Introduction
Regulatory compliance in medical software development is not just a legal obligation; it serves as a cornerstone of patient safety and data security, intricately woven into a framework of laws and guidelines. Developers encounter the challenge of navigating complex regulations, such as HIPAA and FDA standards, which dictate essential practices for ensuring software efficacy and compliance. Organizations must consider how to effectively integrate these requirements into their development processes while fostering innovation and meeting user needs.
This article explores four essential medical software development services that can pave the way to compliance success, offering insights into best practices that enhance both operational efficiency and patient care.
Understand Regulatory Compliance in Medical Software Development
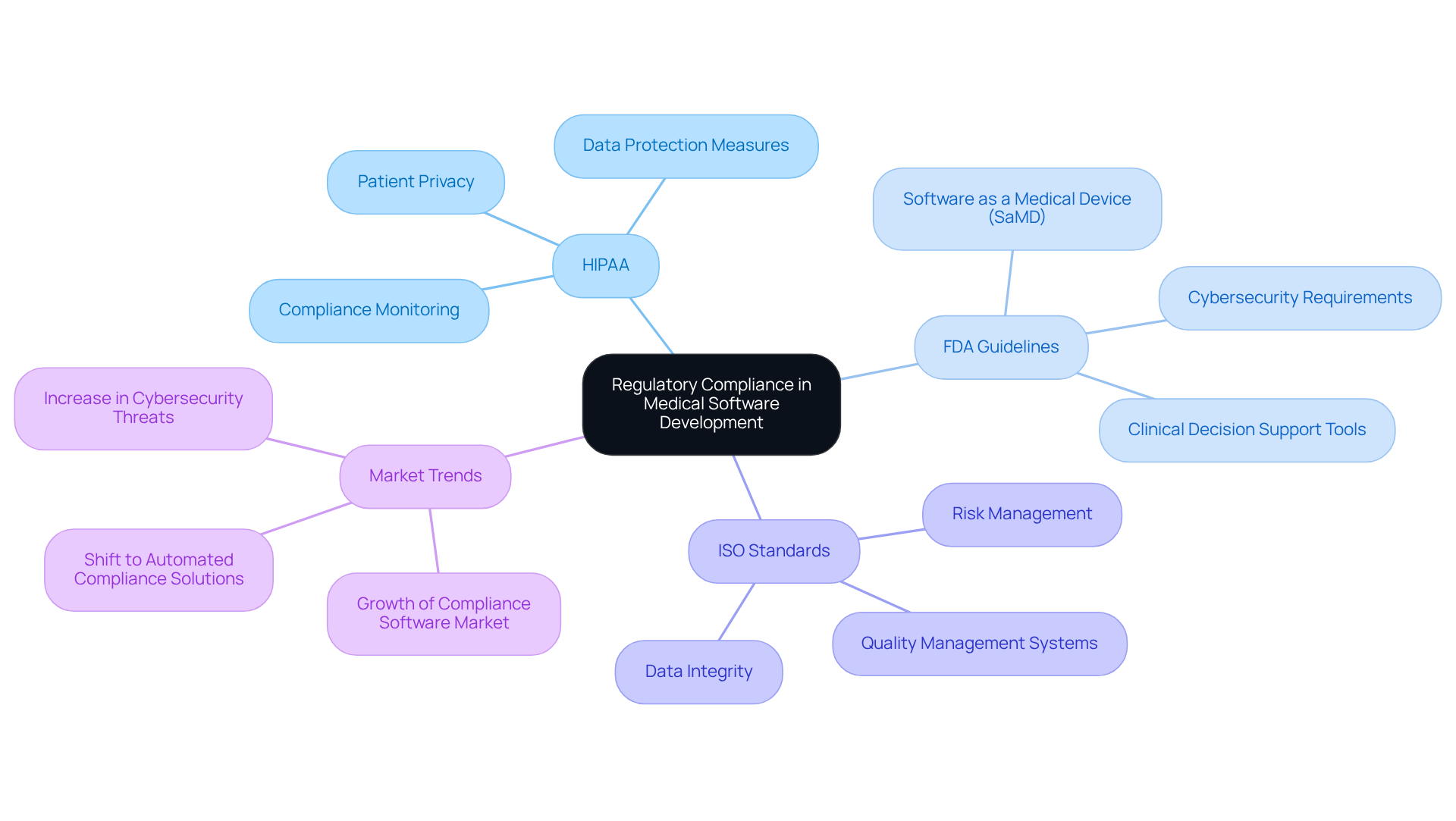
Understanding regulatory compliance is crucial for ensuring software quality, governed by a complex framework of laws and guidelines. Key regulations include:
- The Health Insurance Portability and Accountability Act, which safeguards patient privacy.
