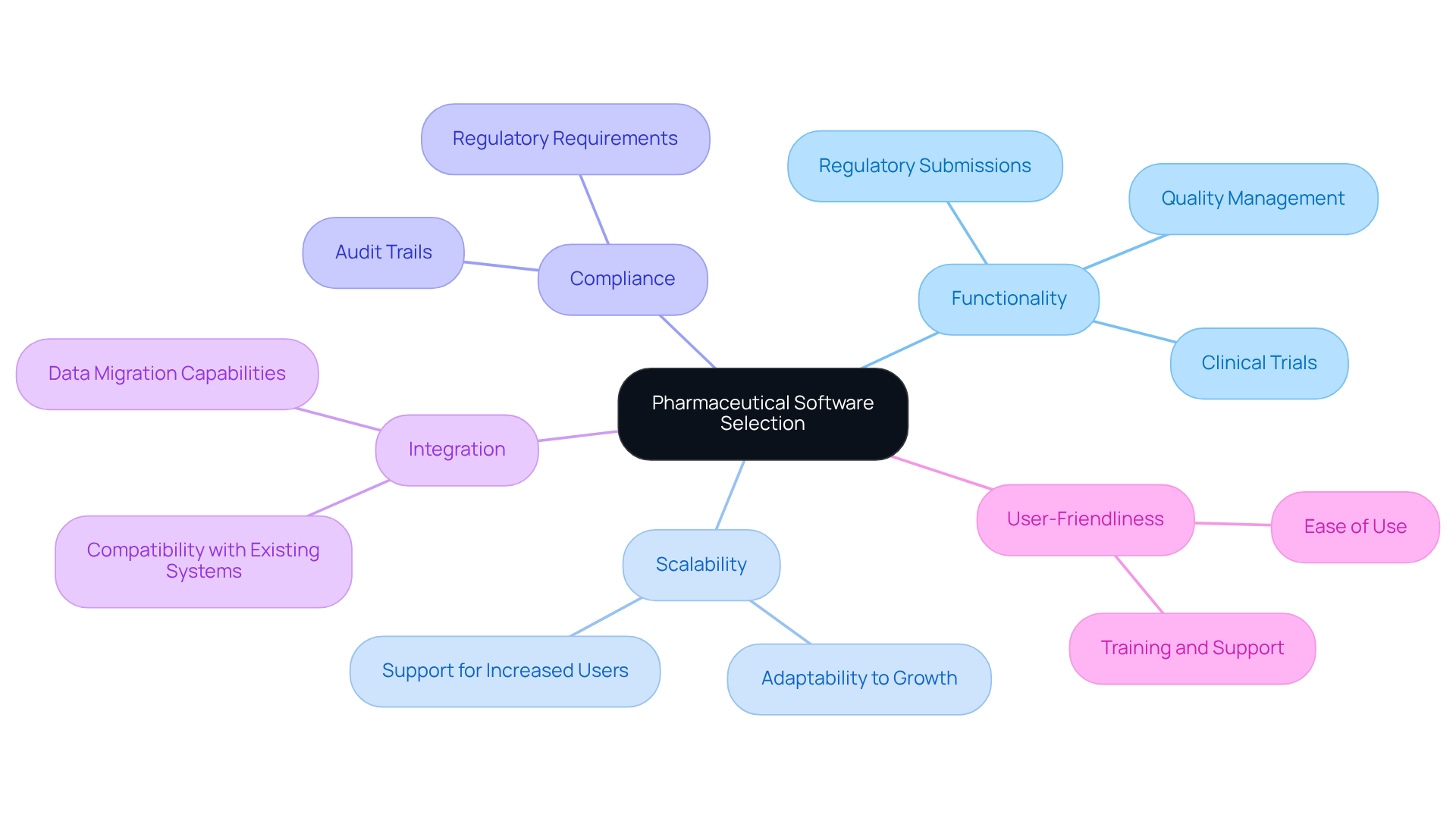
Select Appropriate Pharmaceutical Software Solutions
When selecting , organizations must consider key factors such as functionality, scalability, and compliance capabilities. It is crucial to evaluate applications that support , including , , and quality management. Additionally, companies should assess the application’s ability to integrate with existing systems and its user-friendliness. Conducting a thorough needs assessment and involving end-users in the selection process can significantly enhance adoption rates and overall satisfaction. Moreover, examining from similar organizations can provide valuable insights into the program’s effectiveness.
