Introduction
Quality Assurance (QA) is a fundamental element in regulated industries, where adherence to stringent standards is not just a guideline but a legal requirement. As sectors such as finance, healthcare, and pharmaceuticals encounter heightened regulatory scrutiny, the necessity for effective QA strategies becomes critical. This article examines the complexities of QA within these industries, highlighting tailored approaches that ensure compliance and foster continuous improvement.
How can organizations effectively navigate the intricate regulatory landscape while enhancing their operational integrity and achieving success?
Understand Quality Assurance in Regulated Industries
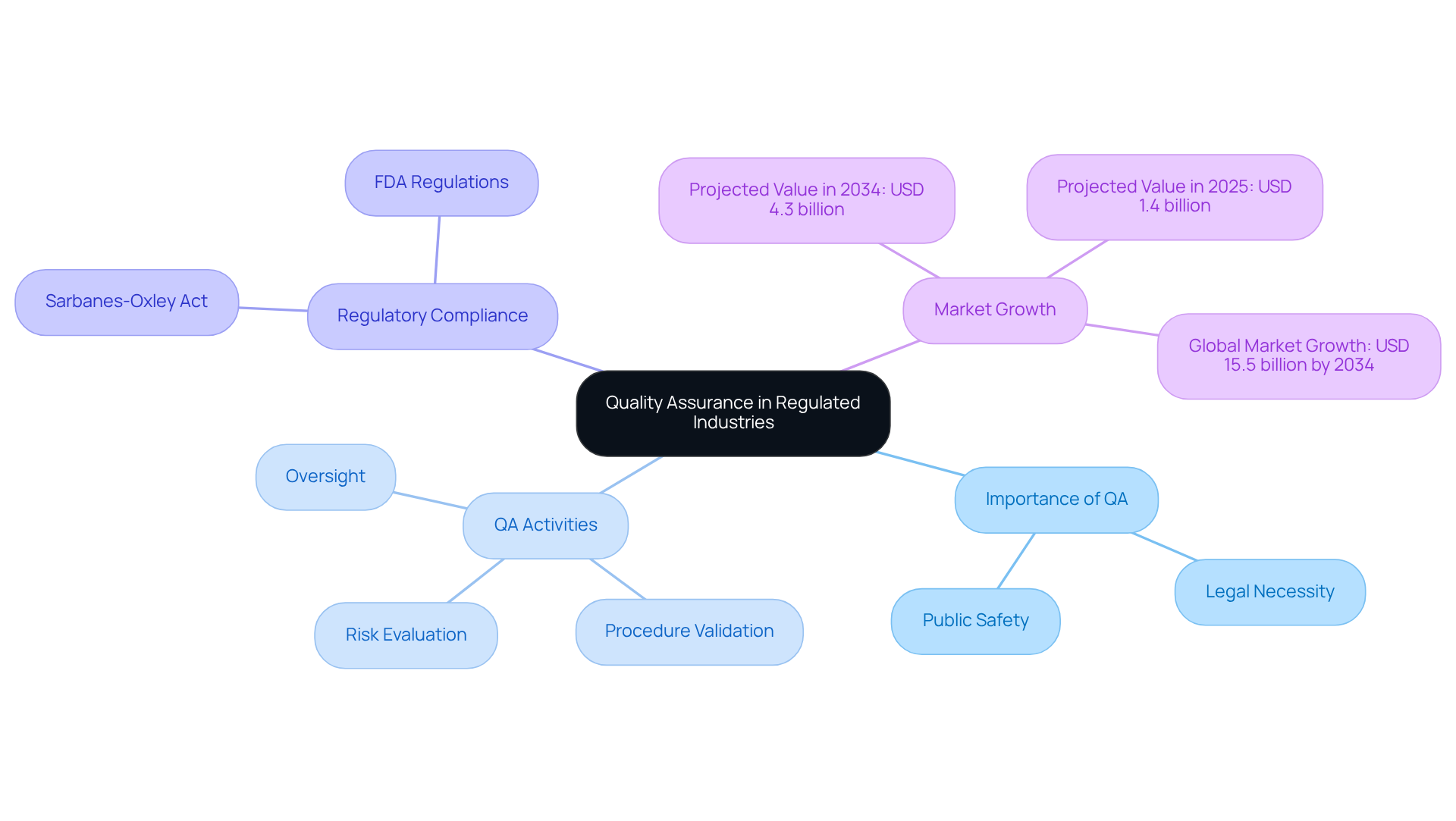
Quality Assurance (QA) in regulated sectors involves systematic methods and practices that ensure products and services meet established quality standards and comply with regulatory requirements. In industries such as finance, healthcare, and pharmaceuticals, QA is not just a best practice; it is a legal necessity. This encompasses a range of activities, including risk evaluation, procedure validation, and oversight, all aimed at safeguarding public safety and ensuring product effectiveness.