- The Food and Drug Administration regulations.
- Adherence to international standards.
Developers must familiarize themselves with these regulations from the outset to avoid costly redesigns and ensure compliance.
Engaging with legal experts can provide vital insights into the specific regulations relevant to your application. This collaboration streamlines the development process and mitigates risks associated with non-compliance. The influence of HIPAA on medical program development is significant, as it mandates stringent data protection measures that must be integrated into the program’s architecture and functionality.
Regular updates to regulations necessitate continuous education and adaptation. Therefore, it is imperative for developers to remain informed about the latest changes to maintain compliance and enhance the software’s market readiness.
Gather Requirements and Define User Needs Effectively
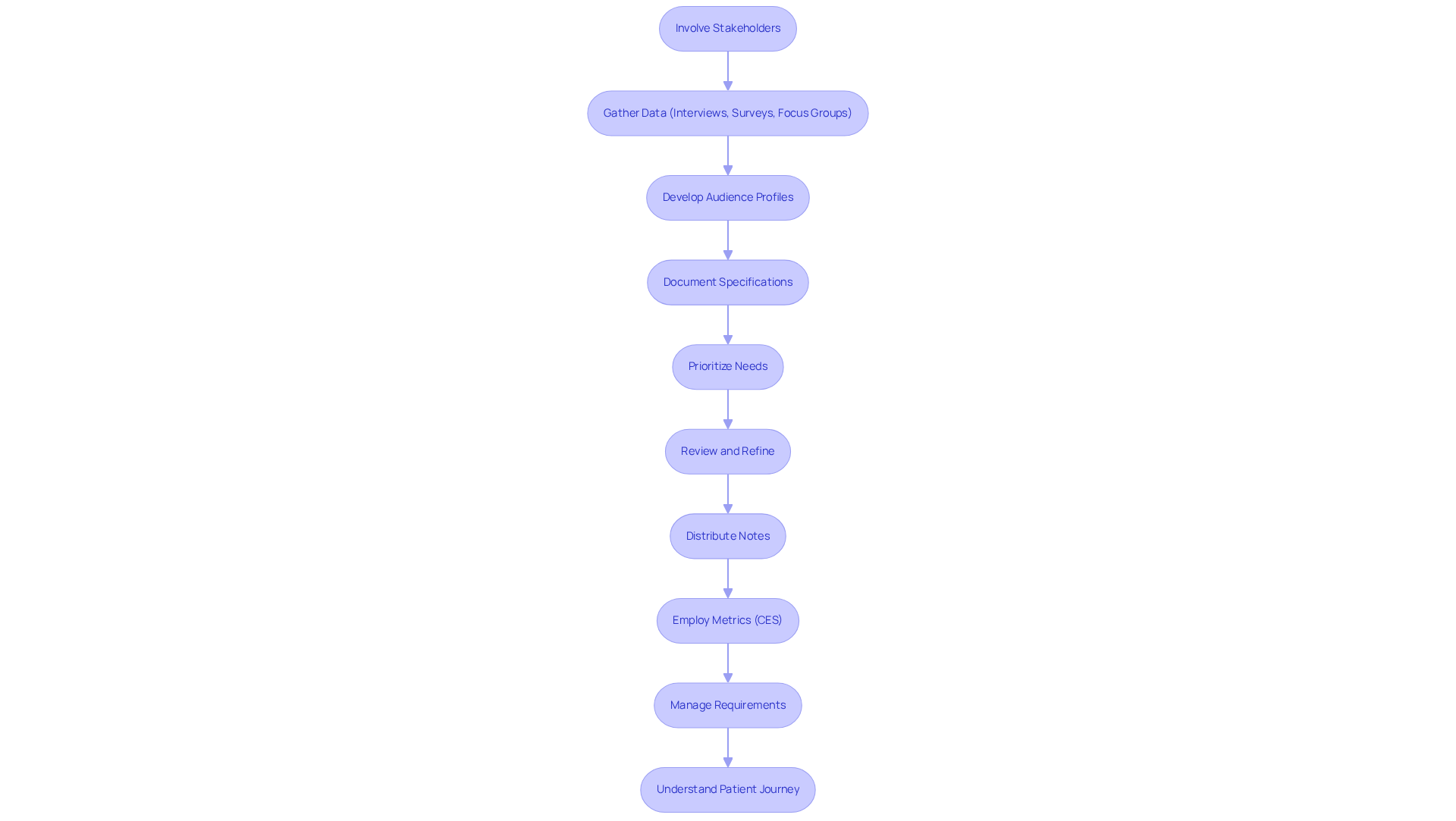
Successful needs collection hinges on the active involvement of stakeholders, including healthcare providers, patients, and end-users, to accurately grasp their needs and expectations. Methods such as interviews, surveys, and focus groups facilitate the gathering of qualitative data that reflects participant viewpoints. Additionally, feedback loops are essential.
