Introduction
In the evolving landscape of healthcare, the need for innovative medical devices is increasingly critical. This article examines ten leading medical device product development companies. These companies are shaping the future of healthcare and setting standards for quality, compliance, and user-centric design.
Navigating regulatory complexities poses significant challenges for companies in the healthcare sector. As the industry navigates challenges such as regulatory complexities and the integration of cutting-edge technologies, these companies are delivering solutions that enhance patient care and safety.
Neutech: Pioneering AI-Driven Engineering Solutions for Medical Devices
Neutech, Inc. stands out in healthcare product development due to its innovative AI-native engineering solutions. The company excels in identifying and nurturing junior talent through its proprietary Residency Program, equipping engineers with deep technical skills and advanced domain knowledge. This training model enables Neutech to deliver high-quality, compliant healthcare products while addressing critical industry challenges, including regulatory adherence and timely market entry.
Neutech’s engineers often integrate into client teams, addressing the common struggle to find reliable engineering support for complex healthcare projects. Our client engagement process begins with a free consultation to understand your needs, followed by the selection of suitable candidates and a structured onboarding process. This commitment ensures that clients not only keep pace with industry changes but also lead in innovation, empowering them to thrive in an evolving healthcare landscape.
Yalantis: Innovating Medical Device Development with User-Centric Design
Yalantis is at the forefront of medical device product development companies, addressing the complexities of user-focused design principles that enhance usability and effectiveness. Incorporating end-user feedback throughout the development process ensures that Yalantis’s healthcare products meet regulatory standards while providing an optimal user experience. Their method includes rigorous testing and validation stages, which are essential for confirming that equipment is safe and effective in practical applications.
The mHealth market is expanding at a compound annual growth rate of nearly 11% and is projected to exceed $300 billion by 2030. Yalantis’s commitment to user experience and its ONC Health IT Certification position it as a formidable competitor among medical device product development companies in the healthcare equipment sector, meeting the critical demand for user-friendly tools that improve patient outcomes.
Benchmark Electronics: Comprehensive Manufacturing Solutions for Medical Devices
In the rapidly evolving healthcare equipment sector, Benchmark Electronics distinguishes itself through its unwavering commitment to quality and compliance. With over 40 years of experience, the company offers comprehensive manufacturing solutions that encompass design engineering, advanced manufacturing, and lifecycle management. Their facilities are adept at managing Class I, II, and III health equipment, ensuring adherence to stringent regulatory standards.
Recent industry data indicates that the healthcare equipment manufacturing sector is projected to reach a market size of $58.0 billion by 2026, with a revenue growth rate of 1.7% CAGR from 2021 to 2026. This growth underscores the critical need for manufacturers to prioritize quality compliance in this rapidly expanding field. Benchmark leverages advanced technologies and a skilled workforce to deliver healthcare products that enhance patient safety and effectiveness.
Industry leaders emphasize that maintaining strict quality standards is essential for success, particularly as healthcare instruments become more complex and regulatory demands intensify. Charles Cohen, President, noted that “manufacturers that embrace innovation, communicate openly, push boundaries, and develop strong customer relationships will be positioned for success in 2026.”
Moreover, the level of competition in the Medical Device Manufacturing industry in the United States is high and increasing, with the impact of import tariffs presenting significant challenges. As Benchmark continues to innovate and adapt, its commitment to quality compliance remains central to its operations. This dedication solidifies the company’s reputation as a trusted partner in the healthcare equipment sector.
DeviceLab: Ensuring Compliance and Market Readiness in Medical Device Development
DeviceLab is a leader in regulatory compliance and product readiness for medical devices, addressing the challenges companies face in this complex landscape. Their dedication to ISO 13485 certification ensures that all processes conform to international quality management standards, a vital element for obtaining approval.
With the FDA’s Quality Management System Regulation (QMSR) set to take effect on February 2, 2026, DeviceLab’s structured development process includes thorough documentation, risk assessment, and validation, ensuring compliance with regulatory mandates while addressing market needs.