For example, in the financial sector, QA processes must comply with regulations like the Sarbanes-Oxley Act, which enforces stringent controls over financial reporting to enhance transparency and accountability. Understanding these frameworks is essential for organizations to navigate the complexities of regulations and maintain operational integrity.
The US Quality Assurance Services Market is projected to grow significantly, expected to reach USD 4.3 billion by 2034, up from an estimated USD 1.4 billion in 2025. This growth underscores the increasing demand for high-quality regulatory solutions. As Zoya Khan notes, ‘By 2026, adherence pressure will transition from policy intent to operational proof,’ highlighting the imperative for organizations to demonstrate their commitment to quality and compliance through measurable actions.
Identify Challenges in QA for Regulated Industries
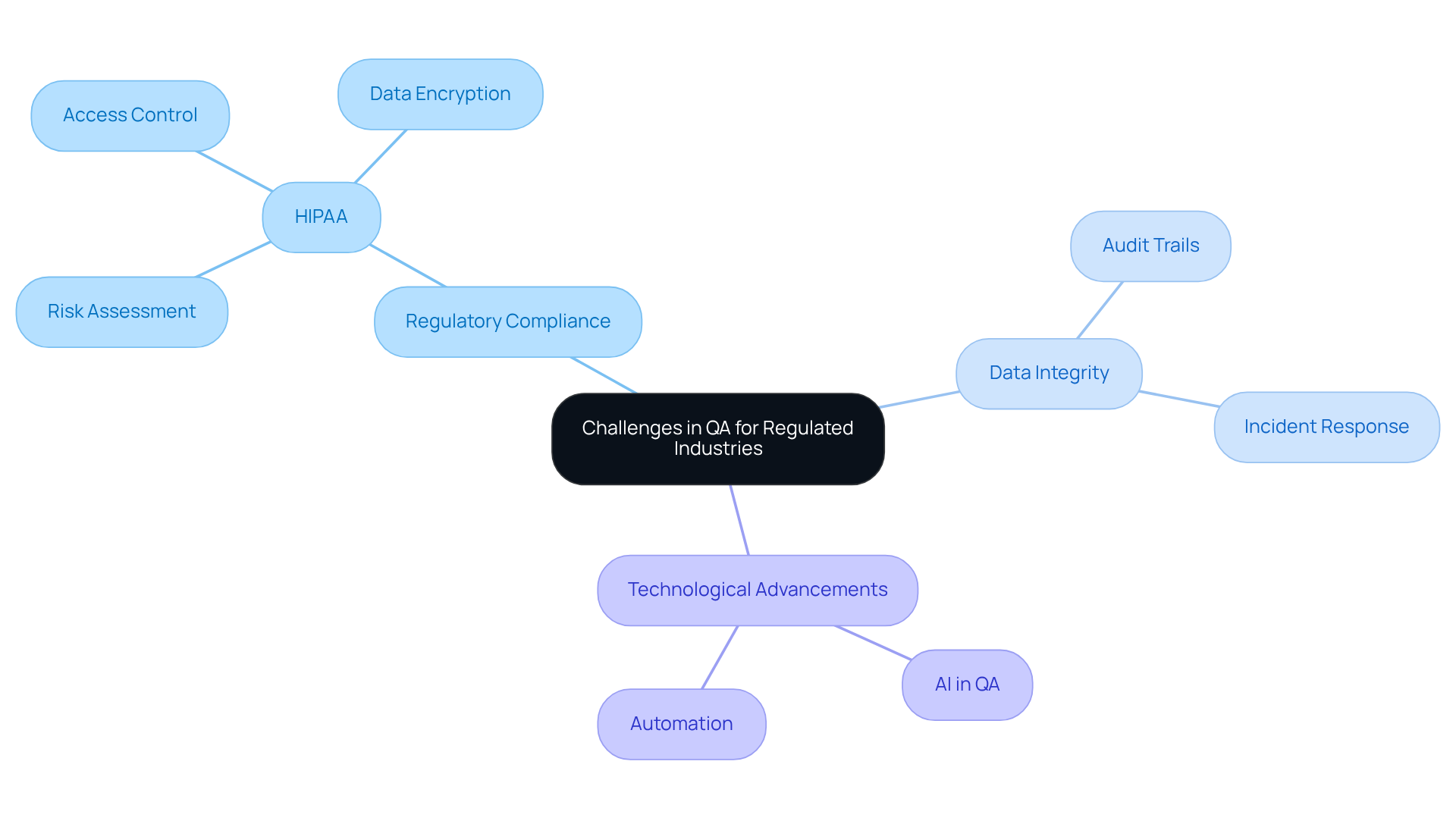
Organizations in regulated industries encounter significant challenges in their QA software processes, particularly when navigating complex regulatory landscapes and ensuring data integrity. A prime example of these challenges is the Health Insurance Portability and Accountability Act (HIPAA), which imposes stringent requirements on data handling and patient privacy. Compliance evaluation must ensure that healthcare software systems adhere to HIPAA regulations, focusing on critical areas such as:
- Risk assessment
- Access control
- Data encryption
Case studies, including those related to hybrid evaluation methods, underscore that entities proactively addressing these regulatory challenges can avoid costly breaches and enhance operational preparedness. Furthermore, the rapid pace of technological advancement necessitates continuous updates to QA software processes to prevent regulatory gaps. As the landscape evolves, QA teams must remain vigilant, adapting their strategies to ensure that their practices not only meet current standards but also anticipate future regulatory changes.