It is crucial to meticulously document these specifications and prioritize them based on their potential impact on usability and functionality. Regularly reviewing and refining these criteria throughout the development lifecycle ensures that the final product aligns with client needs and adheres to regulatory expectations. Furthermore, distributing organized notes to stakeholders after meetings fosters transparency and ensures consensus on expectations.
Employing metrics like the Customer Effort Score (CES) offers a quantitative measure of engagement effectiveness. Continuous management of project scope to prevent scope creep and ensure that the project remains focused on client needs is also essential for enhancing individual experiences and should be integrated into discussions regarding needs collection.
Implement Rigorous Testing and Validation Protocols
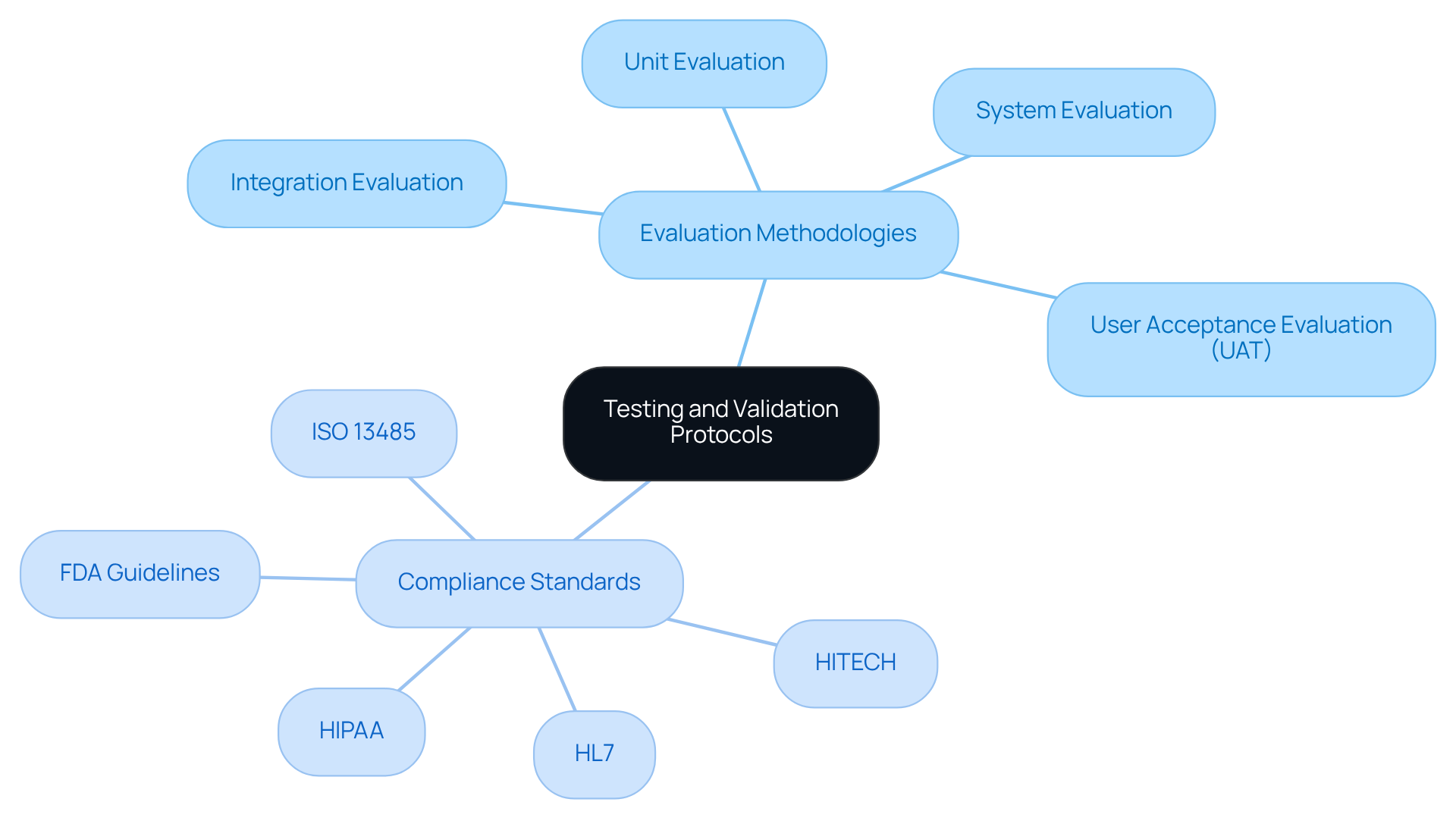
Establishing strict evaluation and validation procedures is essential for ensuring that software meets both functional and regulatory standards. This encompasses various assessment methodologies, including:
- Unit evaluation
- Integration evaluation
- System evaluation
- User acceptance evaluation (UAT)
Compliance evaluation against established standards, such as FDA regulations, is particularly crucial as it verifies that software adheres to necessary legal and ethical standards. For example, ensure adherence to HIPAA guidelines, which are vital for protecting patient data and maintaining confidentiality. Furthermore, automated testing tools, while manual assessments provide valuable insights into user experience and usability.