Additionally, starting in April 2026, the introduction of yearly registration fees for devices in the UK adds complexity that DeviceLab helps clients navigate. This expertise positions DeviceLab as an invaluable ally for companies aiming to introduce innovative healthcare products, particularly in a context where compliance with quality standards is crucial.
Industry experts note that ‘Achieving ISO 13485 certification enhances customer confidence and credibility, demonstrating compliance with government legislation and industry requirements,’ reinforcing DeviceLab’s commitment to quality.
In an environment where regulatory compliance is paramount, DeviceLab’s expertise can be the difference between success and failure for innovative healthcare products.
Velentium Medical: Engineering Excellence in Medical Device Safety
Velentium Medical exemplifies engineering excellence through its unwavering commitment to safety in healthcare tool development. Medical device product development companies provide comprehensive services that encompass the design, development, and manufacturing of Class II and III medical products, ensuring strict adherence to FDA regulations.
In light of the FDA’s updated cybersecurity guidance, Velentium integrates cybersecurity measures throughout the product development lifecycle, addressing critical concerns regarding product security. As Phil Englert, Director of Medical Technology Security, states, “Today, with connected items generating vast volumes of clinical data and playing a central role in diagnosis and treatment, securing them has become essential not only for data protection but also for ensuring care delivery itself.”
This proactive approach enhances compliance with regulations and significantly improves patient outcomes, positioning Velentium as a trusted partner for medical device product development companies that aim to uphold safety standards.
With healthcare instruments averaging 6.2 vulnerabilities per unit, the prevalence of vulnerabilities in healthcare instruments highlights the urgent need for robust cybersecurity measures. Moreover, the partnership between healthcare providers and manufacturers is crucial in preserving equipment security throughout its lifecycle, strengthening the mutual obligation in protecting individual safety.
Addressing these vulnerabilities is not just a regulatory requirement; it is a fundamental aspect of safeguarding patient care.
Kapstone Medical: Enhancing Patient Care Through Innovative Device Solutions
In a rapidly evolving healthcare landscape, Kapstone Medical is dedicated to enhancing patient outcomes through innovative medical equipment solutions. By collaborating closely with healthcare providers, they develop products tailored to specific clinical needs, ensuring that solutions are both effective and user-friendly. Their expertise spans the entire product lifecycle, from initial concept development to commercialization, enabling them to deliver solutions that significantly improve health outcomes. By prioritizing innovation and collaboration, Kapstone solidifies its leadership in healthcare while driving advancements that benefit patients and providers.
As highlighted by Thomas A. Gustafson, Ph.D., the partnership between device manufacturers and healthcare providers is essential for aligning regulatory and coverage expectations, particularly in light of the new RAPID pathway designed to accelerate Medicare coverage for breakthrough devices. Furthermore, the global healthcare technology sector, valued at approximately $456.9 billion in 2020 and anticipated to reach $695.5 billion by 2027, underscores the significance of innovation and collaboration in this swiftly growing field. These strategic partnerships not only enhance patient care but also pave the way for future innovations that could redefine healthcare delivery.
Plexus: End-to-End Product Development for Medical Devices
Plexus addresses the complexities of product development in healthcare instruments, highlighting the role of medical device product development companies in ensuring efficiency and quality are prioritized at every phase. Their comprehensive approach encompasses:
- Design
- Engineering
- Manufacturing
- Supply chain management
This enables clients to expedite their market entry while upholding rigorous compliance standards. Plexus’s expertise in regulatory compliance and commitment to innovation make them a vital partner for health technology firms aiming to enhance product offerings and streamline development processes.