For instance, the FDA’s generative AI pilot initiative illustrates how prioritizing can lead to improved outcomes in drug approval processes. Industry leaders also highlight the importance of selecting that not only fulfill regulatory requirements but also . Specific technological solutions, such as Epic’s ERP capabilities and other platforms designed for clinical trial management, can notably streamline and bolster compliance adherence.
Organizations should remain vigilant about common pitfalls in the application selection process, such as failing to involve or neglecting to conduct a . By adhering to these best practices, companies can ensure they choose the appropriate that align with their operational needs and support their strategic objectives.
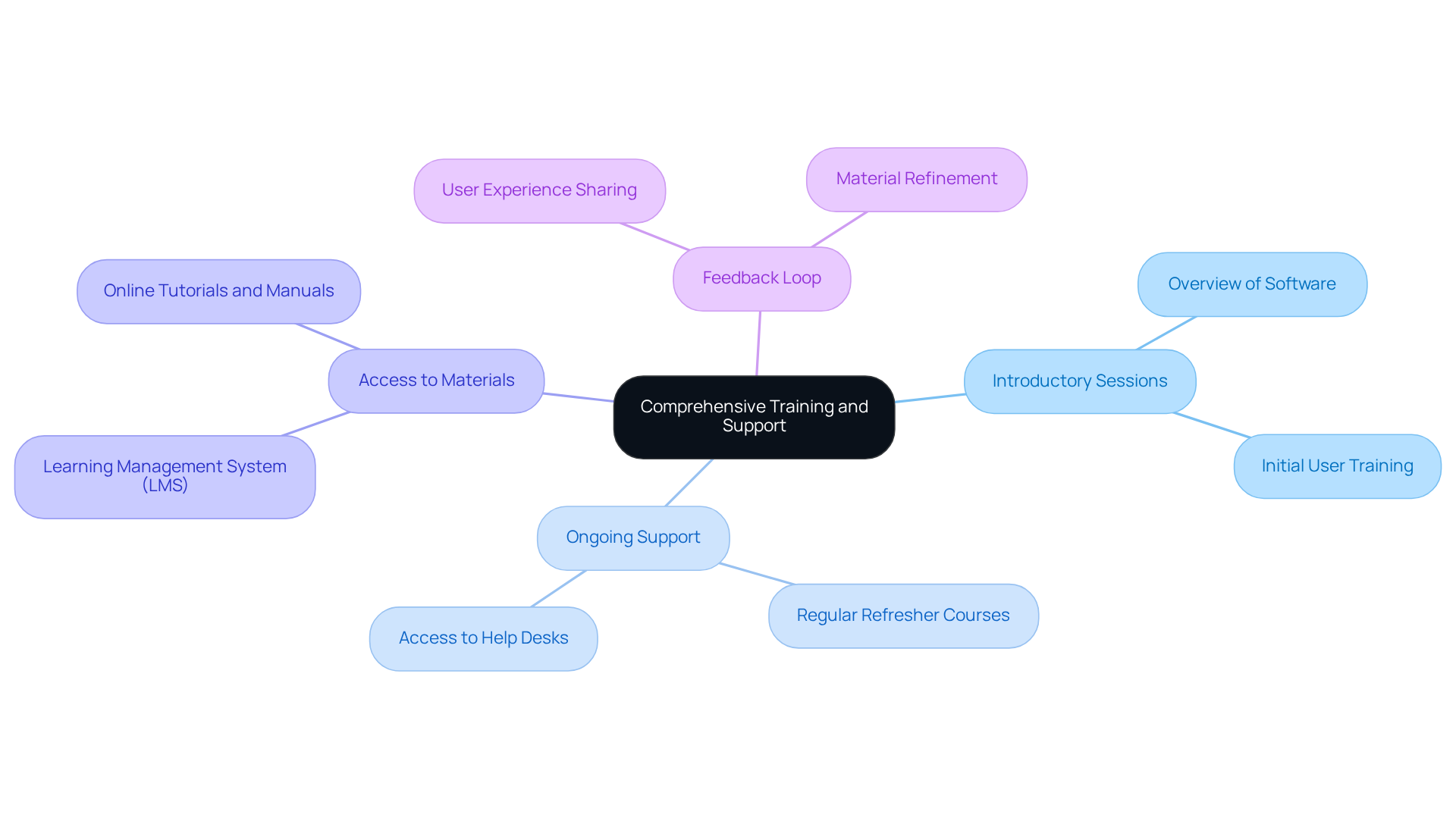
Provide Comprehensive Training and Support
To enhance the effectiveness of , organizations must prioritize for their participants. This encompasses:
- Introductory sessions
- Ongoing support
- such as manuals and online tutorials
Implementing a can significantly streamline the development process, facilitating easy tracking of participant progress and compliance education. For instance, firms with report a 218% increase in revenue, underscoring the financial benefits of investing in customer education.