A clearly outlined testing strategy is critical, specifying the assessment process, success criteria, and documentation requirements. Regular audits and evaluations of these protocols are necessary to uphold compliance and continuously improve quality. As noted by ImpactQA, “Rigorous testing of healthcare applications is thus essential, not merely a best practice but a critical safeguard for patient safety and operational efficiency.” Industry leaders emphasize that testing and validation is a fundamental aspect of software development, as it not only protects patients but also enhances operational efficiency.
Ensure Continuous Improvement and Post-Launch Support
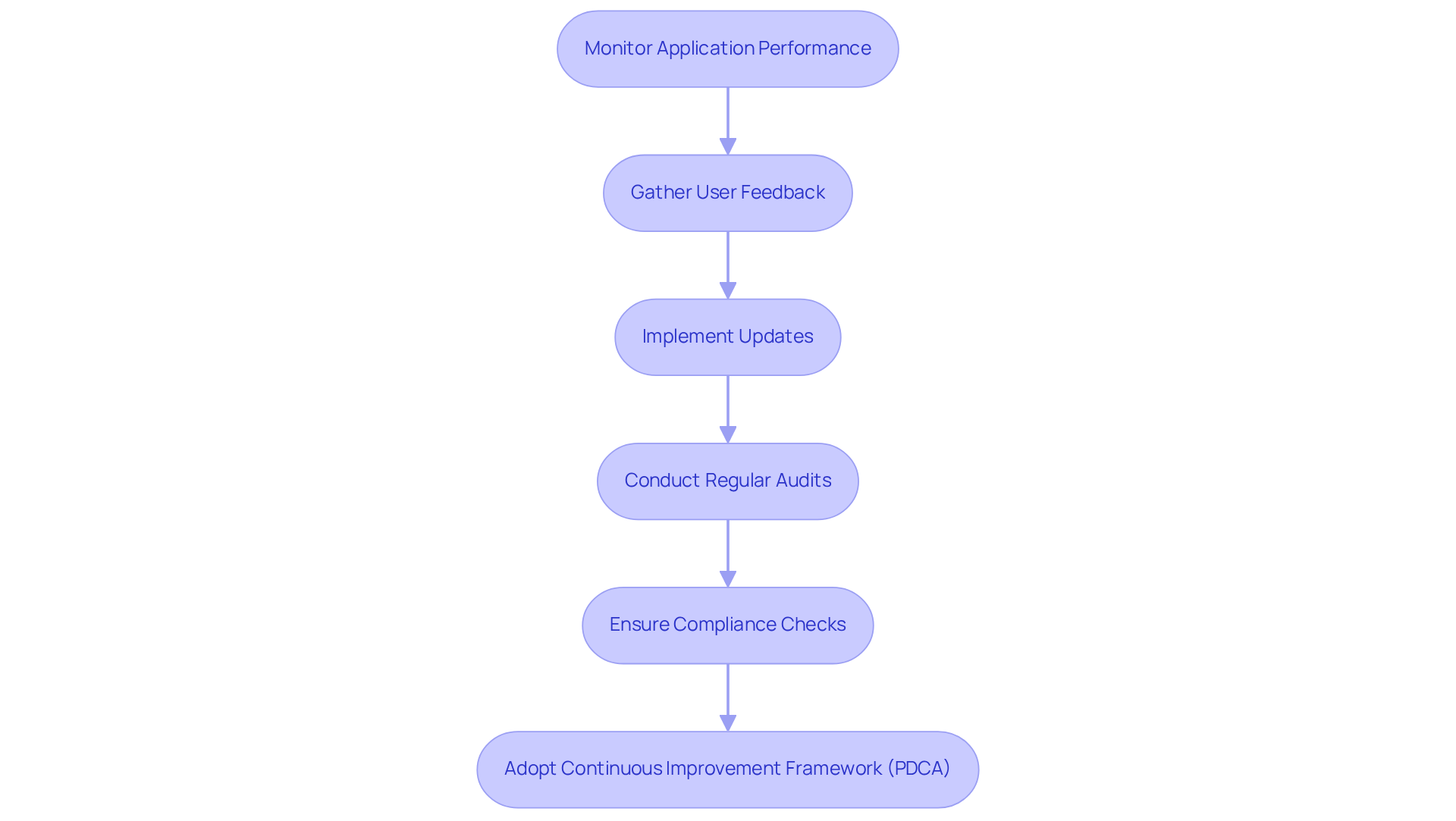
Post-launch assistance and ongoing enhancement are critical for the sustained success of medical software. This process encompasses user training, gathering feedback from users, and implementing updates. Establishing a robust feedback loop with clients regarding software performance yields vital insights into application usability and effectiveness, enabling developers to make informed improvements.
Regular updates are necessary to ensure that the system remains aligned with changing regulations, thereby safeguarding both user safety and organizational integrity. By adopting a continuous improvement model, such as Plan-Do-Check-Act (PDCA), organizations can systematically identify and resolve issues, enhancing overall performance. For example, organizations that have implemented LEAN methodologies have reported significant reductions in pre-op testing times, decreasing from 135 minutes to just 35 minutes, which underscores the effectiveness of these practices.
Moreover, as emphasized by Gary Yates, M.D., employee engagement is essential for retaining staff and attracting talent, ultimately contributing to the long-term viability of the organization.
Conclusion
Regulatory compliance stands as the cornerstone of successful medical software development, ensuring that applications not only adhere to legal standards but also prioritize patient safety and data integrity. By comprehensively understanding and integrating essential regulations such as HIPAA, FDA guidelines, and ISO standards from the outset, developers can effectively navigate the complexities of compliance and avert costly pitfalls in the future.
This article has explored key strategies for achieving compliance success. Engaging stakeholders for accurate requirements gathering, implementing rigorous testing and validation protocols, and ensuring continuous improvement alongside post-launch support are all critical components. These practices not only align software with regulatory expectations but also enhance user experience and operational efficiency.
As the landscape of medical software development continues to evolve, the significance of remaining informed about regulatory changes and best practices cannot be overstated. Embracing a proactive approach to compliance will safeguard patient welfare and position organizations for long-term success in the healthcare industry. By prioritizing these essential services, developers can contribute to a safer, more efficient healthcare environment that ultimately benefits all stakeholders involved.