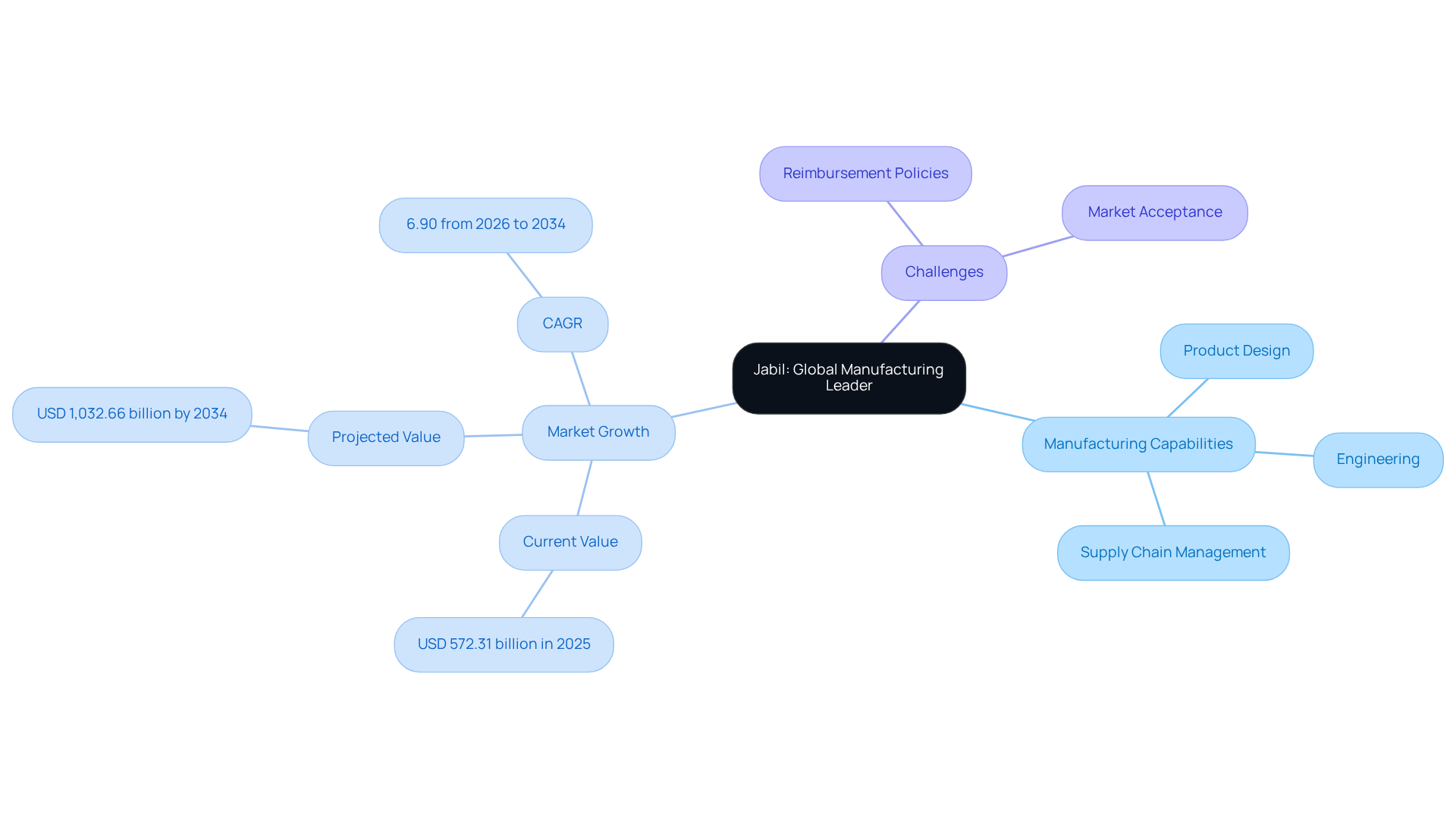
Jabil: Global Manufacturing Leader in Medical Device Solutions
Jabil stands at the forefront of manufacturing solutions in the healthcare industry, offering a comprehensive suite of services that includes product design, engineering, and supply chain management. With over 30 ISO 13485-certified facilities worldwide, Jabil guarantees that every product adheres to rigorous regulatory standards. Their advanced manufacturing capabilities enable the production of a diverse range of healthcare tools, from surgical instruments to diagnostic equipment, all while maintaining a focus on quality and efficiency. Jabil’s commitment to innovation and customer satisfaction establishes it as a trusted partner for medical device product development companies.