Establishing a where individuals can share their experiences and challenges is crucial for refining educational materials and support resources. Regular and updates on new features or regulatory changes are also vital to keep users informed and proficient. Research shows that organizations employing achieve 41% more accurate identification of skill gaps, further enhancing training effectiveness. By cultivating a culture of , pharmaceutical companies can ensure their teams are well-prepared to navigate the complexities of the industry.
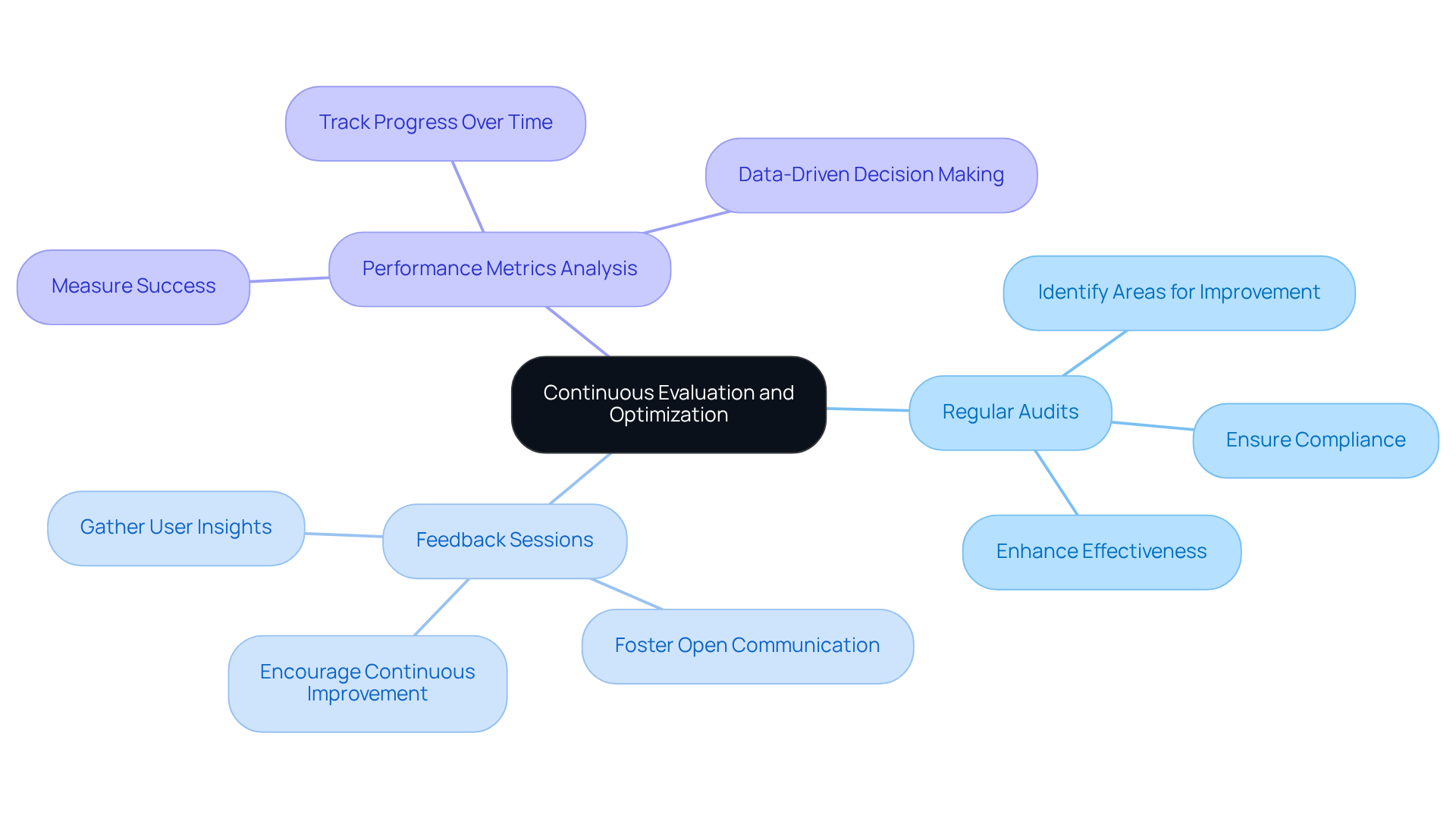
Implement Continuous Evaluation and Optimization
To ensure the effectiveness and compliance of , organizations must implement a robust framework for continuous evaluation. This framework should include:
- Regular audits
- Feedback sessions
These elements are crucial to identify areas for improvement. As Mike Cohn asserts, if a team is not improving month over month, they are no longer agile, underscoring the importance of within . Implementing these methodologies allows for that align with client needs and regulatory changes.
Moreover, leveraging analytics tools offers valuable insights into application usage patterns, enabling organizations to make informed, for optimization. For example, the case study “” demonstrates how can enhance products and improve user experiences. By , companies can swiftly adapt to the evolving landscape of the pharmaceutical sector, ensuring their remain effective and compliant.
Additionally, with December 17, 2025, projected to be the most expensive year in history for IT investments, optimizing software solutions is essential for cost-effectiveness.
Conclusion
In the realm of pharmaceutical software solutions, adherence to best practices is essential for ensuring compliance, efficiency, and overall success. By prioritizing regulatory understanding, selecting appropriate software, providing thorough training, and committing to continuous evaluation, organizations can establish a robust framework that supports their operational goals while safeguarding patient safety and data integrity.
Key insights from this discussion highlight the significance of engaging with regulatory experts, conducting comprehensive needs assessments, and fostering a culture of continuous learning. These elements not only facilitate compliance with FDA and EU regulations but also enhance user satisfaction and operational effectiveness. Moreover, regular audits and feedback mechanisms are crucial for identifying areas of improvement, enabling companies to adapt swiftly to the evolving landscape of the pharmaceutical industry.