Implement Tailored QA Strategies for Compliance
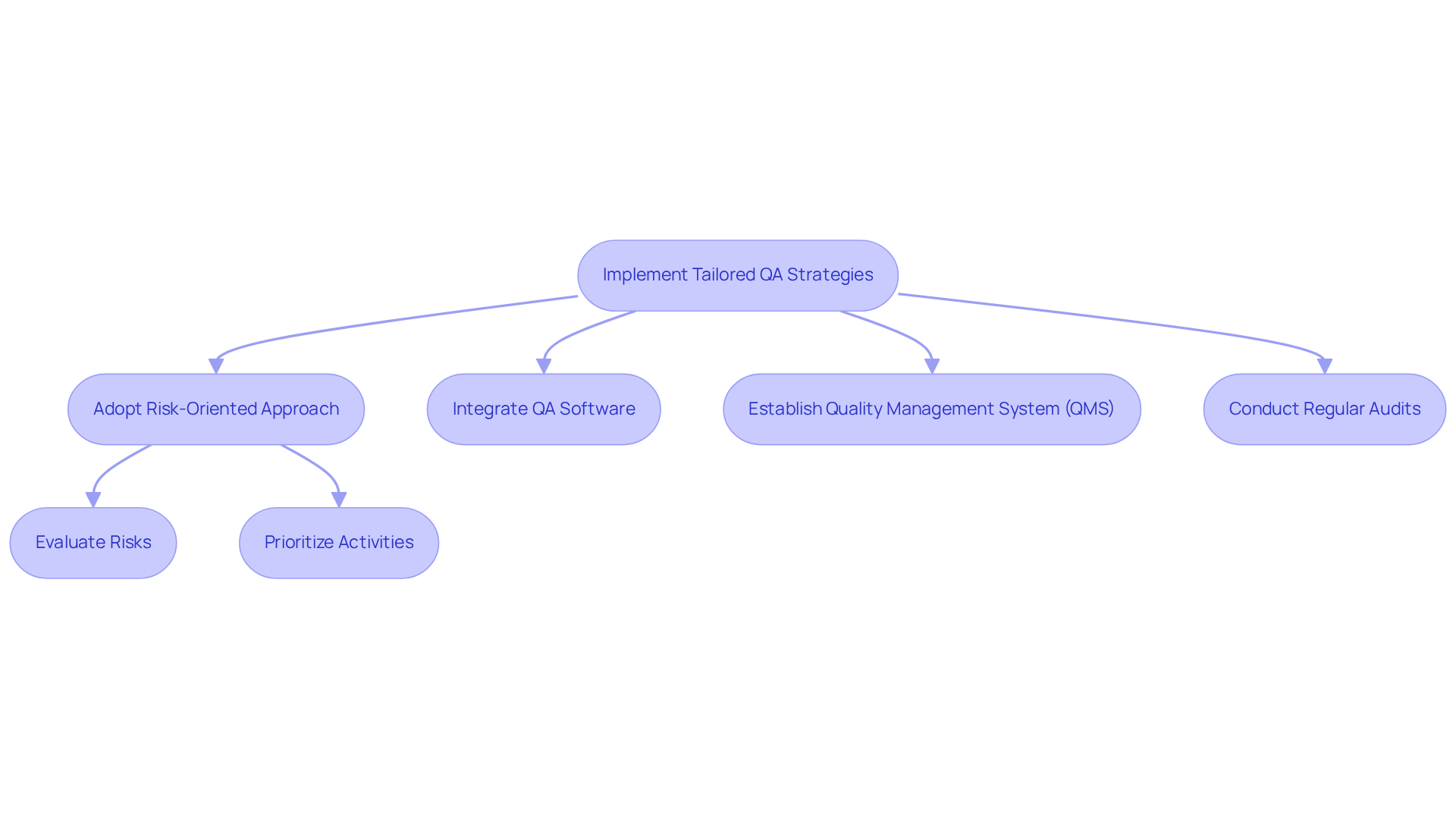
To effectively implement QA software strategies tailored for regulations, organizations must adopt a risk-oriented approach to assessment. This method prioritizes evaluation activities in QA software based on the potential consequences of failures on compliance and operational integrity. The integration of QA software significantly enhances efficiency and accuracy, facilitating continuous verification of regulatory requirements. For instance, in the financial sector, tools such as Selenium and TestComplete can automate the testing of transaction processing systems, ensuring compliance with regulations like the Payment Card Industry Data Security Standard (PCI DSS).