Frequently Asked Questions
Why is regulatory compliance important in medical software development?
Regulatory compliance is crucial for ensuring patient safety and data security, governed by a complex framework of laws and guidelines.
What are the key regulations that govern medical software development?
Key regulations include the Health Insurance Portability and Accountability Act (HIPAA), the FDA’s guidelines for software classified as a medical device (SaMD), and adherence to ISO standards for quality management systems.
How can developers ensure compliance with regulations from the outset?
Developers should familiarize themselves with relevant regulations early in the development process to avoid costly redesigns and ensure compliance.
What role do compliance specialists and legal consultants play in medical software development?
Engaging with compliance specialists and legal consultants provides vital insights into specific regulations and streamlines the development process while mitigating risks associated with non-compliance.
How does HIPAA influence medical program development?
HIPAA mandates stringent data protection measures that must be integrated into the program’s architecture and functionality.
Why is it important for developers to stay updated on regulatory changes?
Regular updates to HIPAA and FDA guidelines necessitate continuous education and adaptation to maintain compliance and enhance the software’s market readiness.
List of Sources
- Understand Regulatory Compliance in Medical Software Development
- Healthcare Compliance Software Market Size & Share Report, 2032 (https://gminsights.com/industry-analysis/healthcare-compliance-software-market)
- htfmarketinsights.com (https://htfmarketinsights.com/report/4393755-health-it-compliance-and-regulations-market)
- coherentmarketinsights.com (https://coherentmarketinsights.com/industry-reports/healthcare-compliance-management-software-market)
- hipaajournal.com (https://hipaajournal.com/healthcare-regulatory-compliance)
- FDA Digital Health Guidance: 2026 Requirements Overview | IntuitionLabs (https://intuitionlabs.ai/articles/fda-digital-health-technology-guidance-requirements)
- Gather Requirements and Define User Needs Effectively
- Successful Requirements Gathering for Healthcare Digital Strategy (https://phase2.io/blog/successful)
- Stakeholder Engagement Effectiveness Statistics (https://zoetalentsolutions.com/stakeholder-engagement-effectiveness)
- Stakeholders’ engagement for improved health outcomes: a research brief to design a tool for better communication and participation – PMC (https://pmc.ncbi.nlm.nih.gov/articles/PMC11979262)
- 12 Best Quotes on the Future of Healthcare (https://klara.com/blog/12-best-quotes-on-the-future-of-healthcare)
- 20 hospital execs’ most thought-provoking quotes on health IT in 2021 – Becker’s Hospital Review | Healthcare News & Analysis (https://beckershospitalreview.com/healthcare-information-technology/innovation/20-hospital-execs-most-thought-provoking-quotes-on-health-it-in-2021)
- Implement Rigorous Testing and Validation Protocols
- healthcareitleaders.com (https://healthcareitleaders.com/blog/insights-from-healthcare-innovators)
- Healthcare Software Testing Services (https://impactqa.com/blog/what-makes-testing-healthcare-software-essential-for-safe-and-high-performing-platforms)
- Utilizing statistical methodologies for efficient verification and validation of medical devices industry designs. (https://linkedin.com/pulse/utilizing-statistical-methodologies-rz8cf)
- forbes.com (https://forbes.com/sites/danmunro/2013/07/09/top-10-quotes-from-harvards-first-forum-on-healthcare-innovation)
- 37 Quotes on Health Care and Health Tech from 2016 (https://medium.com/@r_scott_munro/37-quotes-on-health-care-and-health-tech-from-2016-e34576edfc1f)
- Ensure Continuous Improvement and Post-Launch Support
- These 6 healthcare leaders say quality improvement is an organization-wide effort and a cultural imperative (https://fiercehealthcare.com/healthcare/6-inspiring-quotes-improving-quality-from-6-healthcare-leaders)
- The benefits of effective post-market surveillance for medical devices (https://custom-medical.com/en/knowledge/the-benefits-of-effective-post-market-surveillance-for-medical-devices)
- Inspiring Quotes for Continuous Improvement (https://kpifire.com/blog/quotes-about-continuous-improvement)
- osfinnovation.org (https://osfinnovation.org/about-osf-innovation/case-studies/continuous-improvement-lean)