The global healthcare equipment sector, valued at USD 572.31 billion in 2025, is projected to expand to USD 1,032.66 billion by 2034, with a CAGR of 6.90% from 2026 to 2034. This growth trajectory positions Jabil to capitalize on emerging opportunities, particularly in the in-vitro diagnostics segment, which is expected to dominate the market. However, despite its leadership, Jabil faces significant challenges in navigating reimbursement policies that hinder market acceptance in developing nations. Navigating these complexities will be crucial for Jabil as it seeks to leverage growth opportunities in a rapidly evolving market.
Vantage MedTech: Shaping Healthcare with Innovative Device Development
In an era where healthcare innovation is paramount, Vantage MedTech is at the forefront of developing advanced health equipment. The company prides itself on a group of specialized engineers committed to creating tools that significantly improve healthcare and simplify clinical processes. Vantage MedTech leverages advanced technologies to ensure its products meet and exceed regulatory compliance standards. This commitment to innovation and quality positions Vantage MedTech as a key player among medical device product development companies in the healthcare device sector, driving advancements that lead to better outcomes for individuals and greater efficiency in healthcare.
As the industry evolves, the integration of AI and robotics is expected to enhance surgical precision and reduce operation times, making 2026 a pivotal year for these technologies. The emphasis on consumer-focused healthcare advancements is transforming the landscape, with products becoming more accessible directly to individuals, thus broadening the market for remote monitoring tools and personalized health solutions.
Vantage MedTech demonstrates how specialized engineering teams can proactively address emerging healthcare trends, ensuring that their developments align with the future needs of healthcare.
Suntra MedTech Solutions: Advancing Patient Monitoring with Connected Devices