Furthermore, establishing a robust Quality Management System (QMS) that utilizes QA software, along with regular audits and feedback loops, is essential for maintaining ongoing compliance and fostering continuous improvement. This proactive approach not only mitigates risks but also strengthens the overall regulatory framework for QA software, ensuring that organizations remain resilient in a rapidly evolving regulatory landscape.
However, organizations must be cognizant of common pitfalls associated with implementing a risk-based approach, including:
- Inadequate training
- Poor communication
These issues can compromise the effectiveness of their QA software strategies. Additionally, leveraging insights from industry experts, such as Maurice L. Crescenzi, Jr., who underscores the importance of data and analytics in risk management, can further bolster the credibility of these strategies. By examining real-world examples where a risk-based approach has been successful, companies can better contextualize their QA initiatives and achieve meaningful compliance outcomes.
Emphasize Continuous Improvement in QA Practices
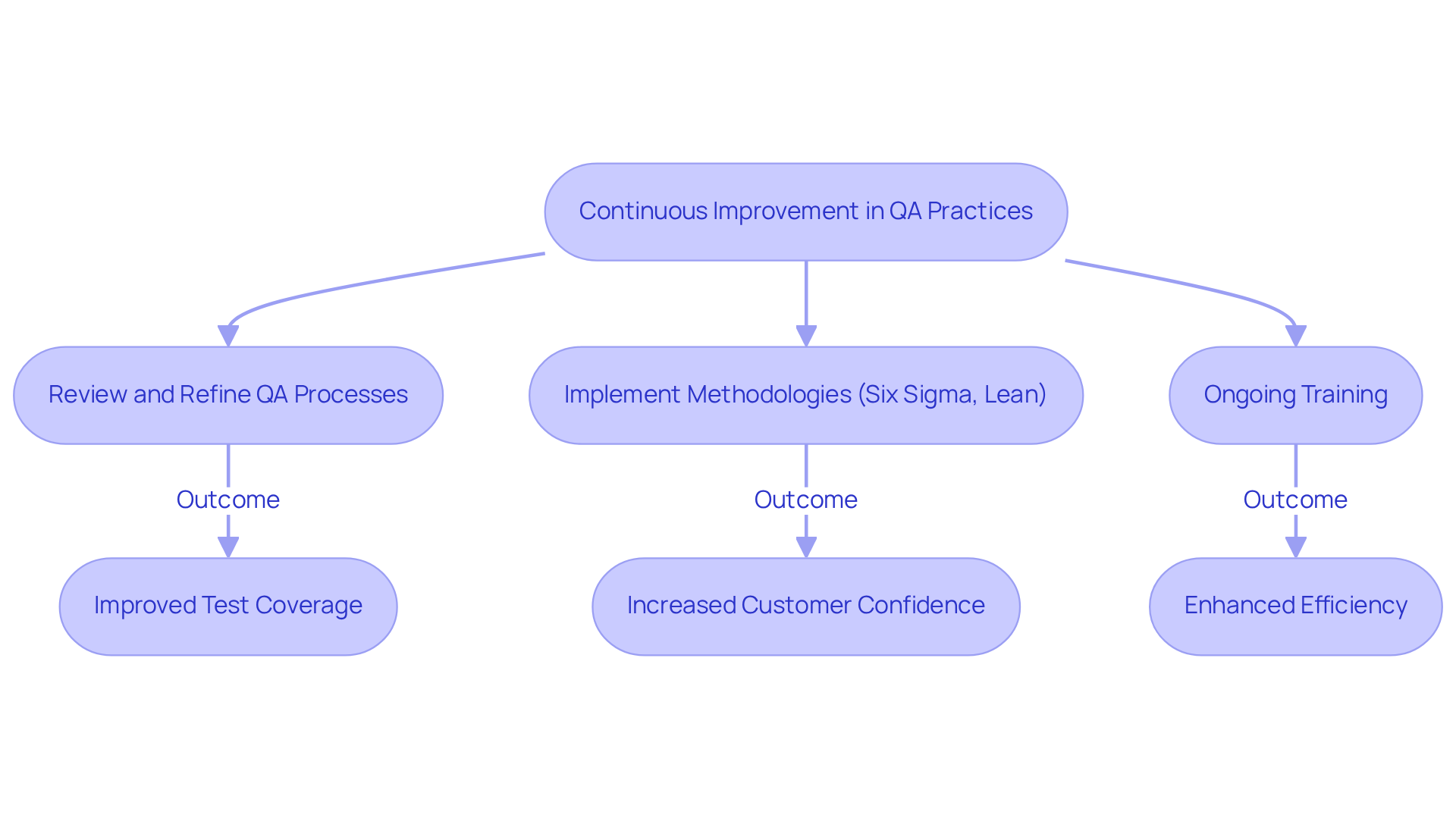
Continuous improvement in QA software practices is essential for organizations operating in regulated industries, where strict adherence to standards is critical. By regularly reviewing and refining their QA software processes, companies can adapt to evolving regulations and industry benchmarks.
Implementing methodologies such as Six Sigma and Lean significantly enhances QA software efficiency by identifying and eliminating inefficiencies. For example, a financial services company mandated 80% code coverage for all new features, which improved test coverage, reduced manufacturing problems, and increased customer confidence.
Ongoing training sessions on the latest regulatory changes for QA software teams ensure that they are well-equipped to maintain compliance. Additionally, leveraging data analytics to monitor QA performance metrics provides valuable insights into areas needing improvement, fostering a culture of excellence and accountability.
Companies that effectively integrate these methodologies often realize up to three times the ROI compared to those managing improvements separately, ultimately driving operational success in a competitive landscape.
Conclusion
Quality Assurance (QA) in regulated industries is not merely a procedural requirement; it is a critical framework that ensures compliance and operational integrity. Organizations must recognize that effective QA strategies are essential for safeguarding public safety and enhancing product effectiveness, particularly in sectors such as finance, healthcare, and pharmaceuticals. As regulatory pressures continue to mount, the commitment to quality must transition from mere policy adherence to demonstrable operational practices.