Ultimately, the successful implementation of pharmaceutical software solutions relies on a proactive approach that integrates compliance, user engagement, and ongoing optimization. As organizations prepare for the future, a commitment to these best practices will streamline operations and position them favorably in an increasingly competitive market. Embracing these strategies will be vital for navigating the complexities of pharmaceutical software in 2025 and beyond, ensuring that innovations translate into improved healthcare outcomes.
Frequently Asked Questions
What are the main regulatory compliance requirements for pharmaceutical applications?
Pharmaceutical applications must comply with various regulations, including FDA guidelines, EU directives, and Good Manufacturing Practices (GMP).
What specific regulations should companies focus on regarding electronic records and signatures?
Companies should focus on the requirements outlined in 21 CFR Part 11, which mandates that electronic signatures be unique to individuals and linked to their corresponding records to prevent unauthorized alterations.
What standards must be considered for data integrity in the EU?
Data integrity standards specified in EU Annex 11 must be taken into account, emphasizing the necessity for robust controls to safeguard electronic records from unauthorized access, alteration, or deletion.
Why is it important to engage with regulatory specialists during application development?
Engaging with regulatory specialists early in the application development process is crucial for identifying potential pitfalls and ensuring that the system is designed with compliance in mind.
What role does training and documentation play in regulatory compliance?
Training and documentation are significant because a compliant system is only effective if the team knows how to use it correctly.
How can companies maintain compliance as regulations evolve?
Companies can maintain compliance by conducting regular evaluations and updates to the system, with proactive internal assessments serving as a vital tool for remaining inspection-ready.
What are the best practices for navigating regulatory adherence in pharmaceutical applications?
Best practices include understanding specific regulations, engaging regulatory specialists, ensuring proper training and documentation, and conducting regular evaluations and updates to maintain compliance.