Healthcare providers often struggle with outdated monitoring methods that hinder timely patient care, making innovative solutions essential. Suntra MedTech Solutions is at the forefront of improving health monitoring through the creation of connected healthcare instruments. Their innovative solutions leverage IoT technology to provide real-time data, enhancing healthcare providers’ ability to monitor patient conditions.
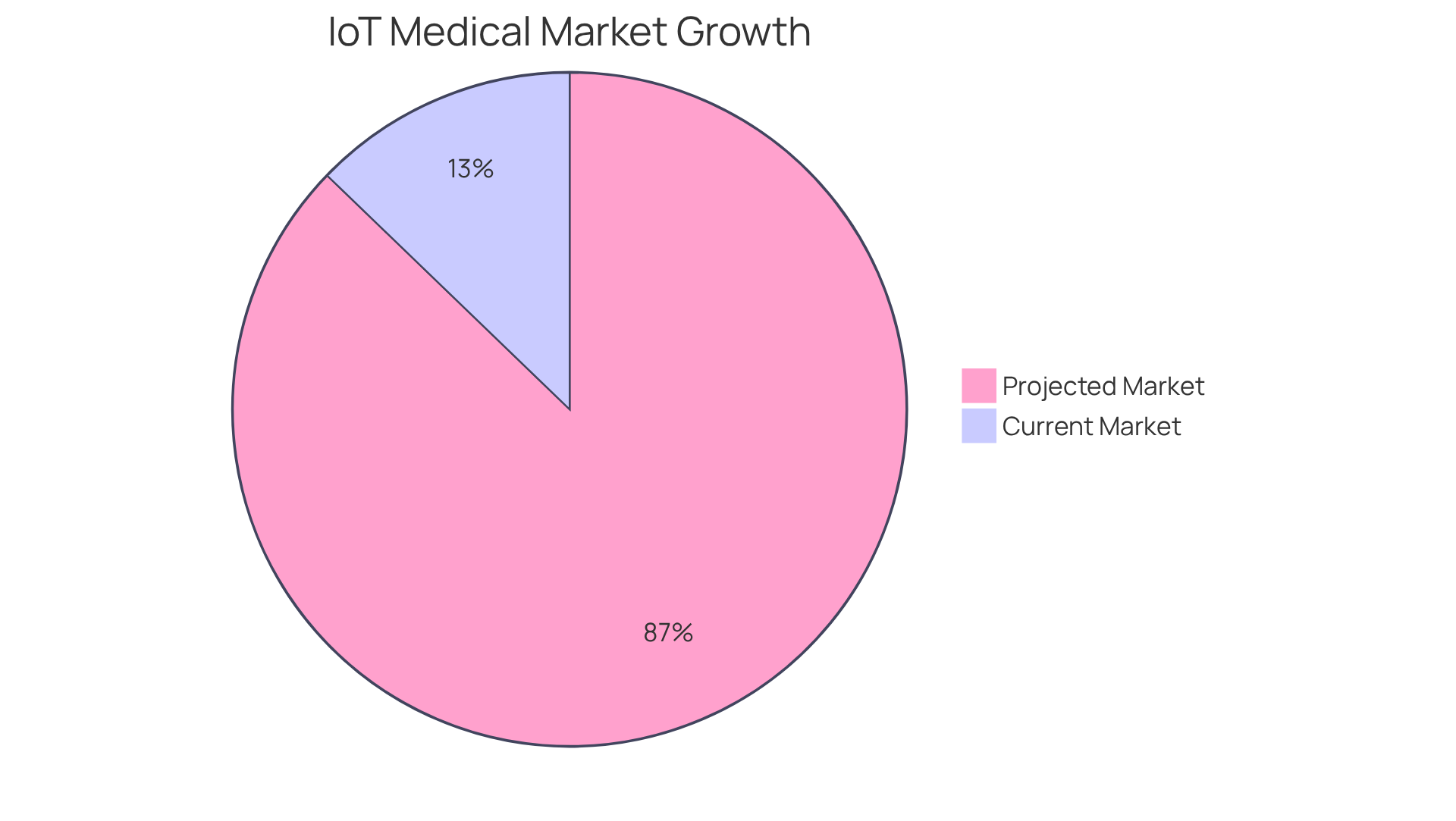
In 2026, the global IoT medical equipment market is projected to expand from USD 68.5 billion to USD 467.2 billion by 2036, with a compound annual growth rate (CAGR) of 21.2% during this timeframe, highlighting the growing significance of connectivity in healthcare. Suntra’s dedication to smooth integration with current healthcare systems guarantees that their products not only operate efficiently but also improve the overall user experience.
Suntra MedTech Solutions prioritizes regulatory compliance and focuses on healthcare professionals’ needs, crucially transforming care and enhancing health outcomes. Furthermore, as the industry evolves, the establishment of dedicated cybersecurity teams within healthcare organizations is becoming essential to protect devices, networks, and databases, ensuring the security of patient data in this increasingly connected environment.
Conclusion
The medical device industry stands at a pivotal moment, where innovation and regulatory compliance are no longer optional but essential for success. Numerous companies are leading the charge in enhancing the efficiency and safety of medical devices while addressing critical regulatory challenges. Firms such as Neutech, Yalantis, and Benchmark Electronics exemplify this commitment. By prioritizing user-centric design and integrating advanced technologies, these companies solidify their leadership in a competitive landscape.
Throughout the article, we have explored the operational strategies and unique strengths of each company, highlighting their vital roles in shaping the future of medical technology. Neutech’s AI-driven solutions, Yalantis’s focus on usability, and DeviceLab’s regulatory expertise illustrate how these firms tackle the complexities of product development. The emphasis on collaboration, quality assurance, and innovative design principles is crucial as the healthcare sector continues to expand and adapt to new demands.