The article highlights several key strategies for successfully navigating the complexities of QA in regulated environments:
- Understanding the regulatory landscape
- Addressing challenges such as data integrity
- Implementing tailored QA strategies
Furthermore, fostering a culture of continuous improvement through methodologies like Six Sigma and Lean enhances efficiency and compliance, ultimately leading to better outcomes and increased stakeholder confidence.
In conclusion, organizations operating within regulated industries must prioritize robust QA practices to thrive in an increasingly complex regulatory framework. By embracing a proactive approach to compliance, leveraging data-driven insights, and committing to continuous improvement, companies can not only meet but exceed regulatory expectations. This commitment protects the organization and reinforces trust with customers and stakeholders, solidifying a foundation for long-term success in the marketplace.
Frequently Asked Questions
What is Quality Assurance (QA) in regulated industries?
Quality Assurance in regulated industries involves systematic methods and practices that ensure products and services meet established quality standards and comply with regulatory requirements.
Why is QA important in sectors like finance, healthcare, and pharmaceuticals?
QA is crucial in these sectors because it is not just a best practice but a legal necessity aimed at safeguarding public safety and ensuring product effectiveness.
What activities are included in QA processes?
QA processes encompass activities such as risk evaluation, procedure validation, and oversight.
Can you provide an example of QA in the financial sector?
In the financial sector, QA processes must comply with regulations like the Sarbanes-Oxley Act, which enforces stringent controls over financial reporting to enhance transparency and accountability.
What is the projected growth of the US Quality Assurance Services Market?
The US Quality Assurance Services Market is projected to grow significantly, expected to reach USD 4.3 billion by 2034, up from an estimated USD 1.4 billion in 2025.
What does Zoya Khan suggest about adherence pressure in the future?
Zoya Khan notes that by 2026, adherence pressure will transition from policy intent to operational proof, emphasizing the need for organizations to demonstrate their commitment to quality and compliance through measurable actions.