With 2026 on the horizon, the pressure for innovation and compliance intensifies, challenging companies to adapt swiftly. Stakeholders must recognize the importance of partnering with leading firms that prioritize quality and safety in their product offerings. Failure to embrace these advancements could hinder patient care and stall progress in healthcare delivery. The trajectory of medical device development will depend on the ability of these companies to navigate challenges and set new benchmarks for excellence.
Frequently Asked Questions
What is Neutech known for in the healthcare product development sector?
Neutech is recognized for its innovative AI-native engineering solutions that focus on developing high-quality, compliant healthcare products while addressing industry challenges such as regulatory adherence and timely market entry.
How does Neutech nurture junior engineering talent?
Neutech has a proprietary Residency Program that equips engineers with deep technical skills and advanced domain knowledge, enabling them to contribute effectively to healthcare product development.
What is the client engagement process at Neutech?
Neutech’s client engagement process starts with a free consultation to understand client needs, followed by selecting suitable candidates and a structured onboarding process to ensure effective integration into client teams.
What design principles does Yalantis focus on in medical device development?
Yalantis emphasizes user-centric design principles that enhance usability and effectiveness, incorporating end-user feedback throughout the development process to ensure regulatory compliance and optimal user experience.
What is the projected growth of the mHealth market?
The mHealth market is expected to grow at a compound annual growth rate of nearly 11% and is projected to exceed $300 billion by 2030.
What distinguishes Benchmark Electronics in the healthcare equipment sector?
Benchmark Electronics is distinguished by its commitment to quality and compliance, offering comprehensive manufacturing solutions that include design engineering, advanced manufacturing, and lifecycle management for various classes of health equipment.
What is the projected market size for healthcare equipment manufacturing by 2026?
The healthcare equipment manufacturing sector is projected to reach a market size of $58.0 billion by 2026, with a revenue growth rate of 1.7% CAGR from 2021 to 2026.
What challenges does the medical device manufacturing industry face?
The industry faces high competition and challenges related to import tariffs, which impact operational costs and market dynamics.
What is the importance of quality compliance in medical device manufacturing?
Maintaining strict quality standards is essential for success, particularly as healthcare instruments become more complex and regulatory demands increase.
List of Sources
- Neutech: Pioneering AI-Driven Engineering Solutions for Medical Devices
- The State of AI in Healthcare: Market Growth and Key Stats (https://patentpc.com/blog/the-state-of-ai-in-healthcare-market-growth-and-key-stats)
- Built to comply: How AI is reshaping the medical device landscape (https://todaysmedicaldevelopments.com/news/built-comply-how-ai-reshaping-medical-device-landscape)
- CMS, FDA Announce RAPID Coverage Pathway for Faster Medical Device Access (https://mpo-mag.com/breaking-news/cms-fda-announce-rapid-coverage-pathway-for-faster-medical-device-access)
- AI in Healthcare Statistics 2025: Revealing the Future of Medicine (https://sqmagazine.co.uk/ai-in-healthcare-statistics)
- FDA’s AI Medical Device List: Stats, Trends & Regulation | IntuitionLabs (https://intuitionlabs.ai/articles/fda-ai-medical-device-tracker)
- Yalantis: Innovating Medical Device Development with User-Centric Design
- TOP 4mental health app developers ranked by user growth (https://infomeddnews.com/top-4-mental-health-app-developers-ranked-by-user-growth)
- FDA requests comment on performance of AI-enabled medical devices (https://healthcarefinancenews.com/news/fda-requests-comment-performance-ai-enabled-medical-devices)
- How to Build a High-Quality IoT Medical Device (MIoT) (https://yalantis.com/blog/medical-iot-solutions)
- Why many existing medical devices fall short of the FDA’s new cybersecurity standards (https://todaysmedicaldevelopments.com/news/why-many-existing-medical-devices-fall-short-fda-new-cybersecurity-standards)
- FDA seeks industry feedback on AI medical device safety monitoring (https://medicaldevice-network.com/news/fda-seeks-industry-feedback-on-ai-medical-device-safety-monitoring)
- Benchmark Electronics: Comprehensive Manufacturing Solutions for Medical Devices
- Benchmark Electronics Q1 2026 slides: upgraded outlook on AI, medical strength By Investing.com (https://investing.com/news/company-news/benchmark-electronics-q1-2026-slides-upgraded-outlook-on-ai-medical-strength-93CH-4647442)
- Benchmark Reports First Quarter 2026 Results and Raises Full Year Outlook (https://businesswire.com/news/home/20260429613284/en/Benchmark-Reports-First-Quarter-2026-Results-and-Raises-Full-Year-Outlook)
- 2026 Forecast: Medical manufacturing industry outlook (https://todaysmedicaldevelopments.com/article/2026-forecast-todays-medical-developments-industry-outlook)
- Medical Device Manufacturing in the US Industry Analysis, 2026 (https://ibisworld.com/united-states/industry/medical-device-manufacturing/764)
- Benchmark Electronics Q1 Earnings Call Highlights (https://marketbeat.com/instant-alerts/benchmark-electronics-q1-earnings-call-highlights-2026-04-29)
- DeviceLab: Ensuring Compliance and Market Readiness in Medical Device Development
- Top Medical Device Regulations to Watch in 2026 (https://xtalks.com/top-medical-device-regulations-to-watch-in-2026-4549)
- The Importance of ISO 13485 Certification to Medical Device Manufacturing | Smithers (https://smithers.com/resources/2022/december/importance-of-iso-13485-certification)
- 5 Facts About ISO 13485 Every Medical Device Manufacturer Needs to Know (https://kapstonemedical.com/resource-center/blog/5-facts-about-iso-13485)
- 8 Benefits of ISO 13485 Certification for Medical Device Manufacturing (https://amtivo.com/us/resources/insights/iso-13485-medical-device-certification-benefits)
- Velentium Medical: Engineering Excellence in Medical Device Safety
- Diving into FDA’s Cybersecurity Expectations (https://mddionline.com/digital-health/diving-into-fda-s-cybersecurity-expectations)
- MedTech and Medical Device Cybersecurity News | MedTech Dive (https://medtechdive.com/topic/cybersecurity)
- FDA Tightens Its Medical Device Cybersecurity Guidance (https://fedtechmagazine.com/article/2026/03/fda-tightens-its-medical-device-cybersecurity-guidance-perfcon)
- FDA Mandates Cybersecurity in Medical Device Development | Velentium Medical posted on the topic | LinkedIn (https://linkedin.com/posts/velentiummedical_medtechcybersecurity-medicaldevicedevelopment-activity-7434621659208245249-wBDg)
- Medical Device Breach Statistics 2026 Report (https://ordr.net/medical-device-breach-statistics)
- Kapstone Medical: Enhancing Patient Care Through Innovative Device Solutions
- Blog – Kapstone Medical (https://kapstonemedical.com/resource-center/blog)
- Strategic partnership created to support early medical device innovation (https://todaysmedicaldevelopments.com/news/strategic-partnership-created-support-early-medical-device-innovation)
- U.S. Health and Human Services Tries Again To Improve Availability of Innovative Devices in Medicare | Advisories | Arnold & Porter (https://arnoldporter.com/en/perspectives/advisories/2026/04/hhs-tries-again-to-improve-availability-of-innovative-devices-in-medicare)
- Medical Technology and Innovation Statistics and Facts (2026) (https://media.market.us/medical-technology-and-innovation-statistics)
- New Plan Could Speed Medicare Coverage for Innovative Devices (https://usnews.com/news/health-news/articles/2026-04-24/new-plan-could-speed-medicare-coverage-for-innovative-devices)
- Plexus: End-to-End Product Development for Medical Devices
- News Archives (https://medicaldevice-network.com/news)
- UK and US deepen regulatory cooperation on medical devices, building on wider pharmaceutical partnership (https://gov.uk/government/news/uk-and-us-deepen-regulatory-cooperation-on-medical-devices-building-on-wider-pharmaceutical-partnership)
- 2026 Medical Device Manufacturing: Optimism Amid Uncertainty (https://mddionline.com/manufacturing/2026-medical-device-manufacturing-outlook-optimism-grows-despite-lingering-uncertainty)
- Leading Companies Reinforce Their Standing in the Medical Device Design and Development Services Market (https://openpr.com/news/4450913/leading-companies-reinforce-their-standing-in-the-medical)
- MD&M West 2026 (https://plexus.com/events/mdm-west-2026)
- Jabil: Global Manufacturing Leader in Medical Device Solutions
- Modern Medical Technology is Shaping a Safer Future|CIO Bulletin (https://ciobulletin.com/medical-technology/usf-and-jabil-medical-technology-partnership)
- Manufacturing giant Jabil signals confidence with higher targets for 2026 (https://digitimes.com/news/a20251218VL208/jabil-2026-manufacturing-revenue-data-center.html)
- Medical Devices Market Size, Share, Global Growth Report 2034 (https://fortunebusinessinsights.com/industry-reports/medical-devices-market-100085)
- Jabil, HSE to develop diagnostic instrumentation (https://todaysmedicaldevelopments.com/news/jabil-hse-develop-diagnostic-instrumentation)
- Medical Device Industry Statistics and Market Trends Driving Global Manufacturing Growth (https://china-greetmed.com/Medical-Device-Industry-Statistics-2026)
- Vantage MedTech: Shaping Healthcare with Innovative Device Development
- 2026 Forecast: Medical manufacturing industry outlook (https://todaysmedicaldevelopments.com/article/2026-forecast-todays-medical-developments-industry-outlook)
- Vantage MedTech names new CEO, CCO and executive chair (https://medicaldesignandoutsourcing.com/vantage-medtech-new-ceo-cco-executive-chair)
- 6 key quotes about innovation’s role in healthcare – Becker’s Hospital Review | Healthcare News & Analysis (https://beckershospitalreview.com/healthcare-information-technology/innovation/6-key-quotes-about-innovation-s-role-in-healthcare)
- 12 Best Quotes on the Future of Healthcare (https://modmed.com/resources/blog/12-best-quotes-on-the-future-of-healthcare)
- Suntra MedTech Solutions: Advancing Patient Monitoring with Connected Devices
- Connected Medical Devices – Suntra MedTech Solutions (https://suntramedtech.com/knowledge-center/insights/connected-medical-devices)
- Medical Device Design & Development Company Updates from Sunrise Labs (https://suntramedtech.com/knowledge-center/company-updates/c/news)
- IoMT: How Connected Medical Devices are Transforming Healthcare in 2026 (https://kandasoft.com/blog/iomt-connected-medical-devices)
- The Pulse of Innovation: How Smart Healthcare is Redefining Patient Outcomes in 2026 (https://usatoday.com/press-release/story/27470/the-pulse-of-innovation-how-smart-healthcare-is-redefining-patient-outcomes-in-2026)
- IoT Medical Devices Market Research and Global Forecast Report 2026-2036: Shift Toward Connected Healthcare Delivery and Rapid Expansion of Remote Patient Monitoring Driving Opportunities (https://globenewswire.com/de/news-release/2026/04/22/3279097/0/en/iot-medical-devices-market-research-and-global-forecast-report-2026-2036-shift-toward-connected-healthcare-delivery-and-rapid-expansion-of-remote-patient-monitoring-driving-opportu.html)
